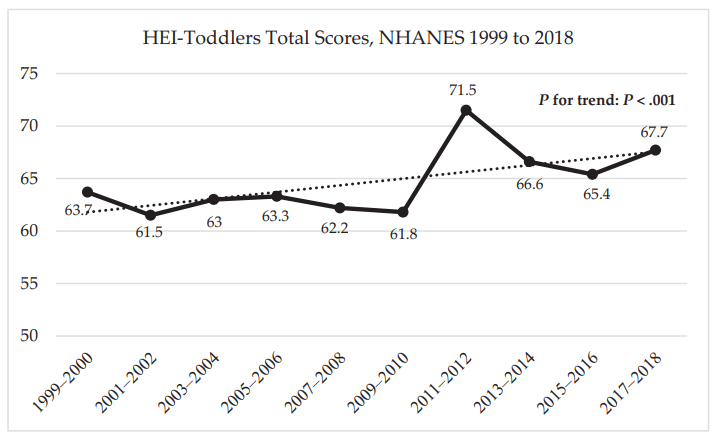
M Zimmer et al. Pediatrics 2024; 154: e2024067783.
Methods: This serial cross-sectional analysis of National Health and Nutrition Examination Survey (NHANES) data was performed with 2541 toddlers from 10 NHANES cycles from 1999 to 2018.
Key findings:
- Toddler diet quality improved significantly from 1999 to 2018 (P < .001), from 63.7
points on average in 1999-2000 to 67.7 points in 2017-2018 - A significant positive linear trend in total diet quality was observed for all socioeconomic status groups (P < .05)
- Several dietary component scores improved, as follows: Whole Fruits (P < .001), Whole
Grains (P 5 .016), Fatty Acids (P 5 .002), Refined Grains (P 5 .009), and Added Sugars
(P < .001)
In the discussion, the authors note that other age groups besides toddlers have seen modest improvements in diet. Even small improvements can be consequential on a population-basis. In adults, a 4-point increase in HEI scores has been associated with a reduced risk of cardiovascular disease. The strongest improvement between 1999 and
2018 in toddler diet quality was in scores for added sugars…attributed to declines in
added sugars from sweetened beverages. “A heightened awareness of added sugars in sweetened drink products among caregivers themselves may have led to a cultural shift
around feeding toddlers less sweetened drinks.”
While in toddlers there was no significant difference in dietary scores based on socioeconomic status, “, the diet quality gap for adults has widened over time by SES,14,15
race and Hispanic origin, and Supplemental Nutrition Assistance Program participation status.”
“Although toddlers do not receive school meals, their diets can be similarly impacted by other large-scale programs like the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC) and the Child and Adult Care Food Program. Changes to the WIC food package have been consistently linked to improved child diet quality, and may have contributed to the gains in toddler diet quality observed in this study.”
Study limitations are related to self-reported dietary intake.
My take: It feels like improving diet quality is like swimming against a strong tide. Yet, this study shows improvements can happen. Ongoing efforts are needed to encourage healthier diets.
Related blog posts:
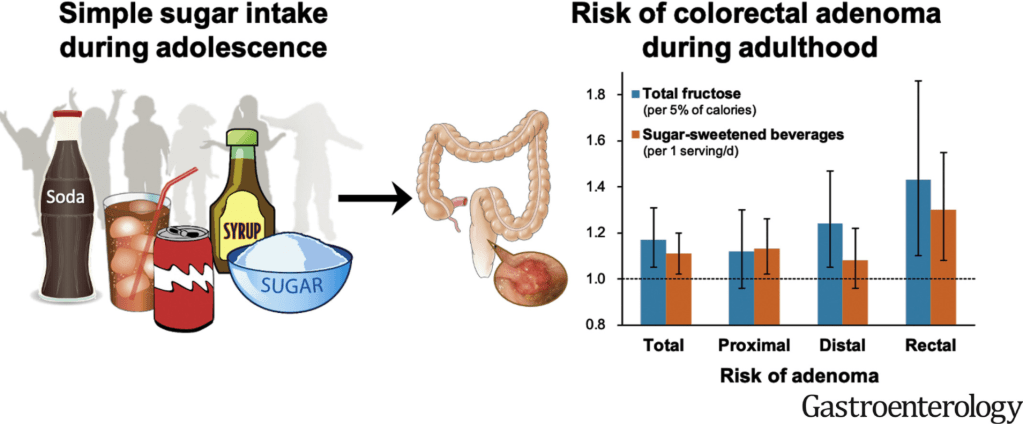
- The Hidden Dangers of Early Sugar Exposure
- NY Times: “Our Food is Killing Too Many of Us”
- Call For Action: Adolescent Nutrition Series
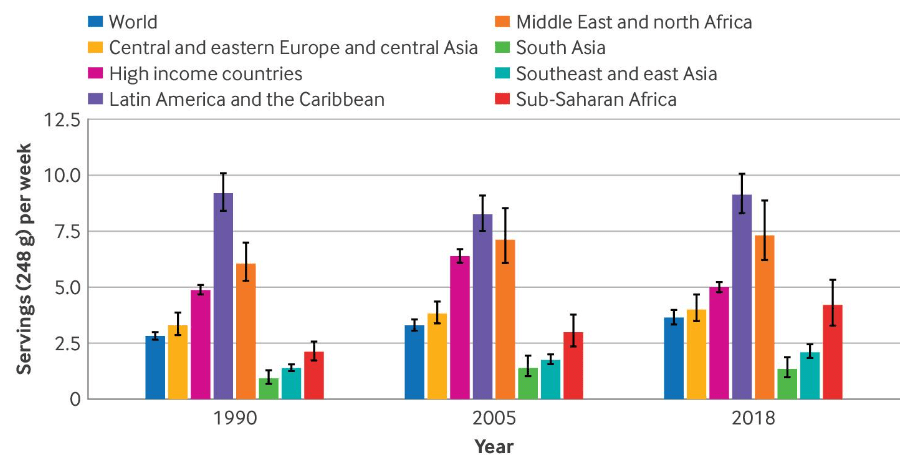
- Worldwide Increase in Sugar-Sweetened Beverage Intake
- The Link Between Ultra-Processed Foods and Irritable Bowel
- Sugary Beverage and Liver Stiffness in Healthy Adults
- Low Free Sugar Diet for Nonalcoholic Fatty Liver Disease in Adolescent Boys
- Deceptive Research: When Sugar Leaves A Bitter Taste
- NY Times: Cutting Sugar Improves Children’s Health in Just 10 Days