A Dimou et al. Hepatology 2024; 80: 266-277. NMR-based metabolomic signature: An important tool for the diagnosis and study of pathogenesis of autoimmune hepatitis
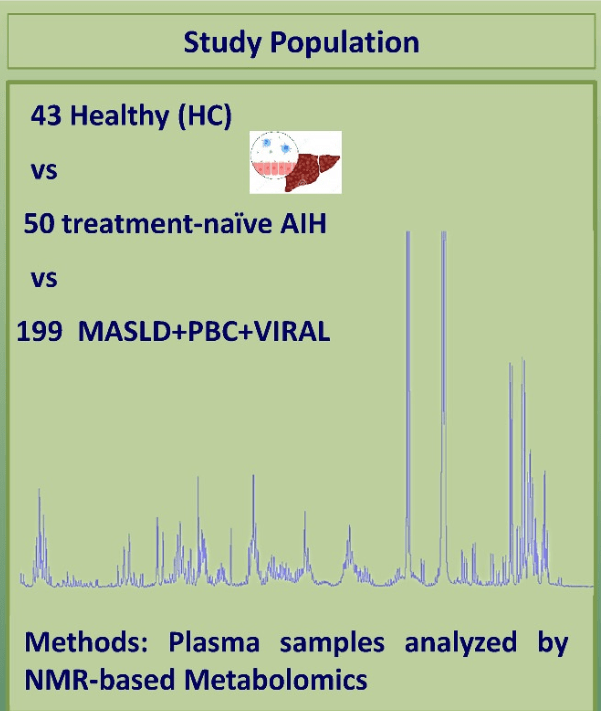
Methods: The authors examined treatment-naive patients with well-established AIH and compared them to healthy controls and those with other liver diseases.
Key Finding:
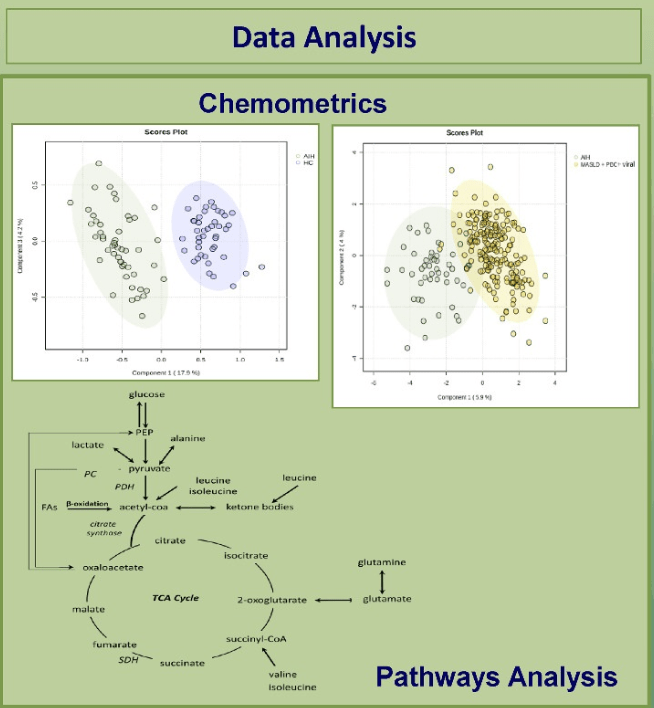
- Fifteen metabolites (out of a total of 52 analyzed) differentiated AIH from disease controls (PBC+chronic viral hepatitis+metabolic dysfunction–associated liver disease) (95% sensitivity and 92% specificity)
In their discussion, the authors review the metabolism of the various metabolites and why they may be altered in AIH. “Our study found that cirrhosis did not seem to affect our results.” In ongoing studies, the authors are trying to determine how these metabolites change with treatment and whether they could be a predictive marker.
My take: Metabolite measurement could be helpful in the diagnosis of AIH as “NMR technology dose not need much sample handling, is highly reproducible, and with low costs.
Related blog posts:
- Diagnosing autoimmune hepatitis
- Is First Line Therapy for Autoimmune Hepatitis Changing? CAMARO Study Results
- Predicting Outcomes in Childhood Autoimmune Hepatitis
- Online Aspen Webinar -COVID-19, Autoimmune Hepatitis (Part 8) (2020)
- 6-Thioguanine Levels in Autoimmune Hepatitis The associated editorial (pg 2-3, N Kerkar) cautions that while “lower levels are sufficient for maintaining biochemical remission…higher levels, similar to that used in IBD, are required for inducing remission.”
- Immune Mediated Disorders Associated with TNF Inhibitors Can Involve the Liver Too