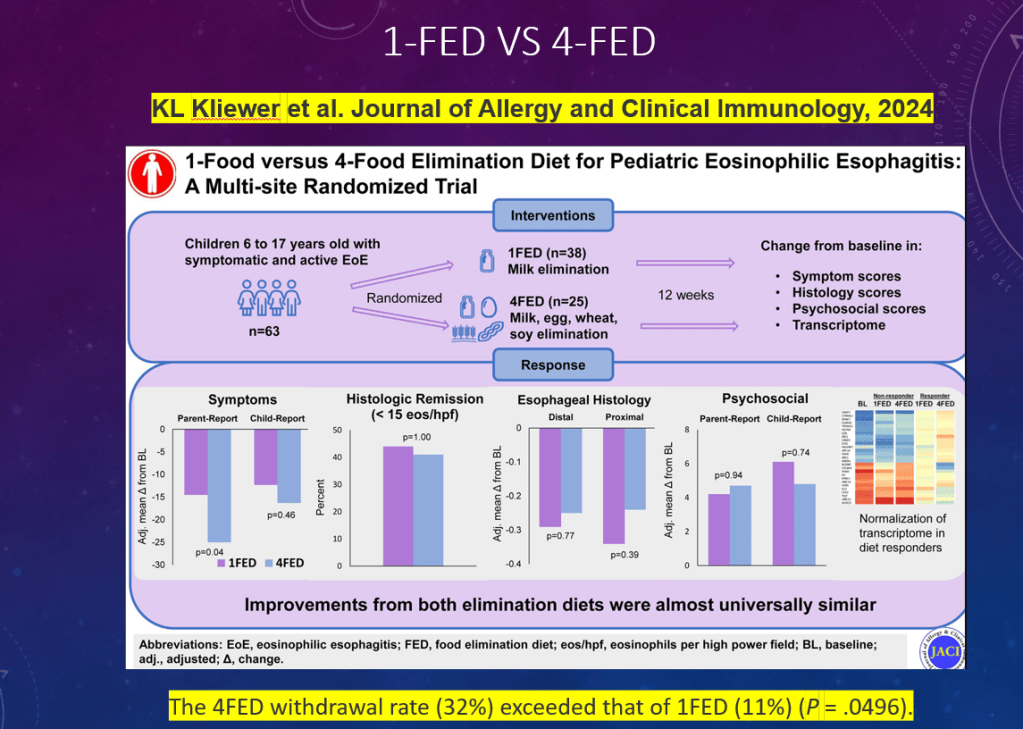
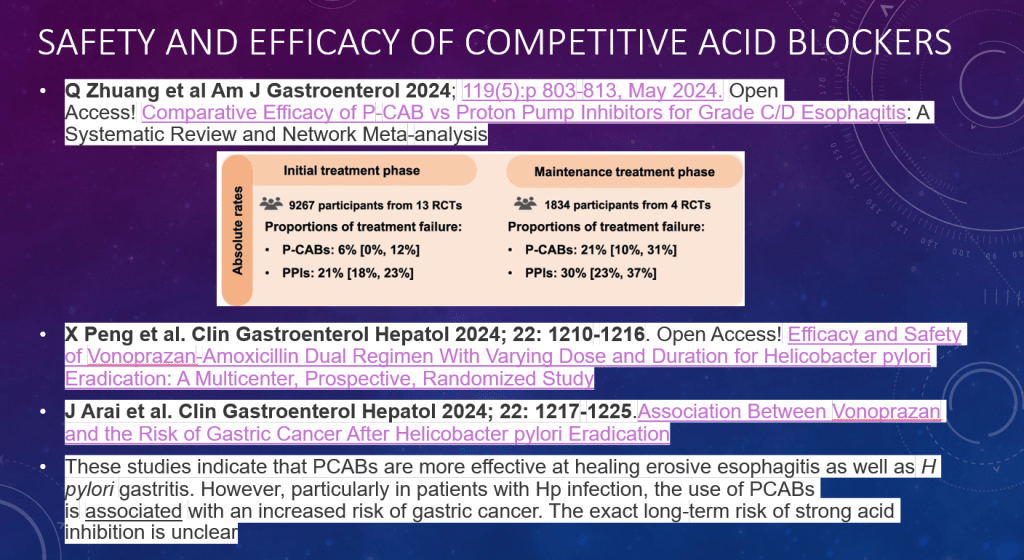
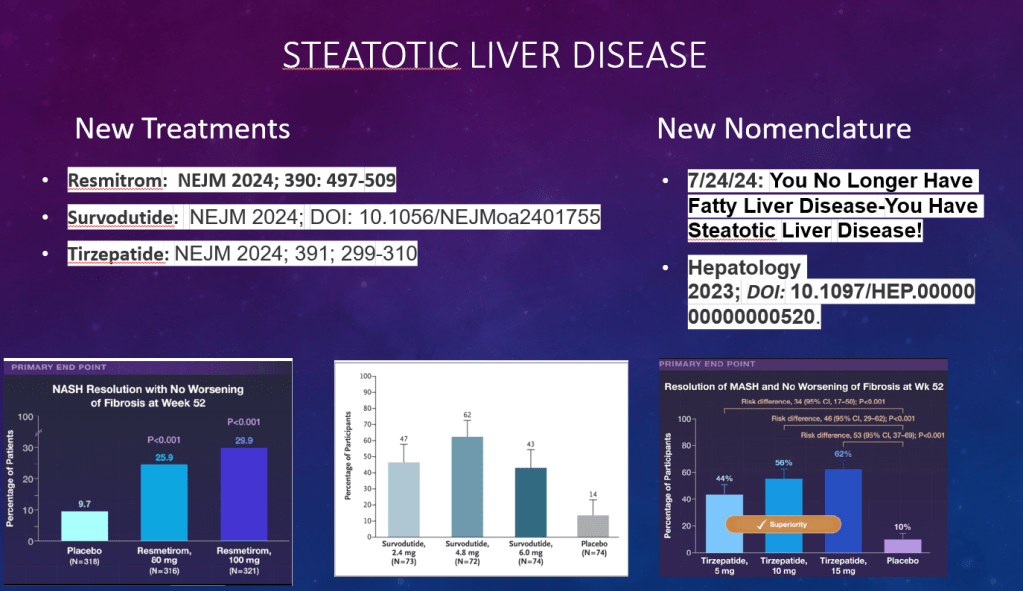
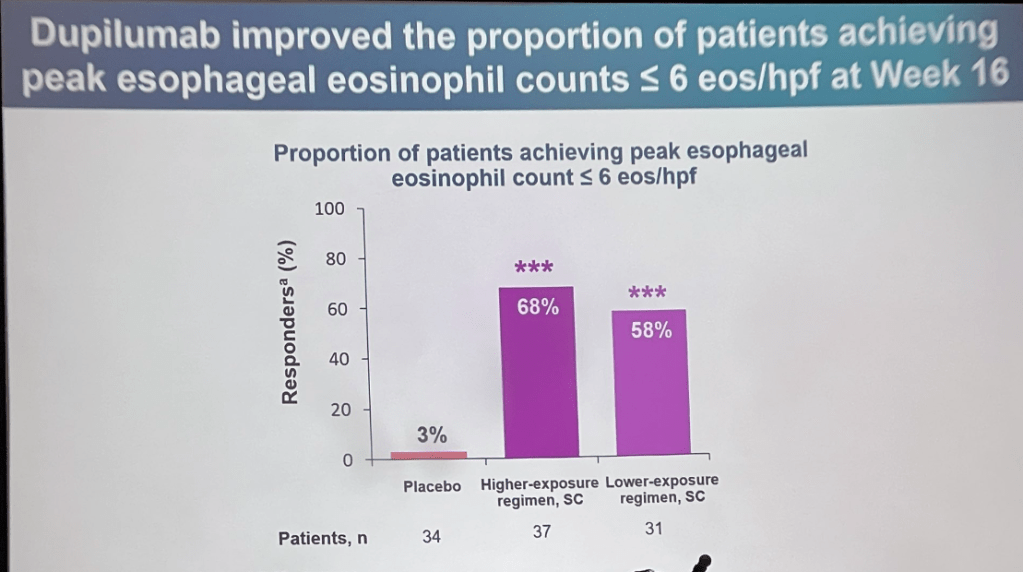
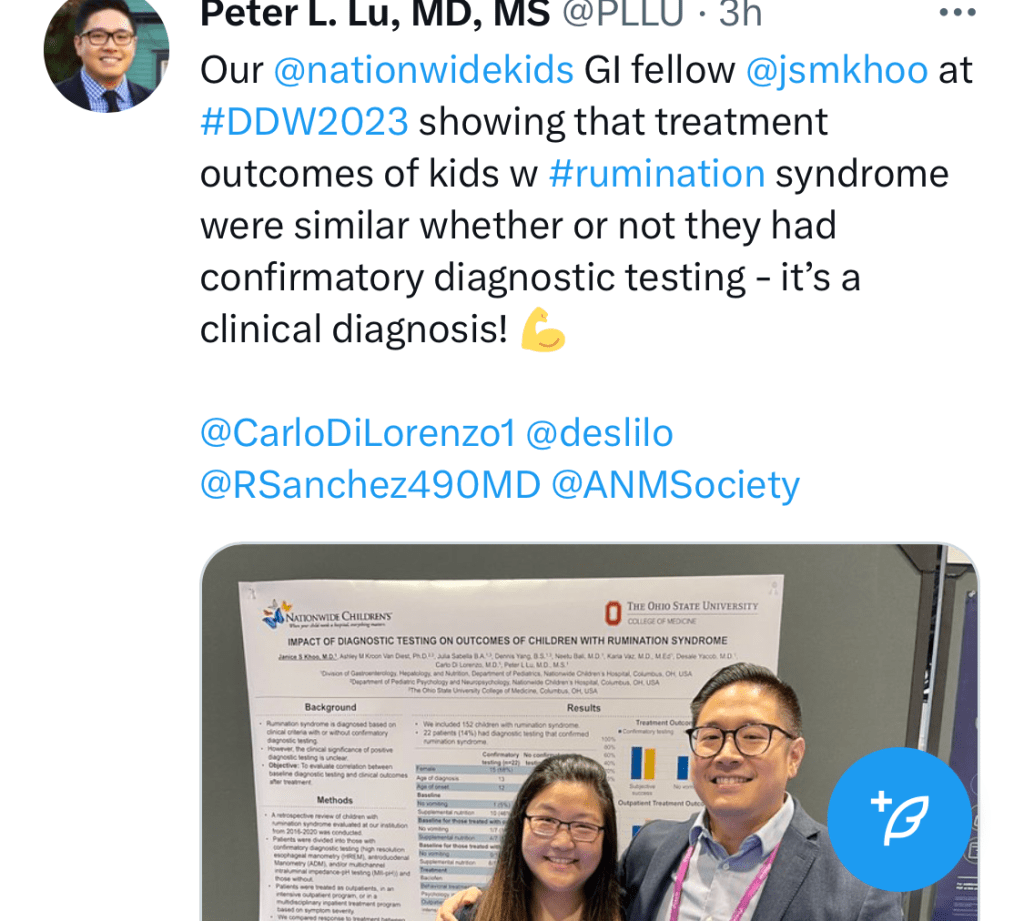
C Thellin, Y Urrutia. Gastroenterology and Endoscopy News, March 17, 2026. Open Acess! ‘Uncorking’ the Esophageal Food Impaction: Why Twisting Works Better Than Pulling
An excerpt from this recent article:
Yet, just as one wouldn’t simply yank a cork from a bottle, it may be time to rethink how we approach food bolus removal. Torsional stress—twisting rather than pulling—may provide a more efficient and atraumatic method for managing these impactions, a technique we refer to as the “corkscrew method.”
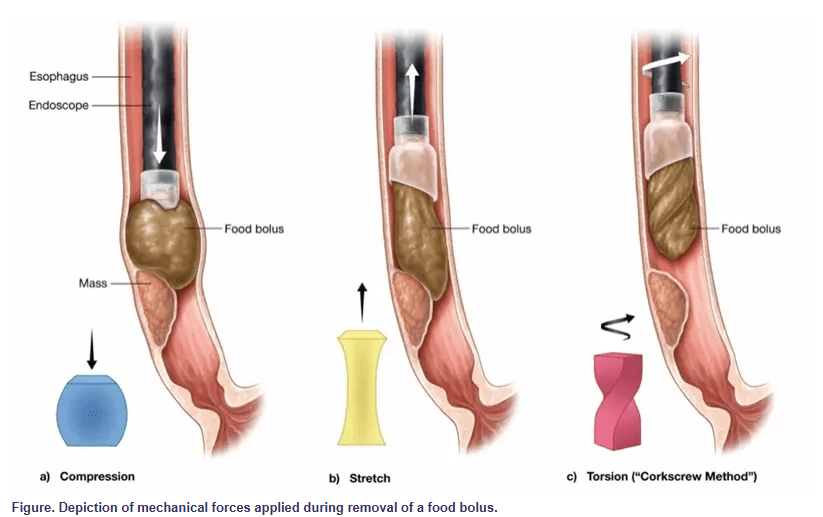
Less force is required because rotation gradually disengages the bolus, decreasing the need for strong pulling or pushing. This method also poses a lower risk for mucosal injury. Shear forces are distributed rather than focused, reducing trauma.
My take: This is probably worth a try for the next food impaction requiring endoscopic removal.
Related blog posts:
- Practical Tips for Eosinophilic Esophagitis
- Adverse Events Following Pediatric Endoscopy –Underestimated Previously
- Foreign Bodies in Children -Expert Guidance
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.