Visser E, Reijntjes M, Heuthorst L et al.The Lancet Gastroenterology & Hepatology, 2026; 11, 190-203. Appendicectomy versus switching to a JAK inhibitor in inducing remission in patients with active ulcerative colitis after biologic therapy failure (COSTA): 1-year results of a multicentre, prospective, cohort study
Last year, the ACCURE Trial (see blog post link below), showed that laparoscopic appendicectomy, in addition to standard medical therapy, significantly reduced the relapse rates for ulcerative colitis (UC) within 1 year. Most patients were treated with mesalamine.
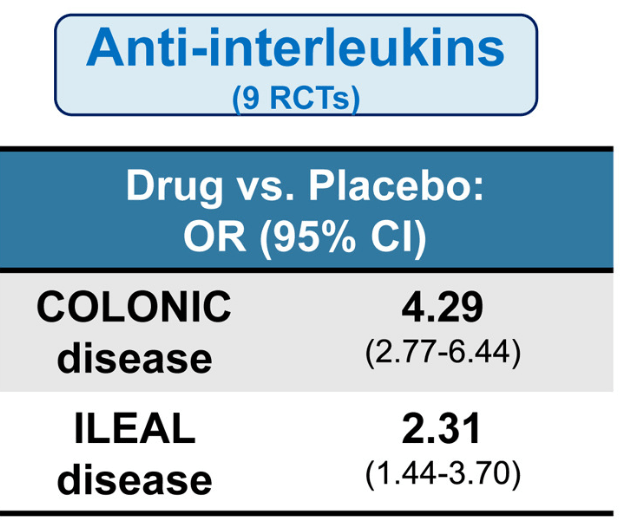
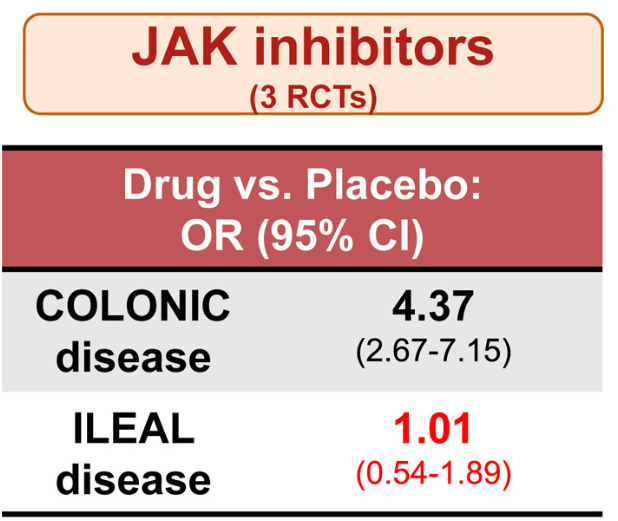
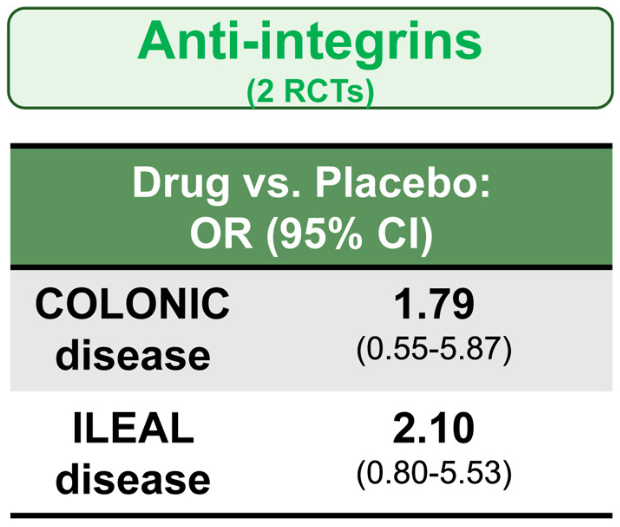
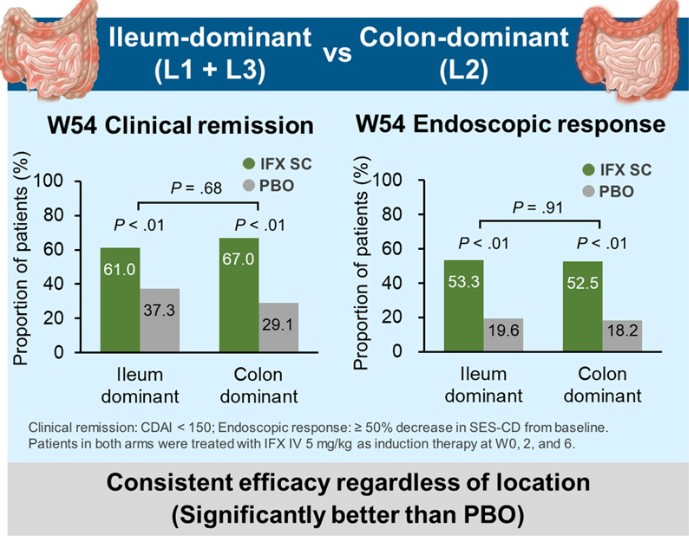
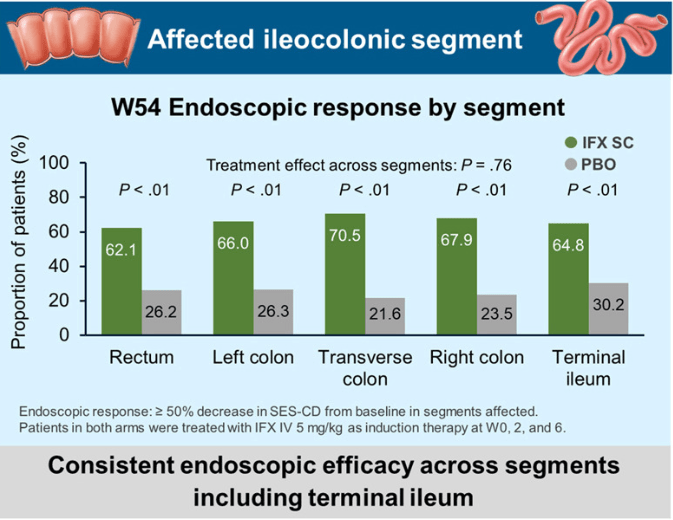
The COSTA study examined adult patients (n=125) who were not responding to advanced therapy. Patients were offered one of three treatments: laparoscopic appendicectomy while continuing their existing advanced therapy at a stable dose; switching their advanced therapy to a JAK inhibitor; or colectomy. 116 patients were included in the modified intention-to-treat-analysis (67 received appendicectomy and 49 received JAK inhibitor (primarily tofacitinib).
Key findings:
- 22 (32.8%) of 67 patients in the appendicectomy group were in clinical remission without therapy failure at 12 months compared with six (12.2%) of 49 patients in the JAK inhibitor group (p=0.016)
- At 12 months, corticosteroid-free clinical remission without therapy failure was attained in 22 (32.8%) of 67 patients in the appendicectomy group compared with six (12.2%) of 49 patients in the JAK inhibitor group (p=0.010). Clinical response in 49 (73.1%) of 67 patients compared with 26 (53.1%) of 49 patients (p=0·025), and endoscopic response in 31 (48.4%) of 64 patients compared with 11 (25.6%) of 43 patients (p=0·018)
My take (borrowed in part from the authors): “Appendicectomy as an adjunct to advanced therapy in biologic-exposed patients with active ulcerative colitis was associated with higher clinical remission rates at 12 months compared with switching to a JAK inhibitor.” We don’t know how appendectomy would influence disease course in pediatric patients. We also don’t know how this information will be incorporated into adult guidelines.
Interestingly, there have been studies (two cited below) indicating that appendectomy lowers the risk of developing inflammatory bowel disease:
- RE Andersson et al. NEJM 2001;344:808-814. Appendectomy and Protection against Ulcerative Colitis. Conclusion: “Appendectomy for an inflammatory condition (appendicitis or lymphadenitis) but not for nonspecific abdominal pain is associated with a low risk of subsequent ulcerative colitis. This inverse relation is limited to patients who undergo surgery before the age of 20 years.”
- A Kiasat et al. Colorectal Dis. 2022 Apr 6;24(8):975–983. Childhood appendicitis and future risk of inflammatory bowel disease – A nationwide cohort study in Sweden 1973–2017)
Related blog post: ACCURE Trial: Appendectomy As an Adjunct Ulcerative Colitis Treatment Plus One