C DiLorenzo, M Saps et al. Gastroenterol 2026; 170: 1367-1387. Open Access! Lower and Biliary Disorders of Gut–Brain Interaction: Child and Adolescent
Before reviewing this article, I wanted to point out that Dr. DiLorenzo, who was the 2025 ANMS Lifetime Achievement Award recipient, overcame great hardship to become a leader in neurogastroenterology. Elsewhere in this same issue, Wong et al (Gastroenterol 2026; 170: 1190-1204) point out that in Italy (& Spain) there is not even a word for bloating!
Key points:
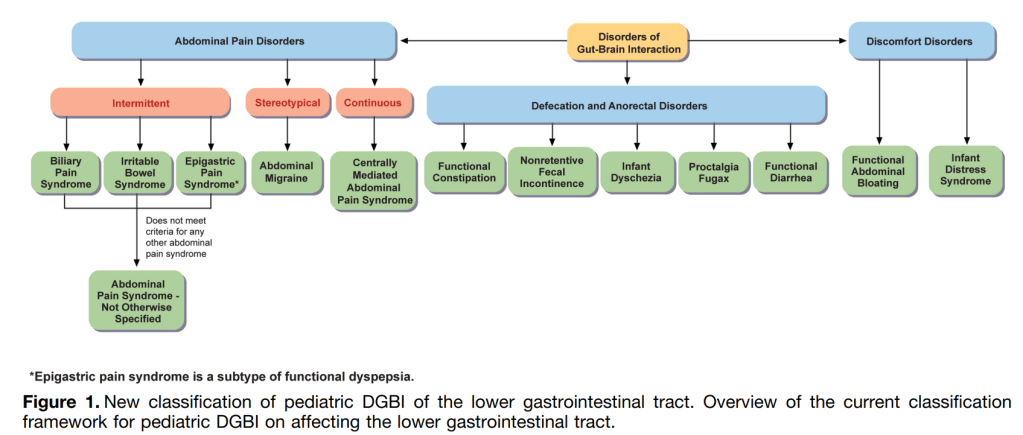
- For the Rome V recommendations, the Pediatric Committees decided to depart from the age-based divisions used in Rome IV
Irritable Bowel Syndrome:
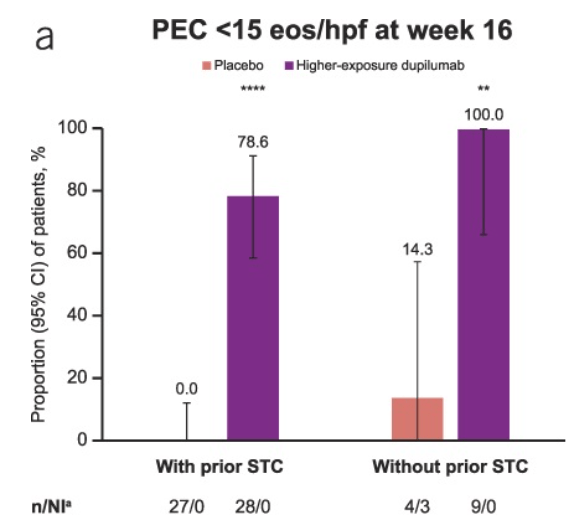
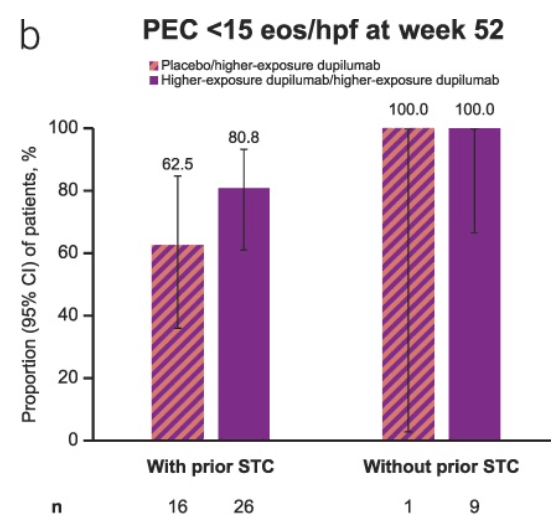
- “In Rome IV, for those children with constipation, there was an attempt to differentiate functional constipation (FC) from IBS by first attempting to treat constipation.7 …the Rome V Committee proposes a shift akin to that described in the adult IBS criteria, with a focus on the predominant symptom of abdominal pain as the differentiating factor between IBS-C and FC.”
- Pathophysiology of IBS includes early life events, heightened nerve sensitivity, and increased gut permeability. Also, “more than 90% of children and adolescents with IBS identify at least 1 food that exacerbates their GI symptoms.34,35“
- Evaluation: “testing for celiac disease is recommended in those with IBS-D” and possibly testing for parasites and fecal calprotectin
- Psycholological features: “studies have reported the association between abdominal pain–related DGBI and clinically evident as well as subclinical anxiety and depression.43–47 Anxiety and depression are as likely to follow as to precede pain48 and are not the main factors influencing pain outcomes.49–51…hildren with IBS may have increased school absenteeism, sleep disturbances, multisite pain, and functional disability.54,55“
- Treatments: Hyponotherapy, cognitive behavioral therapy, percutaneous nerve field stimulation, dietary interventions, probiotics, peppermint oil, psyllium, and pharmacologic interventions (lack of RCT evidence with most medications). Dietary intevertions: “The majority of lactose challenge RCTs in children with abdominal pain–predominant DGBI do not support the role of lactose as the trigger of the child’s symptoms.60…Given concerns for abnormal eating behaviors and the potential occurrence of avoidant and restrictive food intake disorders, it is strongly recommended that a dietitian be involved in any restriction diet and that liberalization of the restriction be instrumented when possible.65” Related blog post: Treatment Guidelines for Pediatric Irritable Bowel Syndrome
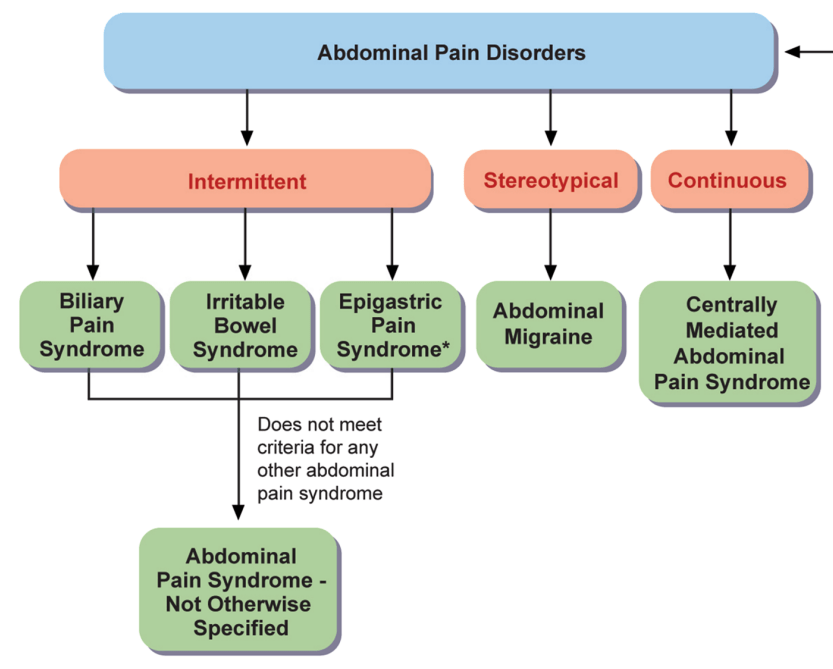
Abdominal Pain Syndrome–Not Otherwise Specified
- “The committee revised the criteria to differentiate intermittent pain (APS-NOS) from constant pain (CAPS) and specified that pain in APS-NOS should not be exclusively associated with meals, menses, or bowel movements.”
Biliary Pain Sydrome
- “A key difference is the requirement for pain to be in the right upper quadrant with or without epigastric pain, helping distinguish it from functional dyspepsia.99“
- “Biliary dyskinesia may resolve spontaneously, with conservative treatment often showing equivalent or better outcomes than cholecystectomy in long-term follow-up.104 Therefore, cholecystectomy should be considered only when other nonsurgical treatments have been appropriately trialed and have failed to improve symptoms…surgery may not alleviate symptoms or may exacerbate symptoms or result in complications.”
Abdominal migraine

- “In cases of overlapping symptoms with cyclic vomiting syndrome, the predominant and most bothersome symptom will guide the primary diagnosis.”
- “There are no US Food and Drug Administration–approved medications or evidence-based guidelines for treating AM in children…Treatment should be individualized…Children with frequent and debilitating episodes may benefit from prophylactic therapy, as some evidence suggests that antimigraine medications.”
Centrally Mediated Abdominal Pain Syndrome
- “Continuous pain as in the case of CAPS is much less frequent. There is no specific epidemiologic data for this diagnosis, as CAPS was not part of previous pediatric Rome Criteria.”
- “Some of the treatment strategies listed in the ESPGHAN-NAPGHAN guidelines related to IBS and FAP-NOS may apply to this condition as well.68 Brain–gut therapies are strongly recommended,122,123 given the central sensitization that is likely present in these patients.”
Functional Abdominal Bloating
- “Functional abdominal bloating is a recent addition to the pediatric Rome Criteria.”
- “Potential organic causes of both bloating and distention include small bowel bacterial overgrowth, celiac disease,199 congenital sucrase-isomaltase deficiency, and other malabsorption disorders.195“
Infant Distress Syndrome
- “IDS is a new name proposed by the Rome V Committee in lieu of the term “infant colic.” The Rome V Committee agrees that this syndrome of excessive crying in infancy belongs to the DGBI group because there is evidence for a role of both brain and gut in its pathophysiology. However, the term “colic” suggests that the pain arises in the colon, which has not been proven to date. “
- “The Rome V committee, however, agreed that this criterion of 3 hours was arbitrary and that many infants present to the pediatrician with excessive crying of a duration of less than 3 hours per day but with severe impact on at least 1 of the caregivers.”
- Pathophysiology: “The pathophysiological mechanisms underlying IDS are still poorly understood, but IDS is likely to be a multifactorial disorder with GI, neurologic, and psychosocial disturbances.207 The pathogenesis of excessive crying may be closely related to the development of the GI microbiome.”
- Maternal Psychology: “Maternal anxiety has been consistently found to be both a preceding and concurrent condition of excessive crying…However, depression seems to be a result of IDS, with excessive crying and maternal depression exacerbating simultaneously in a vicious cycle.214“
- Treatment: “The cornerstone of helping infants with IDS is to validate the infant’s symptoms and the emotional burden of the parents, reassure the parents that their child is healthy, and educate them about the self-limited nature of IDS and the need for support by family members…Probiotics may reduce crying time in infants with IDS…Evidence for the effectiveness of dietary modifications to treat IDS is scarce and presents a significant risk of bias.222 However, removing cow’s milk from the infant’s diet or from the maternal diet in those who are breastfed may be beneficial..Tthe evidence for using …proton pump inhibitors is very weak.224“
My take: This is a very useful article and worth reading. I like the change in terminology from colic to infant distress syndrome and labeling IBS-C instead of FC when patient has predominantly abdominal pain.