H Khalili et al. Clin Gastroenterol Hepatol 2019; 17: 123-29. Using data from two prospective Swedish cohort sutdies with 83,042 participants (age 44-83 yrs), the authors determined that there was “no evidence for association between consumption of sweetened beverages and later risk of” Crohn’s disease or Ulcerative Colitis.
WJ Sandborn et al. Gastroenterol 2019; 156: 946-57. This study published data from 354 patients who received subcutaneous abrilumab, an anti-alpha4beta7 antibody as a treatment for moderate-to-severe colitis. This 8 week treatment increased the odds of remission compared with placebo.
B Wynne et al. Gastroenterol 2019; 156: 935-45. This study showed that a psychological intervention termed “acceptance and commitment therapy (ACT)” was effective in a randomized controlled trial in reducing stress and depression in patients with quiescent or mildly-active IBD (n=122). With ACT, the “primary aim is to encourage subjects to adopt positive life values and to accept adverse experiences, including thoughts, feelings and sensations that are an inevitable consequence of life.” All program materials are available in article supplement: Full text and supplement: https://doi.org/10.1053/j.gastro.2018.11.030
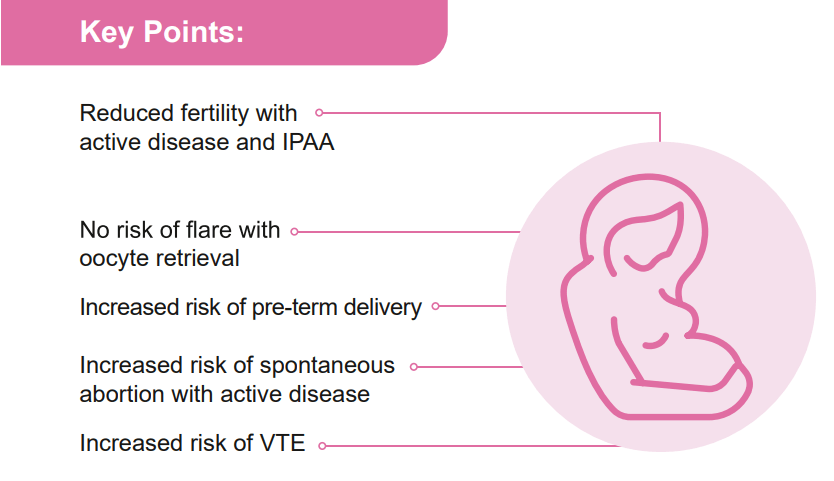
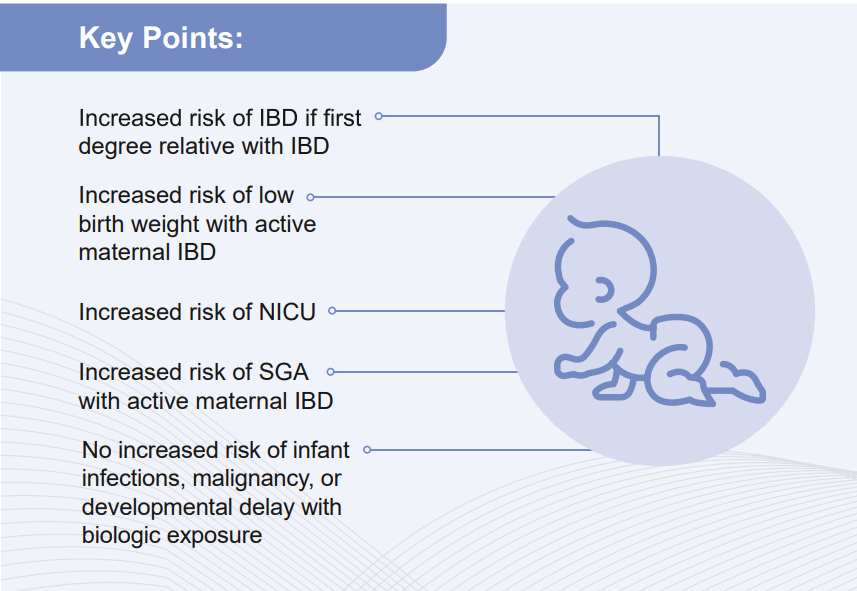
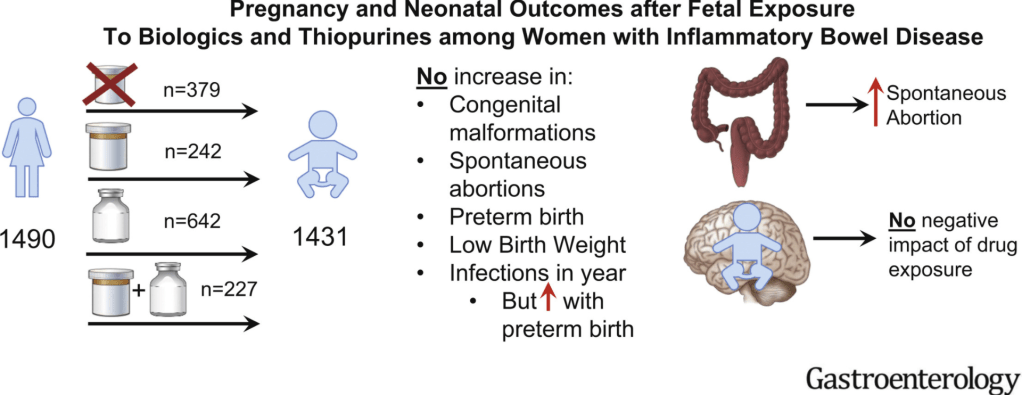
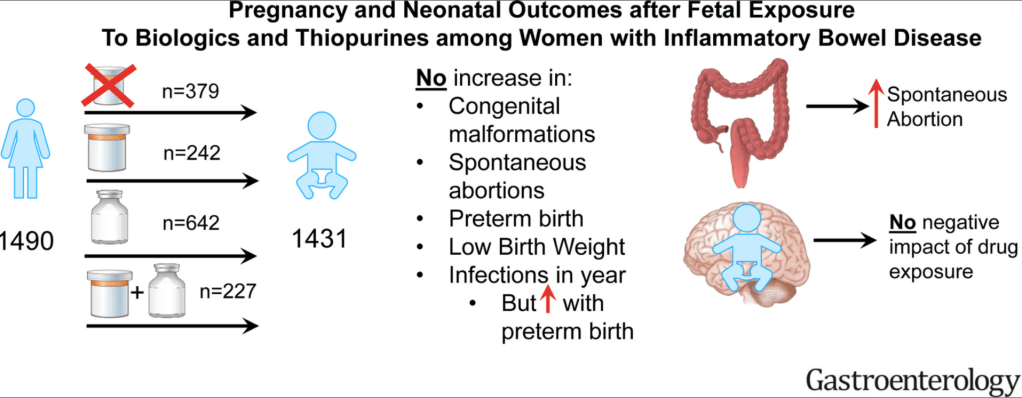
D Duricova et al. Inflamm Bowel Dis 2019; 25:789-96. This study included 72 consecutive children born to mothers with IBD treated with anti-TNF therapy during pregnancy (2007-16) along with 69 unexposed controls. Key findings: Anti-TNF therapy exposure in utero was NOT associated with a negative impact on postnatal complications, including infections, allergy, growth, or psychomotor development. Findings are limited by the small number of participants.
AW Gridnal et al. Inflamm Bowel Dis 2019; 25:642-45. The authors examined the frequency of financial conflicts of interest (FCOI) among authors of 11 relevant clinical practice guidelines for IBD in the US, the UK, Canada, and Europe. Key finding: FCOI were frequently present with 19% prevalence among US authors, 56% in UK, 84% in Canada, and 94% in Europe.
KN Weaver et al. Inflamm Bowel Dis 2019; 25:767-74. This retrospective study examined the efficacy of ustekinumab for Crohn’s disease of the pouch in 56 patients; 73% had previously been treated with anti-TNF therapy, vedolizumab or both. Key finding: 83% demonstrated a clinical response 6 months and 60% with endoscopic improvement after induction with ustekinumab. Clinical response was defined as “any improvement in symptoms …including a decrease in bowel movements, pain, or fistula drainage.”

Retiro Park, Madrid
Thanks to Jennifer