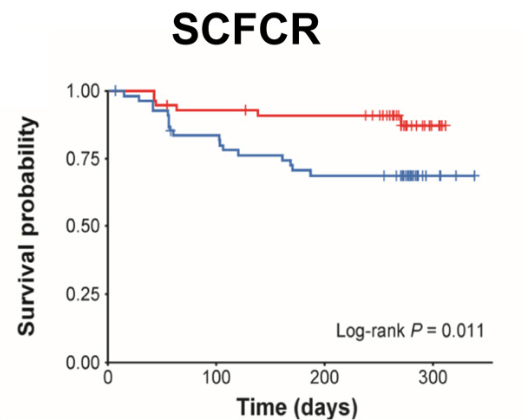
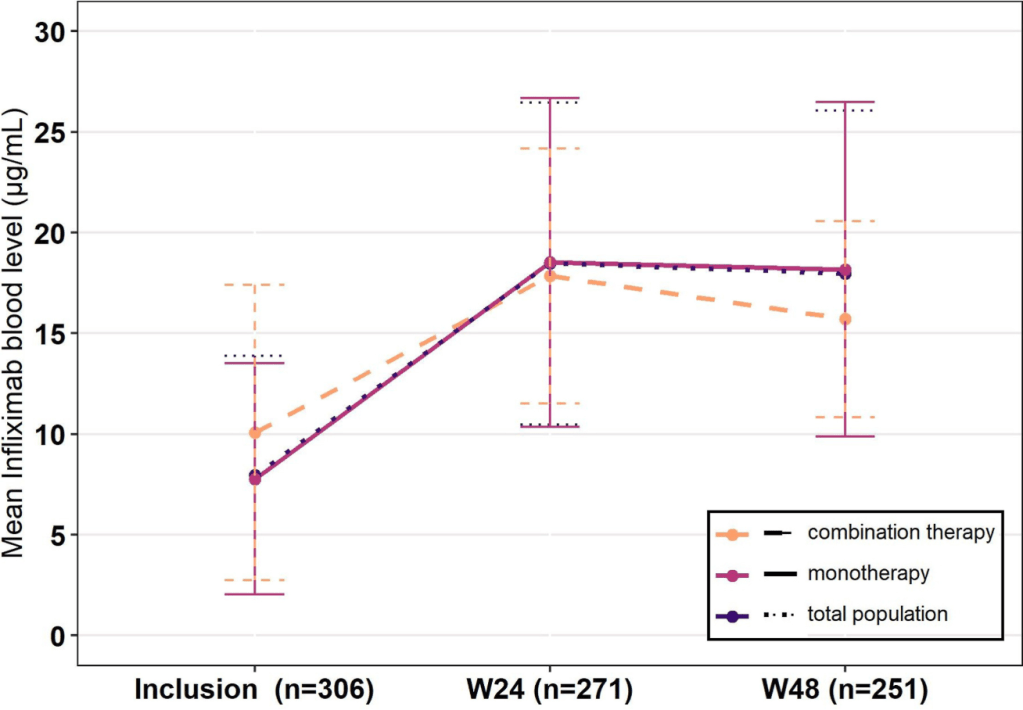
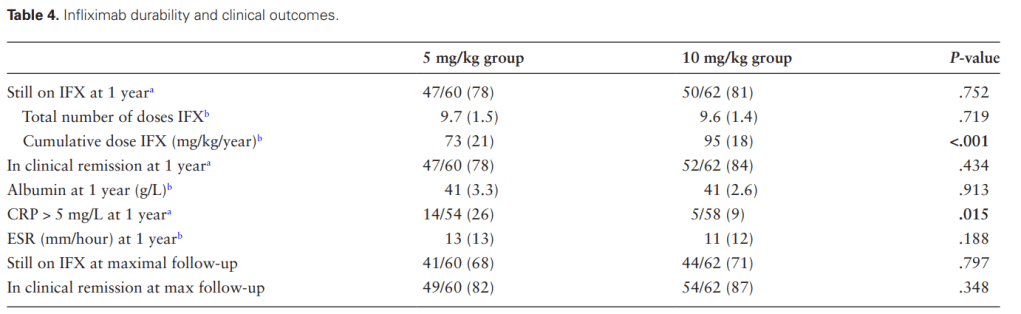
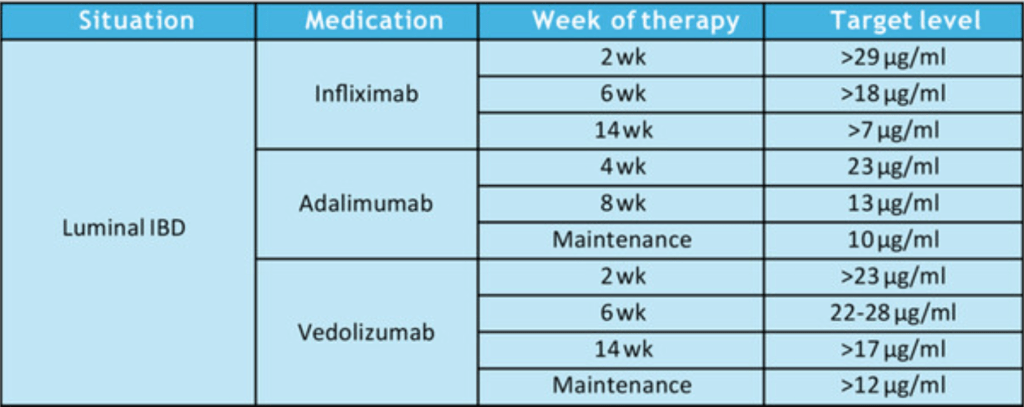
- HH Lee et al. Clin Gastroenterol Hepatol 2026; 24: 923-931. Efficacy of Advanced Therapies in Achieving Remission by Disease Location in Crohn’s Disease: A Systematic Review and Meta-analysis
- Sands B et al. Clin Gastroenterol Hepatol 2026; 24: 1111-1120. Open Access! Endoscopic Response to Subcutaneous Infliximab by Disease Location: A Post Hoc Analysis of the LIBERTY-CD Study
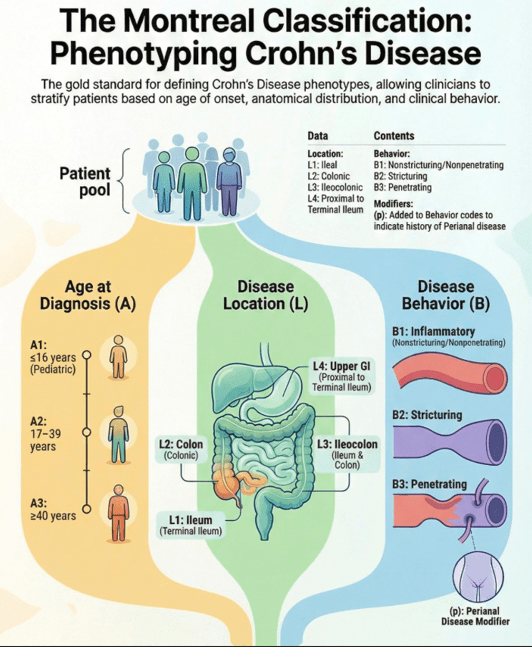
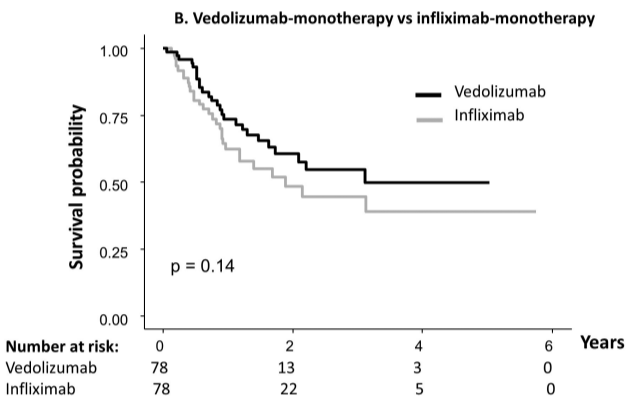
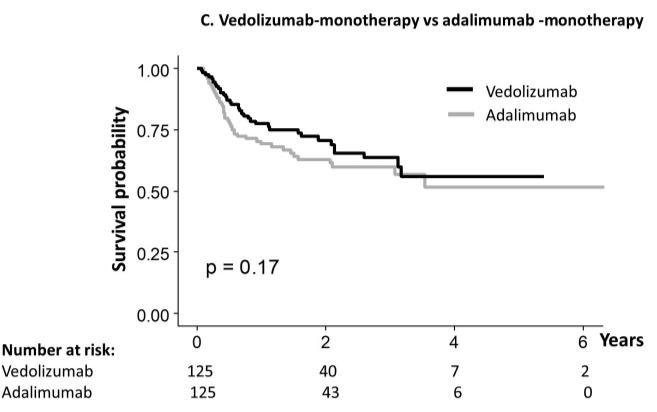
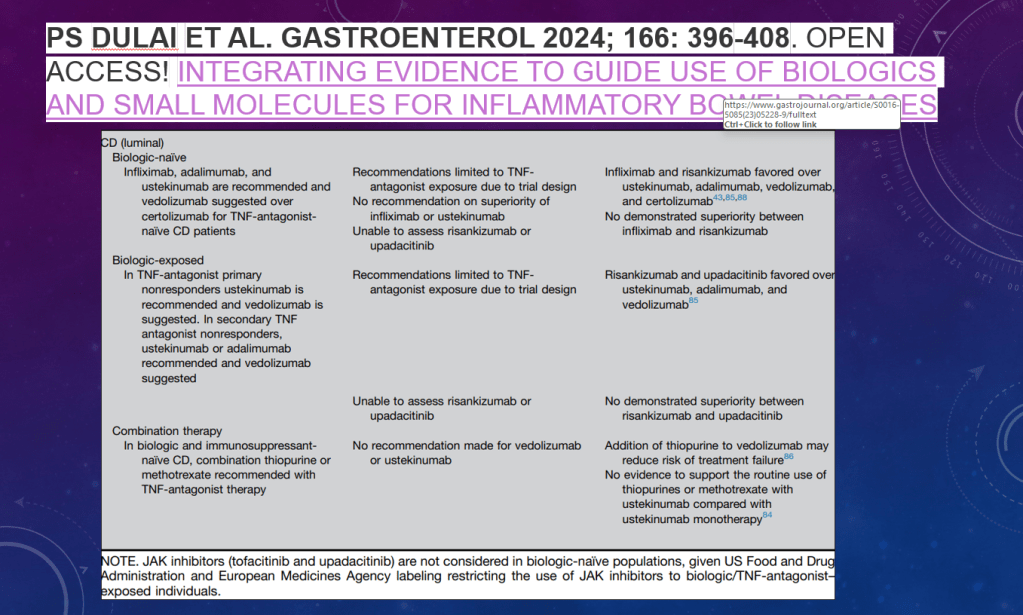
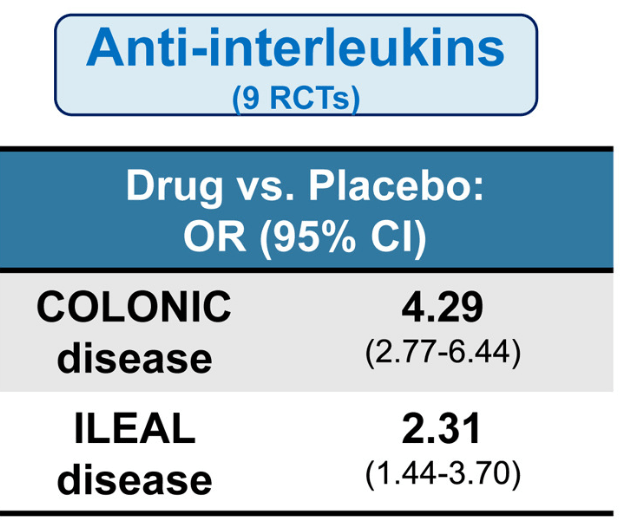
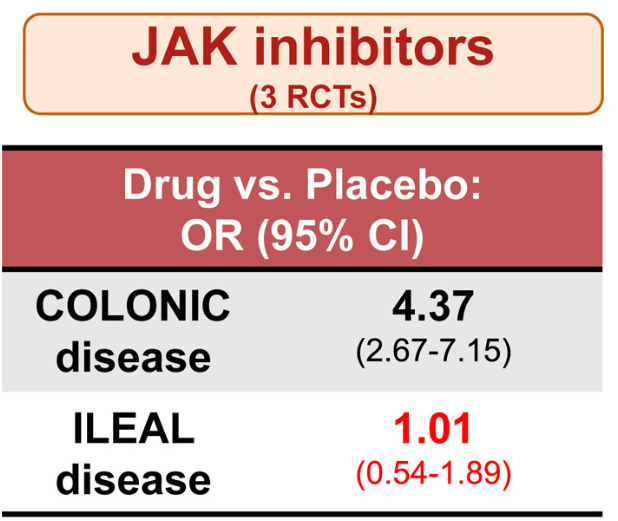
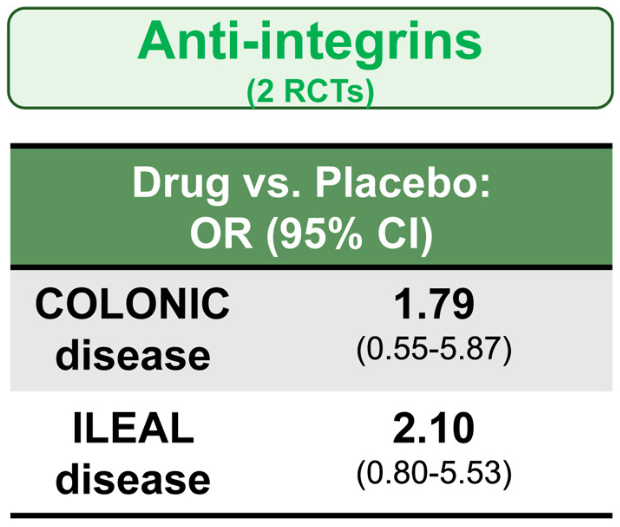
Methods: In the study by Lee et al, the authors identified 14 randomized controlled trials in 3139 patients with moderate-to-severe CD who were treated with different advanced therapies vs placebo, and reported efficacy in inducing clinical remission, stratified by disease location (isolated colonic vs ileal disease, excluding ileocolonic disease). The authors did not identify any RCT of TNF antagonists that reported induction of remission by disease location.
Key findings:
- All advanced therapies had better success with colonic disease rather than ileal disease
- Anti-interleukins (eg. IL23 o rIL12/IL23 agents) worked best for ileal disease among these advanced therapies
- JAK inhibitors did not work well for ileal disease, but performed well for colonic disease
- Anti-integrins, like vedolizumab, had some efficacy for ileal disease but generally a lower clinical remission rate than other agents
- Lmitations included the use of clinical remission as the primary outcome
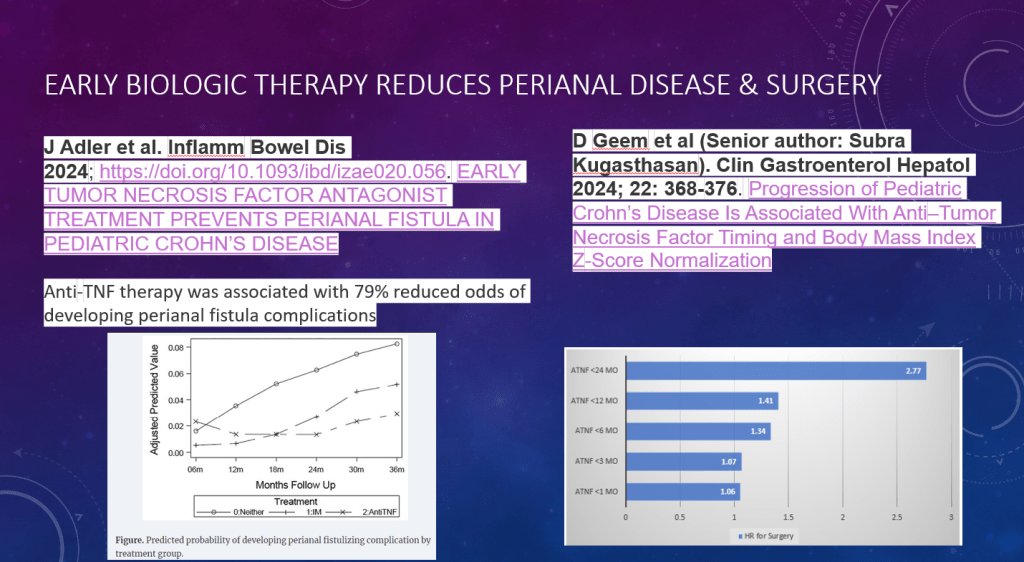
- While this study did not provide data on anti-TNF therapy, in the discussion the authors note that “TNF antagonists may have advantages in small bowel CD…infliximab demonstrated the highest rate of improvement in large ileal ulcers (>0.5 cm).” [ref#45] “Additionally, infliximab has been reported to reduce fibrostenosis-associated inflammation, [Ref#46] making it currently the most suitable therapeuctic option for small bowel CD.”
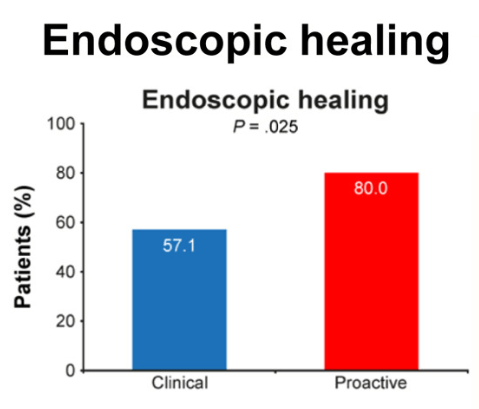
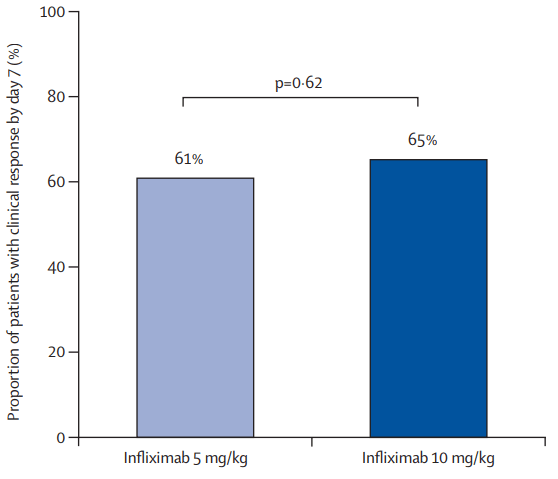
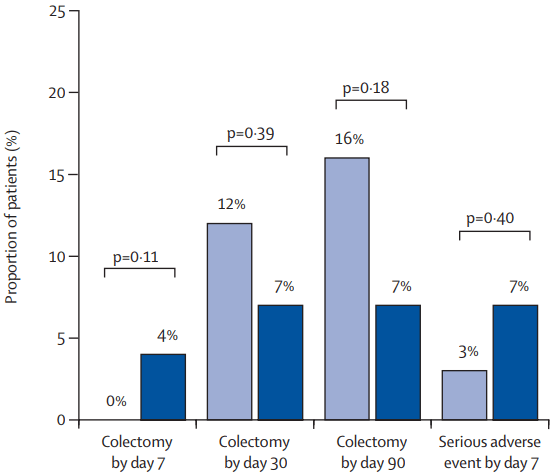
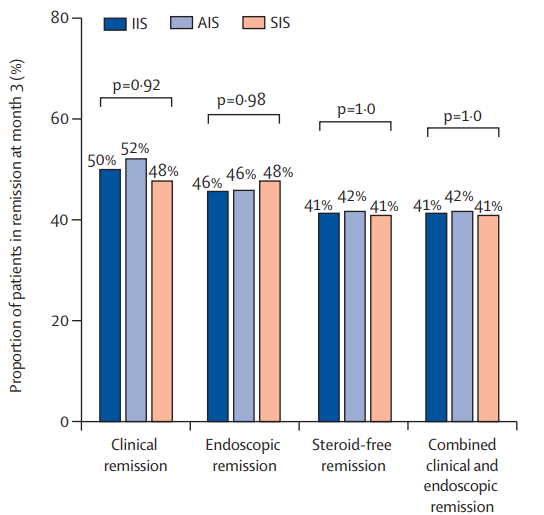
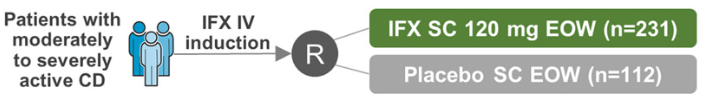
In the study by Sands et al, this post-hoc analysis included week 10 responders (n=329) to intravenous IFX induction therapy who were randomized to receive IFX SC 120 mg every 2 weeks or placebo (PBO) during maintenance therapy.

Key findings:
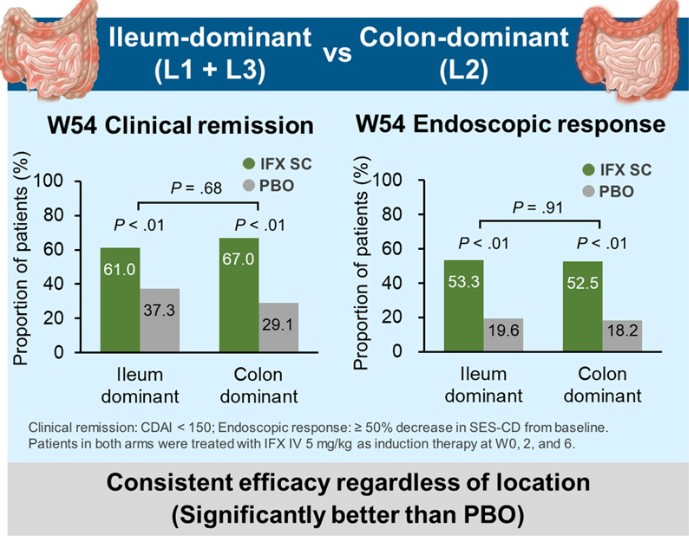
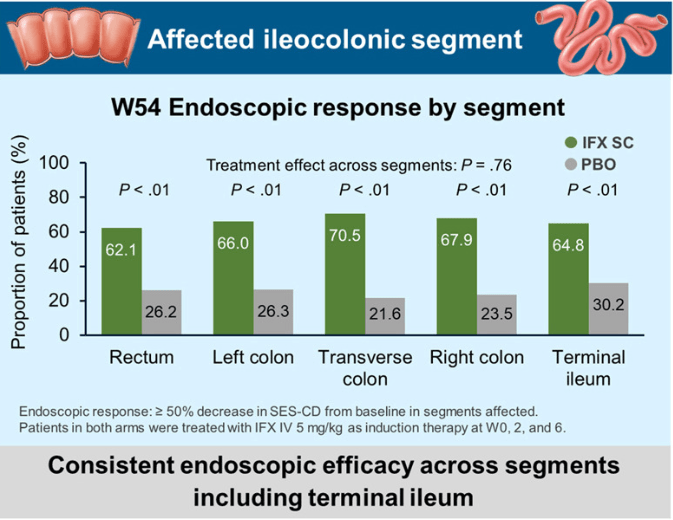
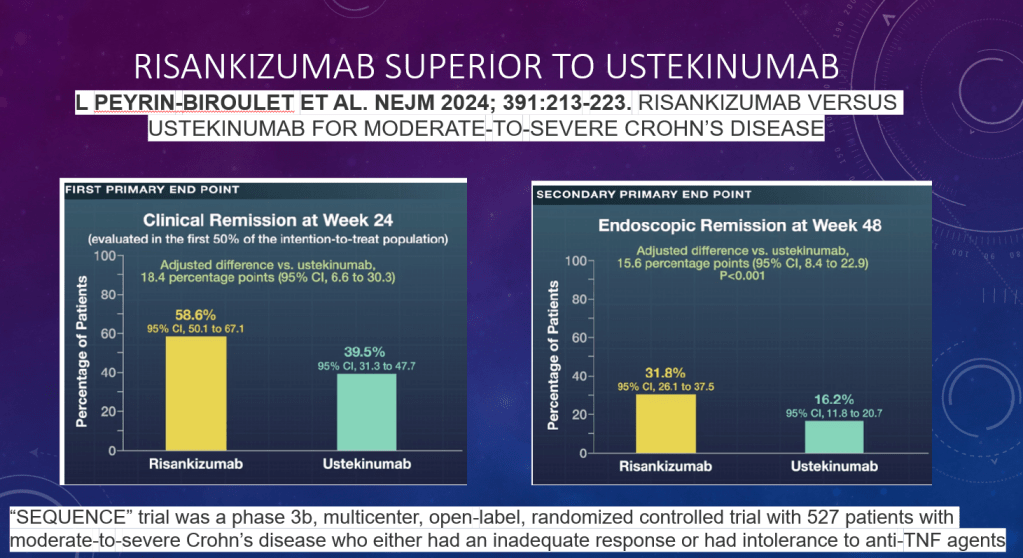
My take: These two studies indicate that anti-TNF agents (particularly infliximab) and IL-23 type agents are most effective for Crohn’s disease affecting the ileum. JAK inhibitors are best for colonic disease.
Related blog posts:
- Dr. Maria Oliva-Hemker: Positioning Therapies for Pediatric Crohn’s Disease
- Spotlight: AGA Living Clinical Practice Guideline on the Pharmacologic Management of Moderate-to-Severe Crohn’s Disease
- AGA Living Clinical Practice Guideline on the Pharmacologic Management of Moderate-to-Severe Crohn’s Disease
- Is It RISKy Not To Use Anti-TNF Therapy for Pediatric Crohn’s Disease?
- Comparative Evidence and Positioning Advance Therapies for Inflammatory Bowel Disease
- Upadacitinib vs Risankizumab for Crohn’s Disease