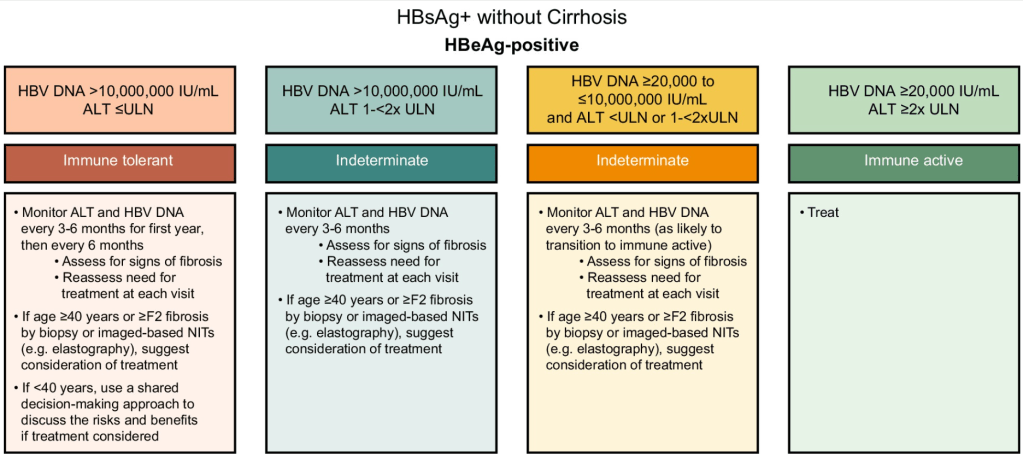
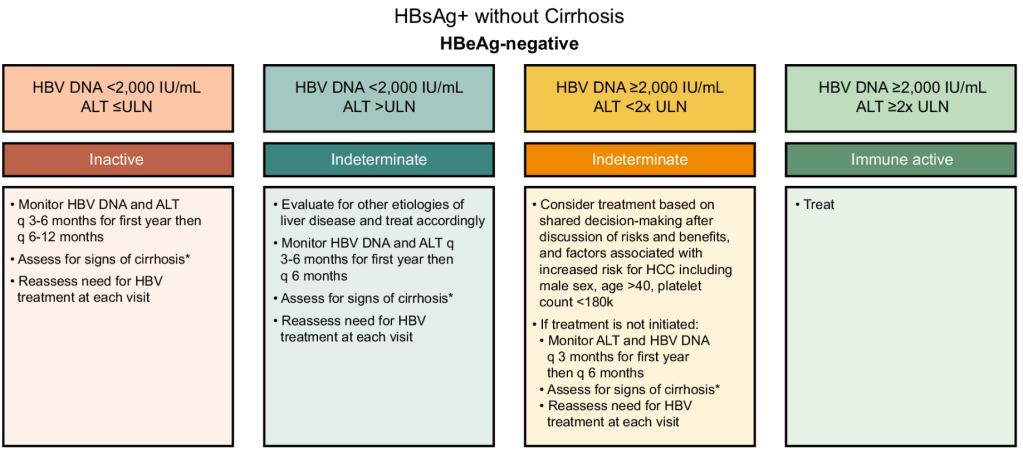
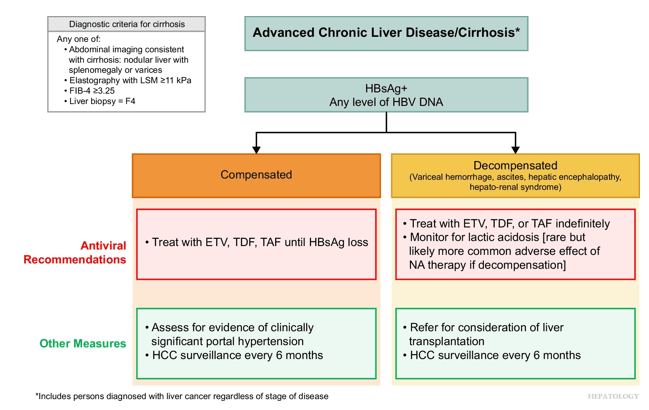
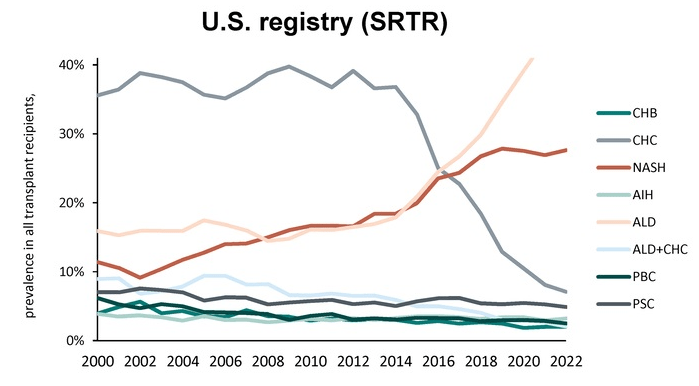
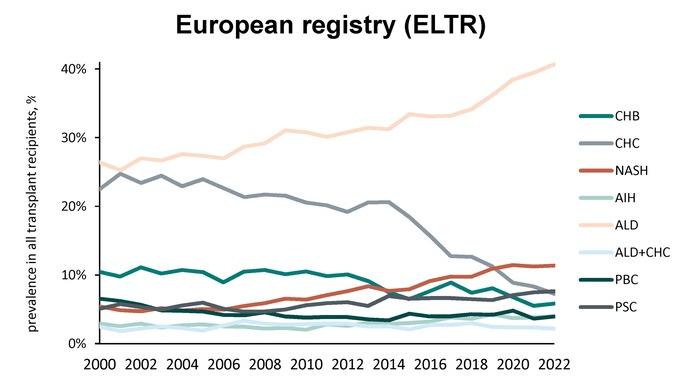
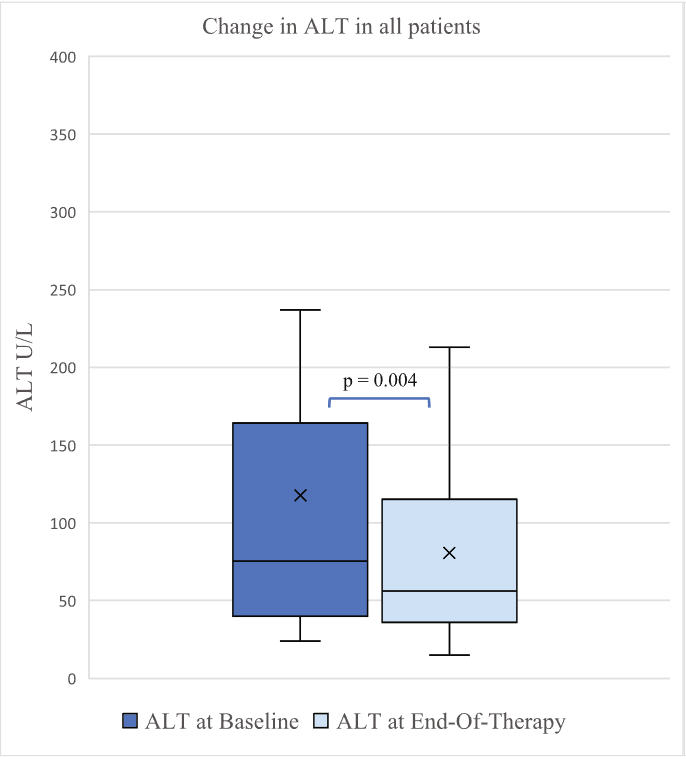
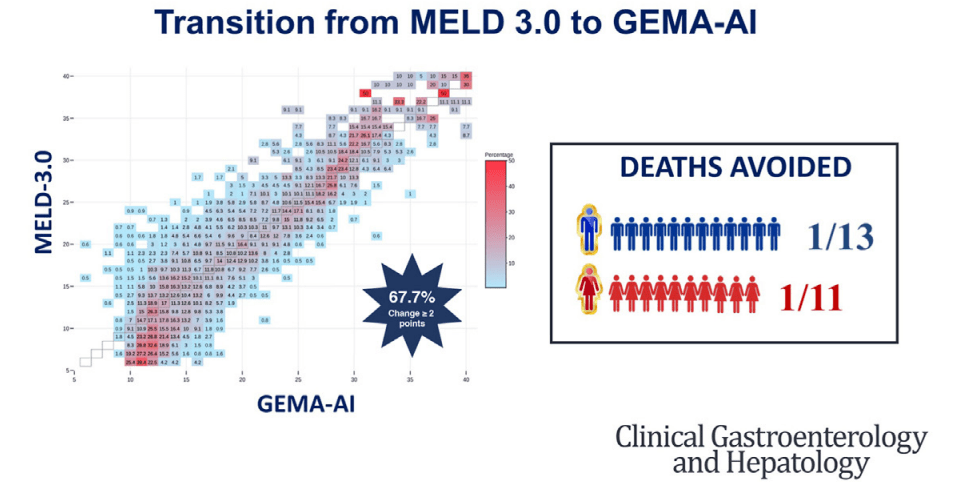
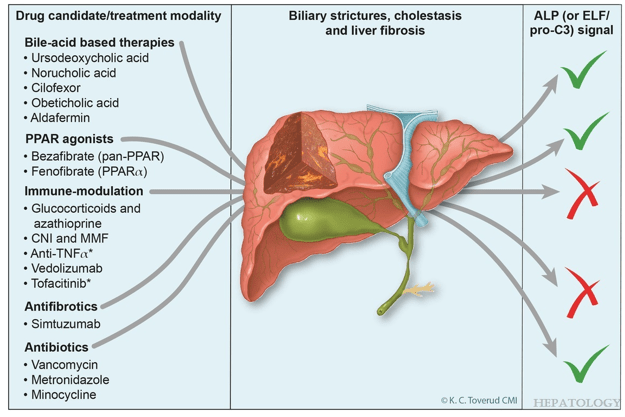
- D Wild. Gastroenterology & Endoscopy News, March 2026: 11.. Open Access! ‘Liver Detox’ Bestsellers Pull in $39M a Year, Despite Thin Evidence
- Referenced study: A Telbany et al. American Journal of Gastroenterology 2026;121[1]:171-178. Liver Cleansing Imposters: An Analysis of Popular Online Liver Supplements
An excerpt:
A cross-sectional study of the 20 highest-ranked purported “liver-cleansing” products sold on Amazon found that they generated almost $40 million in annual revenue, yet their detoxification claims and ingredients were unsupported by data and, in some cases, these products caused adverse events…evidence assessment showed that even the most common ingredients have limited scientific support.
The most common ingredient in the analyzed products was milk thistle, which appeared in 19 of 20 products. This was followed by dandelion root (13) and turmeric root (13). Other, less common ingredients included zinc (12), beetroot (11), artichoke extract (10), choline (10), ginger (9), chicory (8), and berberine (7)…
The investigators recommended “several policy improvements, including mandatory pre-market safety testing, standardized manufacturing practices, enhanced post-market surveillance, and requirements for scientific substantiation of marketing claims.”
My take: Herbal and dietary supplements can cause drug-induced liver injury, sometimes life-threatening. It would be best for those who claim these products are effective to provide proof of this.
Related blog posts:
- Influencer$ and $upplements (2026)
- Are Muscle-Building Supplements Risky for Teens?
- FDA’s Very Limited Ability to Regulate Dietary Supplements