A series of articles details the 2021 AGA Guidelines for Crohn’s disease (CD) including a clinical practice guideline (pg 2496-2508), a clinical decision support tool (2509-2510), a spotlight summary (pg 2511), a technical review (2512-2557), and a review of the recommendations (pg 2557-2262). I will highlight the first article.
JD Feuerstein et al. Gastroenterol 2021; 160: 2496-2508. Full text: AGA Clinical Practice Guidelines on the Medical Management of Moderate to Severe Luminal and Perianal Fistulizing Crohn’s Disease
Full text: Spotlight
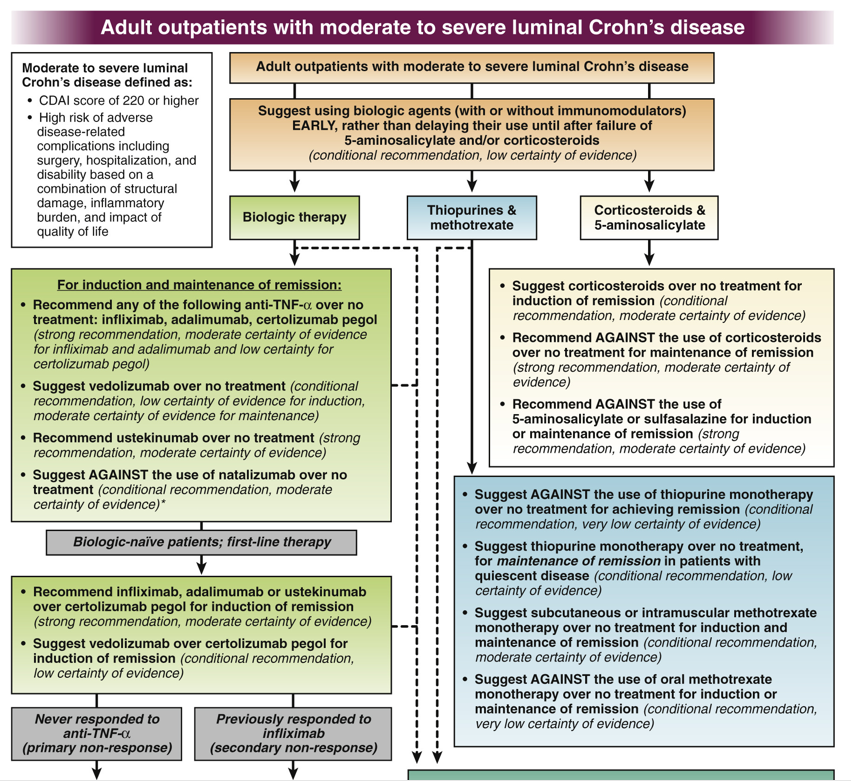
For me the most important of their recommendations was #7:
- In adult outpatients with moderate to severe CD, the AGA suggests early introduction with a biologic with or without an immunomodulator rather than delaying their use until after failure of 5-aminosalicylates and/or corticosteroids.
Other points:
- For moderate to severe CD, the authors support the use of anti-TNF therapy in combination with immunomodulators, support the use of ustekinumab and vedolizumab, and support the use of SC/IM (but not oral) methotrexate.
- The authors are AGAINST the use of mesalamine/sulfasalazine products as well as corticosteroids for maintenance of remission for CD
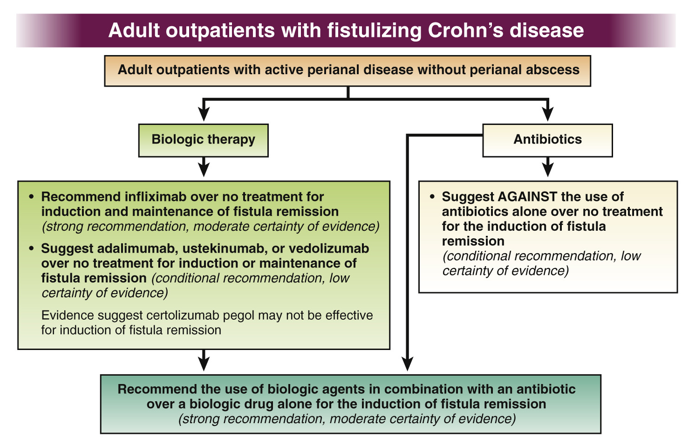
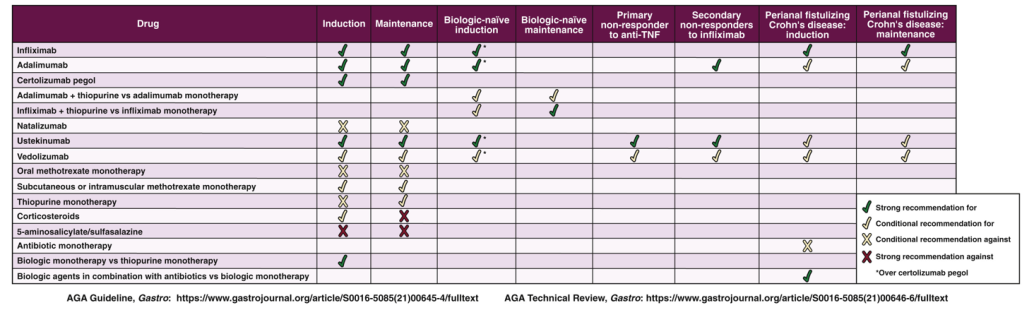
- See full text link to Table 3 for summary of all of their recommendations: AGA Clinical Practice Guidelines on the Medical Management of Moderate to Severe Luminal and Perianal Fistulizing Crohn’s Disease
From Spotlight: