NV Castelle et al. Clin Gastroenterol Hepatol 2022; 20: 465-467. Open Access: Patients With Low Drug Levels or Antibodies to a Prior Anti-Tumor Necrosis Factor Are More Likely to Develop Antibodies to a Subsequent Anti-Tumor Necrosis Factor
Design: Retrospective case-control study in 5828 patients (2171 cases, 1445 controls). Subjects needed to have consecutively orders of 2 anti-TNF therapies (infliximab (IFX) prior to adalimumab (ADM) or vice versa).
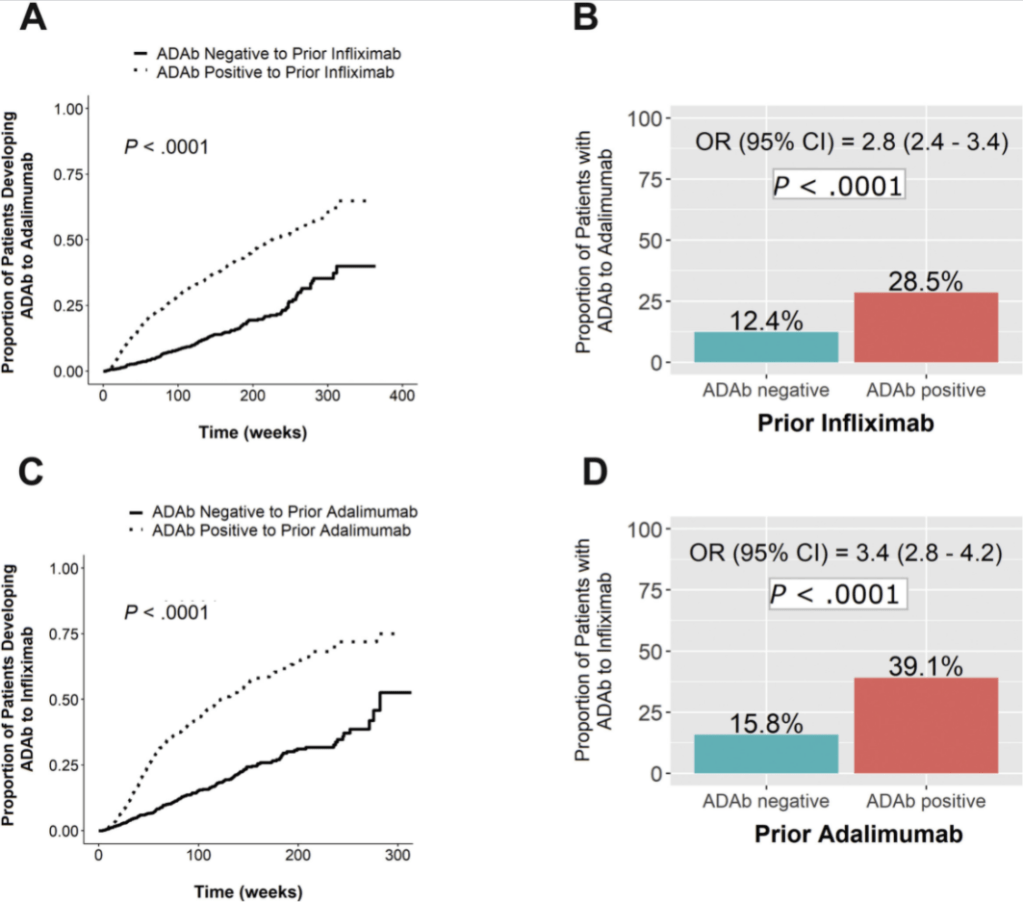
Key findings:
- Before switch from IFX to ADM, median (interquartile range) IFX serum concentrations were lower in cases versus control subjects (1.0 μg/mL [1.0–1.0] vs 11.7 μg/mL [4.2–27.1]; P < .0001).
- As noted in figure below, prior antidrug antibodies ADAb) to anti-TNF agent was associated with development of ADAb with 2nd anti-TNF agent. This risk was >2-fold higher when switching from IFX to ADM (B in Figure) and even more when switching from ADM to IFX (D in Figure)
- Increasing concentrations of ADAb to IFX were associated with higher proportions of patients developing ADAb to ADM (P < .0001). In contrast, increasing concentrations of ADAb to ADM did not result in a significantly higher proportion of patients developing ADAb to IFX.
My take:
- In my experience, many patients with subtherapeutic anti-TNF levels, even those with ADAb, can remain on initial anti-TNF with more frequent dosing and often with combination therapy. So before jumping off the initial treatment, make sure it has been optimized.
- In those who do start a 2nd anti-TNF agent, the authors recommend using combination therapy (with an immunomodulator) which “leads to lower rates of clinical failure and more favorable pharmacokinetics, compared with monotherapy.”
- “Alternatively, a strategy with optimized monotherapy using proactive TDM may be effective as well, but remains to be assessed in a prospective manner.”
Related blog posts:
- Can Therapeutic Drug Monitoring with Monotherapy Achieve Similar Results as Combination Therapy for IBD?
- Expert Consensus: New Recommendations for Therapeutic Drug Monitoring
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.
