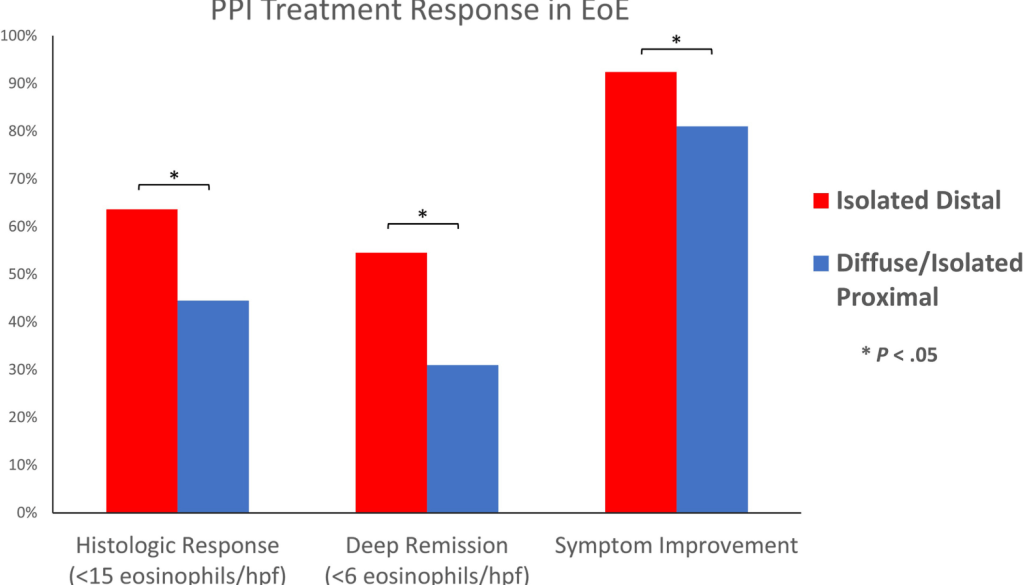
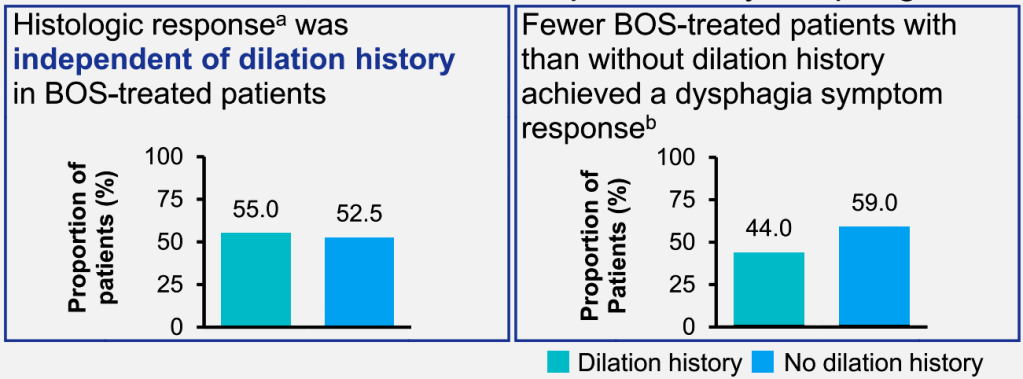
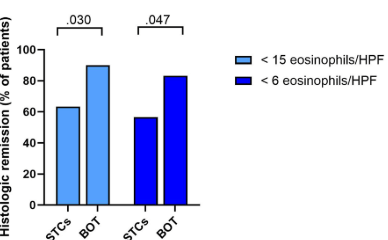
FB Murray et al. Am J Gastroenterol 2026; 121: 130-139. Loss of Response to Off-Label Swallowed Topical Corticosteroids in Eosinophilic Esophagitis Can be Overcome by a Switch to an Esophageal-Targeted Budesonide Formulation
A quick glance at this study gives the impression that off-label swallowed topical corticosteroids (olSTC) are an inferior treatment to the budesonide orodispersible tablet (BOT) as many patients who had either a non-response to olSTC achieved remission with BOT.
Methods: This study from the Swiss Eosinophilic Esophagitis Cohort Study with 340 patients (mean age 43 years) analyzed prospectively collected data. Twenty-six percent had prior olSTC nonresponse (n=66) , 16% were in remission with prior olSTC (n=44), and 58% were STC-naïve.
Here were the key results according to the authors (Figure 2):
- Histologic remission (<15 eos/hpf) was achieved in 62% who had not responded to olSTC previously and in 72% who had prior olSTC response (P=0.094)
- The authors conclude that “our results provide conclusive evidence that off-label STC cannot be translated into corticosteroid-refractory disease per se.”
Here’s the main problem with this study:
- “Most patients in Switzerland have been treated with a rather low-dose olSTC regimen (</ = to 0.5 mg per day).” Typical BOT dosing is 2 mg per day. Thus, the authors are comparing the use of 4-times the amount of budesonide in the BOT group to the olSTC non-responders.
- Their discussion states that “most patients with EoE without a response to olSTC are not truly corticosteroid-refractory but respond to an esophageal adjusted topical corticosteroid formulation…In the United States, a BOS [suspension] has been recently approved…Due to similar mechanism of action, it can by hypothesized that patients without prior response to olSTC will respond to BOS in similar rates as shown in our study.” Yet, there is no proof that the formulation made a difference in this study; the more likely explanation is that patients previously who had not responded to olSTC were under-treated.
My take:
- In patients with eosinophilic esophagitis who have not responded to topical steroids, it is important to make sure that they were prescribed the right dose and that they are actually taking the medication.
- Future studies of esophageal formulations should be compared to off-label STC using the same dose to determine if the formulations confer additional benefits.
- Getting the right dose is important for every malady.
Thanks to Ben Gold for sharing this publication with me.
Related blog posts:
- Long-Term Treatment of Eosinophilic Esophagitis with Budesonide
- Budesonide Tablet vs Off-Label Corticosteroids in Eosinophilic Esophagitis
- Orodispersible Budesonide Tablets for Eosinophilic Esophagitis
- Delivery Vehicle and Outcomes for Budesonide-Treated Eosinophilic Esophagitis
- Budesonide FDA-Approved for Eosinophilic Esophagitis
- Budesonide for Maintaining EoE Remission
- Give the Right Dose (for H pylori) -It Works Better!