There is a well-described “evidence-practice gap” which has been cited to describe the lag between initial discovery and widespread, evidence-based clinical implementation (M Beauchemin et al. ANS Adv Nurs Sci. 2019 Oct-Dec;42(4):307–324); it has been suggested that this gap is typically about 17 years.
I was looking this up after reading a “practice tips” article on H pylori and was surprised at how infrequentlly practitioners follow guidelines for optimal treatment.
M Ventoso, SF Moss. Gastroenterology & Hepatology 2026; 22: 80-86. Open Access! Practice Tips From the Updated Helicobacter pylori Treatment Guidelines
Background: “The American College of Gastroenterology (ACG) recently published H pylori management guidelines6 with significant updates to the prior recommendations published in 2017.7 Importantly, the 2017 management guidelines relied heavily upon extrapolation from studies performed outside of North America. Since then, new data from US patients have become available that highlights the rising rates of resistance of commonly used antibiotics for treatment of H pylori.”
Key points:
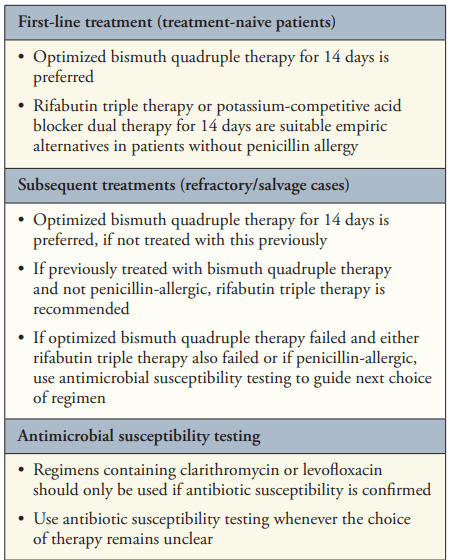
- “Bismuth Quadruple Therapy Is First Line and Should Be Optimized” (see below). A 14-day course is recommended (except when using Pylera which is a 10 day treatment).
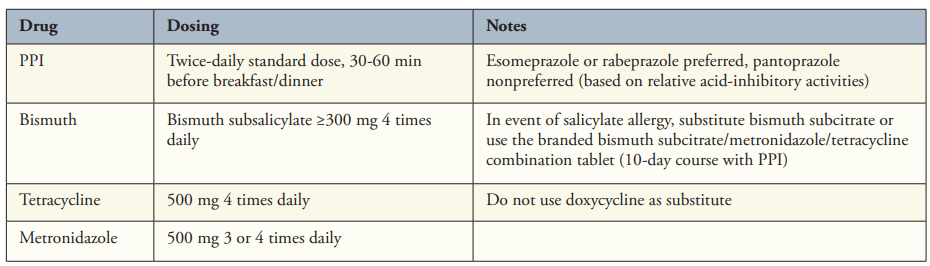
- “3 new regimens have received US Food and Drug Administration (FDA) approval for H pylori therapy, each based on high levels of gastric acid inhibition. “
- “The first is a combination tablet of rifabutin with amoxicillin and omeprazole (Talicia, RedHill Biopharma), given in daily doses of 150 mg, 3 g, and 120 mg, respectively. If the combination tablet is not available, generic equivalents can be substituted at similar doses (omeprazole 40 mg and amoxicillin 1 g each 3 times daily). Because the lowest available dose of generic rifabutin is only 150 mg, the best option is likely 150 mg twice daily”
- “The second regimen is based on vonoprazan (Voquezna, Phathom Pharmaceuticals), the first potassium-competitive acid blocker (P-CAB) approved in the United States. substituting the P-CAB vonoprazan for lansoprazole in clarithromycin triple therapy produced higher eradication rates (81% compared with 69%). Similar statistically high rates (77%) were achieved with a dual vonoprazan-amoxicillin combination in the same trial, leading to FDA approval of both the triple and dual regimens14“
- For “both rifabutin- and vonoprazan-containing regimens use amoxicillin and cannot be used in the confirmed penicillin-allergic patient. For patients with unproven penicillin allergy (approximately 10% of the US population), allergy testing is recommended”
- “Use Clarithromycin or Levofloxacin Only If Antimicrobial Susceptibility Is Confirmed“
- “The majority of practitioners continue to use clarithromycin-based triple therapy and have not followed the 2017 ACG H pylori guidelines …It is striking that clarithromycin-PPI triple therapy still dominates the US market, comprising greater than 80% of all treatments in 2016 to 2019.11 Notably, in approximately half of the cases where clarithromycin-PPI triple therapy failed, exactly the same prescription was given a second time.11 Thus, a massive implementation gap exists.”
My take:
- While this article focuses on adult guidelines, there are similar issues in pediatrics. There is an even greater need for research involving children with H pylori. As vonoprazan appears to improve eradication rates, more guidance is needed regarding drug regimens with vonoprazan in kids.
- I frequently see patients who have received empiric clarithromycin-based therapy. Based on this article, I should be less surprised that this is so commonplace.
Related blog posts:
- Dr. Benjamin Gold: 2024 Pediatric H pylori Guidelines (Part One)
- Dr. Benjamin Gold: 2024 Pediatric H pylori Guidelines (Part 2)
- QI Project: Increasing H Pylori Eradication
- 5 Rights and H pylori Treatment
- Give the Right Dose (for H pylori) -It Works Better!
- Helicobacter Pylori Stool Susceptibility in Children –How Good Is It?
- Understanding FDA Approval of Vonoprazan-Based Therapies for Helicobacter Pylori
- Synergistic Dangers: Helicobacter Pylori and Cancer Genes
- AGA: Best Practice Advice for Refractory H pylori
- Treating Helicobacter Pylori Lowers The Risk of Gastric Cancer
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.
