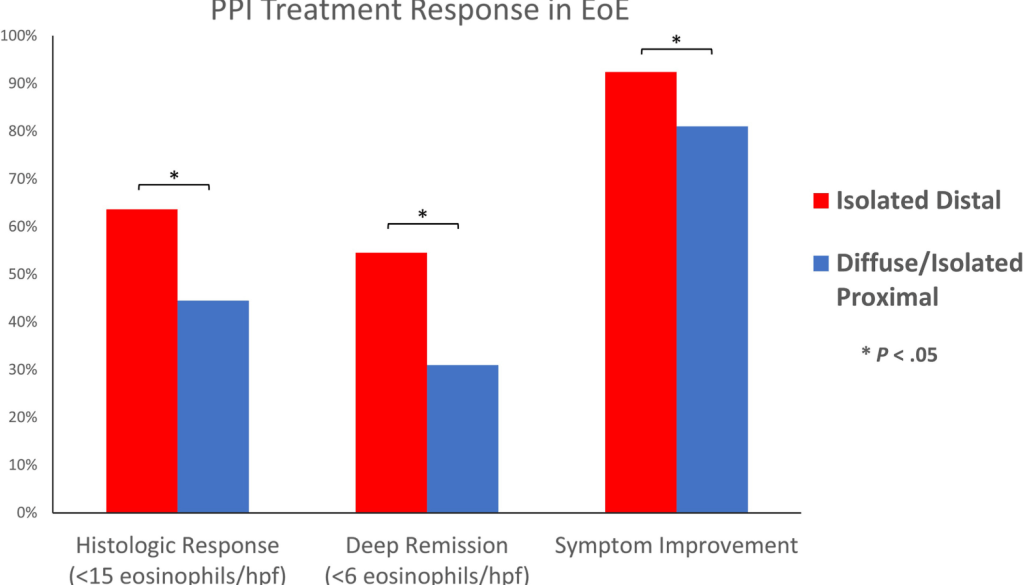
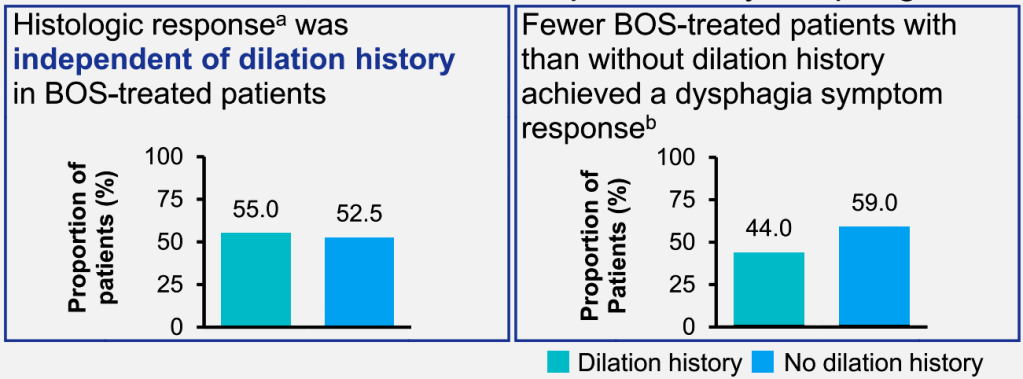
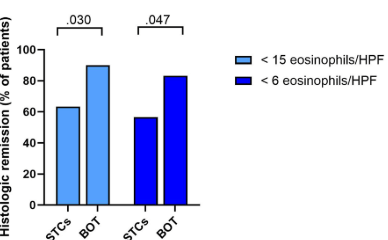
Since I completed my pediatric residency and pediatric gastroenterology fellowship in Cincinnati, I receive their bulletins. A recent title (Link: EoE: The Fifth Member of the Atopic March) prompted me to review the source article. The Cincinnati summary noted that “children with AD [atopic dermatitis] within the first two years of life are 10 times more likely to develop EoE [eosinophilic esophagitis] by age 8.”
Source article: WC Chang et al. Journal of Allergy and Clinical Immunology 2026; 157: 999-1001. Open Access! Longitudinal enrichment of eosinophilic esophagitis in children with AD: The MPAACH cohort
Background: The atopic march refers to the natural history of allergic disease manifestations as they develop in childhood.1 Classically, the march begins with atopic dermatitis (AD), followed sequentially by food allergy (FA), asthma, and allergic rhinitis (AR),1 but there is significant heterogeneity in the timing, order and organ(s) affected.2
Methods: Using the Mechanisms of Progression of Atopic Dermatitis to Asthma in CHildren (MPAACH) cohort,4 a US longitudinal early-life cohort of children (n=700) with AD, the authors examined the associated epidemiology of EoE with other members of the atopic march.
Key findings:
- “Of 700 MPAACH participants, 10 have EoE, and in all cases, EoE developed after AD onset. Of these 10 subjects, 2 had EoE at enrollment, whereas 8 developed EoE subsequent to enrollment in MPAACH”
- “To date, 48.9% of the children in MPAACH had developed at least 1 allergic comorbidity (FA, AR, and/or asthma) by age 8 years versus 90% of the children with EoE (P = .009), highlighting the strong association between EoE and other atopic diseases”
- “Despite no difference in skin barrier quality or AD severity, the children with EoE were significantly more likely to have food sensitization (as defined by at least 1 positive result of a skin prick test to a food allergen [60% vs 28% (P = .039)]) and FA (70% vs 13% [P < .001])…suggesting that the esophageal epithelium may be the site of allergen penetration and immune activation, independent of the skin barrier”
My take: There was a 10-fold higher risk of EoE (1.4% or 10 of 700) in this cohort of children with AD compared to the general population. However, I was expecting a much higher prevalence in this population. This could be due to the fact that patients are seeing me for GI symptoms rather than for AD. It is possible, as well, that the number of EoE patients would be higher if there was a prospective evaluation (e.g. endoscopy).
Related blog posts:
- Quality Forum: Understanding Food Allergy Testing (Part 2) & Atopic Dermatitis
- Quality Forum: Understanding Food Allergy Testing (Part 1)
- “The Truth About Allergies and Food Sensitivity Tests”
- Poorly-Conceived Allergy Testing Can Lead to Unnecessary Diet Restrictions and Complications | gutsandgrowth
- Best Allergy Articles 2021 (Part 5): Allergy Test Ordering | gutsandgrowth
- Eczema Rarely Linked to Food Allergy Dave Stukus: Many parents are told that if they can find the ‘cause’ of their child’s eczema and eliminate exposure, then their skin will improve. Unfortunately, this is not the case because the cause of eczema is a disrupted skin barrier, which leads to excessive water loss, dryness and itching. Children with eczema, especially those with persistent, severe cases affecting most of their body, are at higher risk to develop allergies and asthma as they get older….In rare instances, specific foods may be a major contributor to a child’s eczema, but this is the exception and typically affects infants less than one year of age with truly unmanageable, severe eczema, despite good daily skin care.
- How Allergy Testing Can Lead to More Allergies
- What’s Wrong with “I Want My Kid Tested For Food Allergies”
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition