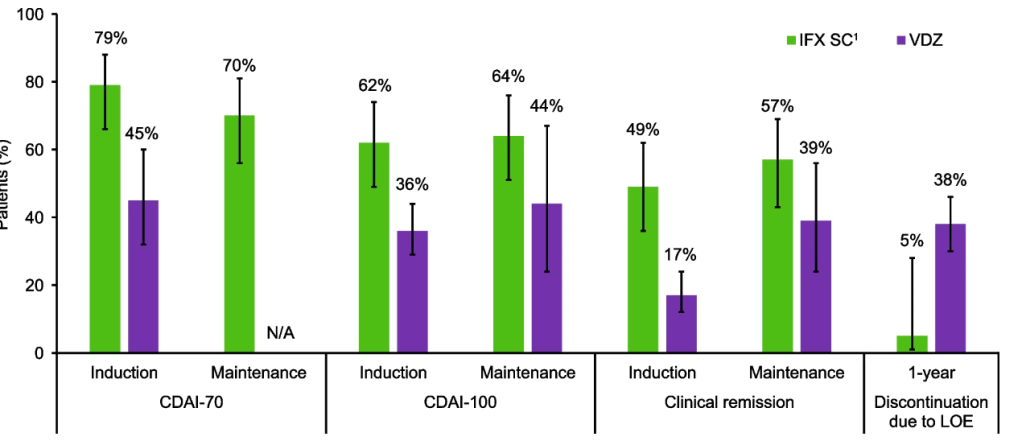
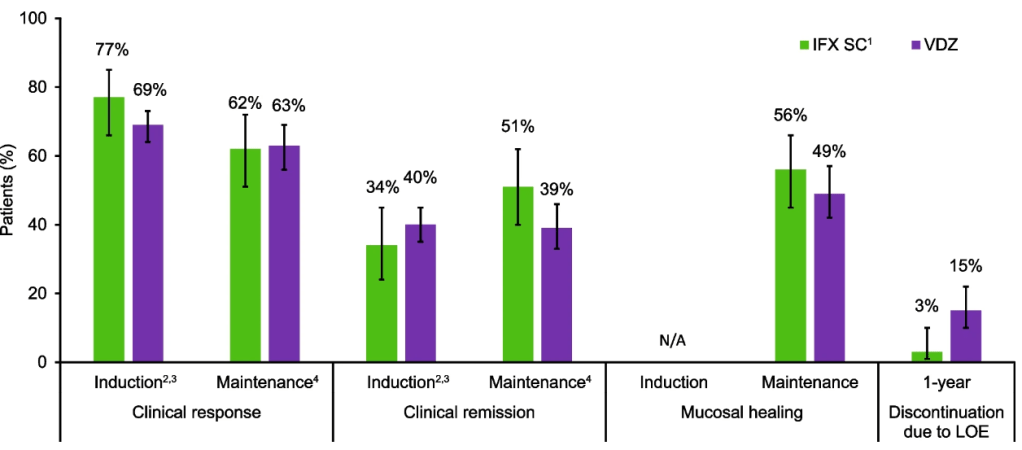
Lancet Gastroenterol Hepatol 2026; 11: 12-21. Vedolizumab in early and late Crohn’s disease (LOVE-CD): a phase 4 open-label cohort study
Methods: Eligible patients were adults aged 18–80 years with moderate to severe Crohn’s disease (Crohn’s Disease Activity Index [CDAI] 220–450, with ulcers at endoscopy). Patients were divided into two groups: those with early Crohn’s disease, n=86 (defined as a diagnosis less than 2 years ago and naive to advanced treatment [naive or only treated with corticosteroids or immunomodulators, or both]); and those with late Crohn’s disease, n=174 (defined as a diagnosis more than 2 years ago and previously treated with corticosteroids, immunomodulators, and anti-TNF agents). The primary endpoint was the proportion of patients with clinical and endoscopic remission (defined as CDAI ≤150 and SES-CD <4) at both week 26 and 52.
Key findings:
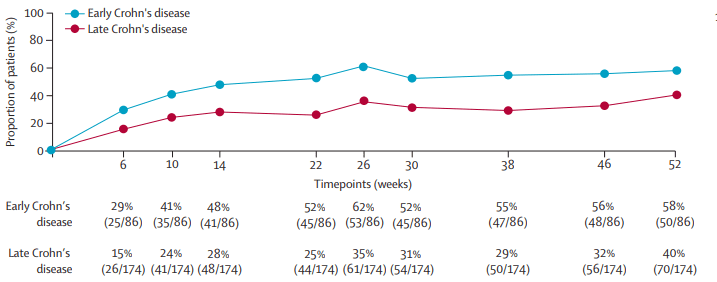
- Clinical and endoscopic remission at both week 26 and 52 was achieved in 27 (31·4%) of 86 patients with early Crohn’s disease versus 15 (8·6%) of 174 patients with late Crohn’s disease (difference 22·8%, 95% CI 12·6–33·7)
- Serious adverse events occurred in three (3·5%) of 86 patients with early Crohn’s disease versus 46 (26·4%) of 174 patients with late Crohn’s disease and included infections (one [1·2%] vs 13 [7·5%]), surgery (none vs eight [4·6%]), intestinal obstruction (none vs four [2·3%]), exacerbation of Crohn’s disease (one [1·2%] vs six [3·4%]), and malignancy (none vs three [1·7%])
Discussion:
- “After 52 weeks of open-label treatment, almost 60% of patients with early disease achieved clinical remission and more than 50% were in endoscopic remission. By contrast, deep remission rates at both weeks 26 and 52 were observed in less than 10% of patients with late Crohn’s disease (ie, those with longstanding disease and previous exposure to anti-TNF
agents).” - “Despite the earlier stage at which vedolizumab was initiated, disease severity was comparable betweenthe early and late Crohn’s disease groups, from a clinical (median CDAI 255 vs 259), endoscopic (median SES-CD 9 vs 12), and biochemical perspective (median serum C-reactive protein 9 mg/L vs 8 mg/L).”
- “A pivotal observation in LOVE-CD was that dose intensification after week 26 (ie, doubling the dose) in patients without an endoscopic response did not lead to higher endoscopic remission rates, despite significantly higher serum vedolizumab concentrations. This
finding suggests that the dosing schedule that was originally designed and approved is optimal for most patients and saturates the target. Patients who do not respond most likely have other dominant immune pathways that are activated and remain unaffected by
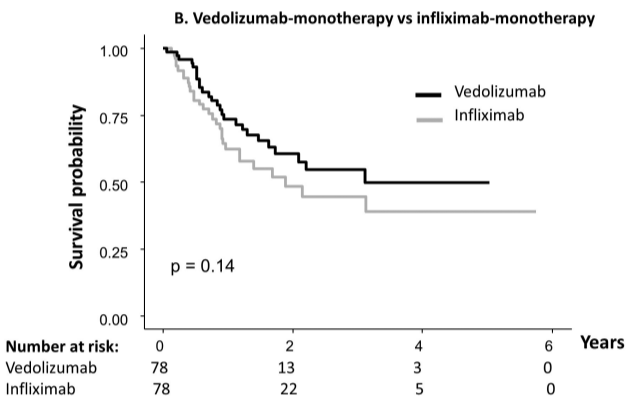
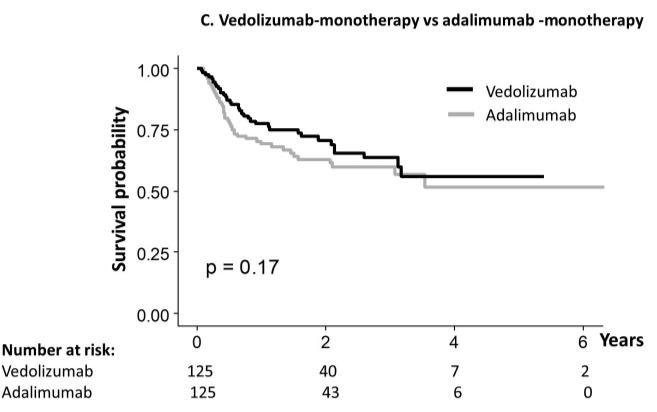
vedolizumab.” - “All three classes of biologics approved for the treatment of Crohn’s disease perform better when initiated early in the disease course.”
My take: For Crohn’s disease (CD), vedolizumab should be mainly used in those without prior biologic therapy. In addition, changes in vedolizumab dosing based on concentrations is much less helpful than it is with anti-TNF agents.
Related blog posts:
- Prior Exposure to TNF Antagonists May Increase Response to JAK Inhibitors in Patients with Ulcerative Colitis (& lower response to vedolizumab) (2025)
- Should Vedolizumab Be Used as a First Line Agent for Crohn’s Disease?
- Vedolizumab’s Impact on Postoperative Crohn’s Disease Recurrence
- Dr. Maria Oliva-Hemker: Positioning Therapies for Pediatric Crohn’s Disease
- What is Mild Crohn’s Disease and How to Treat It
- Comparative Efficacy of Biologics for Crohn’s Disease
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.