MG Ghany et al. Hepatology 83(4):p 974-997, April 2026. Open Access! AASLD IDSA Practice Guideline on treatment of chronic hepatitis B
Yesterdays’ blog post summarized the 6 key questions that are reviewed in the updated guidelines. Today’s post provides more details on treatment and a direct link to the practice guideline.
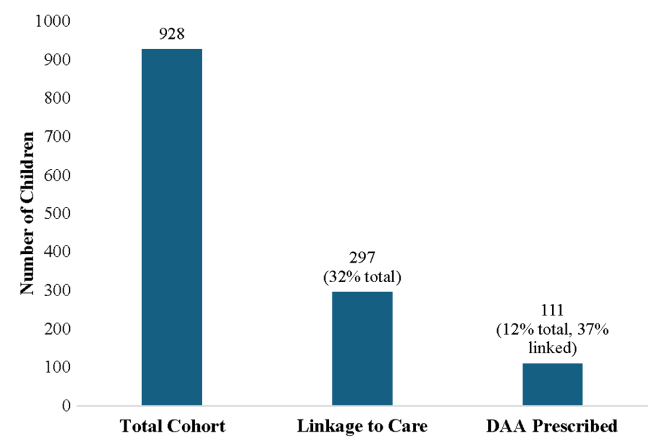
Background: “CHB affects ~258 million individuals (2022) globally8 and leads to an estimated 1.1 million deaths annually due to complications of cirrhosis and HCC…This guideline focuses on six specific issues related to management of CHB. Other recommendations for management of CHB addressed in the 2018 guidelines will continue to be applicable and the reader is referred to this document.1“
Treatment Recommendations for Patients without Cirrhosis Based on HBeAg Results, HBV DNA and ALT Values
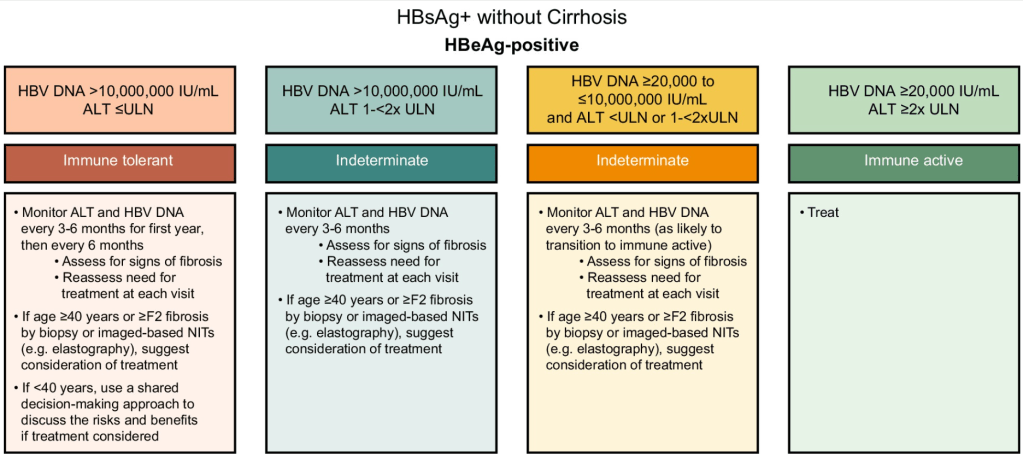
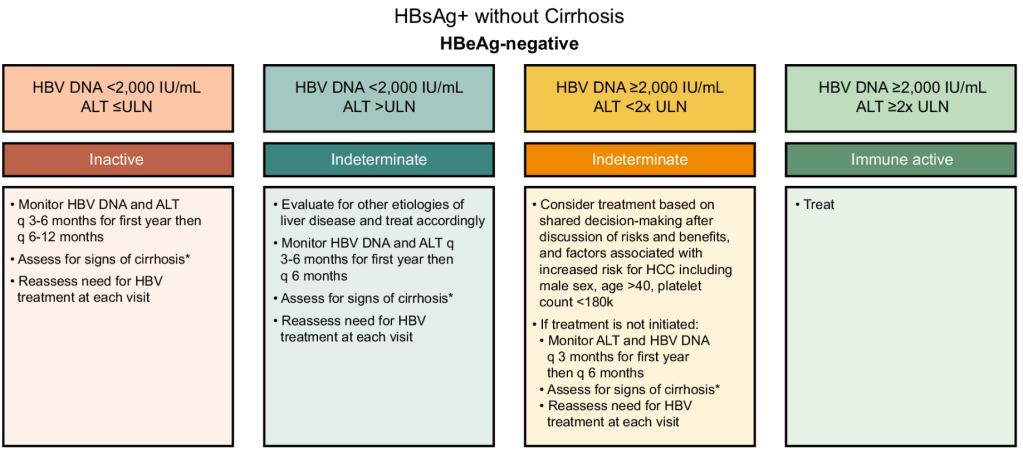
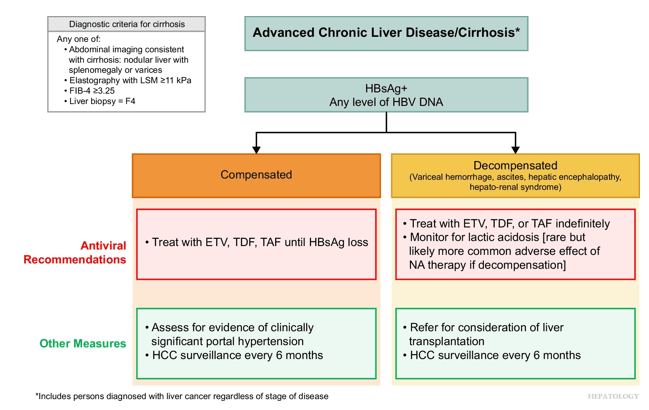
AASLD guidelines favor continuing nucleos(t)ide analogs (NAs) therapy until loss of HBsAg. However, there are additional criteria in new the guidelines for shared decision-making on stopping therapy in those with very low HBsAg levels (<100 IU/mL).
Criteria for stopping NA therapy if ALL are met:

Criteria for resuming therapy immediately (need only 1)

Other Criteria for resuming therapy

Related blog posts:
- Is It Time to Revise Hepatitis B Treatment Guidelines? (2023)
- Comprehensive 2018 AASLD Guidance for Chronic Hepatitis B (2018)
- Scientific Basis for Current Hepatitis B Vaccine Strategy (2025)
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.