T Mayr et al. JPGN 2021; 72: 115-122. Optimized Trientine-dihydrochloride Therapy in Pediatric Patients With Wilson Disease: Is Weight-based Dosing Justified?
In this retrospective study with 31 children with Wilson’s disease (most of whom had had previous penicillamine), those who received more than 20 mg/kg/day of trientine therapy had increased adverse effects compared to those who received less than 20 mg/kg/day: 63% vs 7%; median followup was 60 months. In addition, there was not increased response to higher doses. The authors note that trientine had lower incidence of adverse effects compared to penicillamine and “appears to be the preferred” as a first-line treatment.
J Teckman et al. (ChiLDReN Network). J Pediatr 2020; 227: 81-86. Longitudinal Outcomes in Young Patients with Alpha-1-Antitrypsin Deficiency with Native Liver Reveal that Neonatal Cholestasis is a Poor Predictor of Future Portal Hypertension
In this prospective cohort with 350 participants (all with either PiZZ (90%) or PiSZ (10%) and native livers), 278 (79%) entered the cohort (in 2007 or later) without portal hypertension and 18 developed portal hypertension during follow-up. Portal hypertension was defined by development of ascites, varices or combination of splenomegaly/thrombocytopenia. Thirty participants required liver transplantation; 2 patients died during 1077 person-years of follow-up. Median length of followup was 2.5 years. My take: While most children with Alpha-1-Antitrypsin Deficiency do well, monitoring is warranted as some will develop progressive liver disease (even in the absence of neonatal cholestasis).
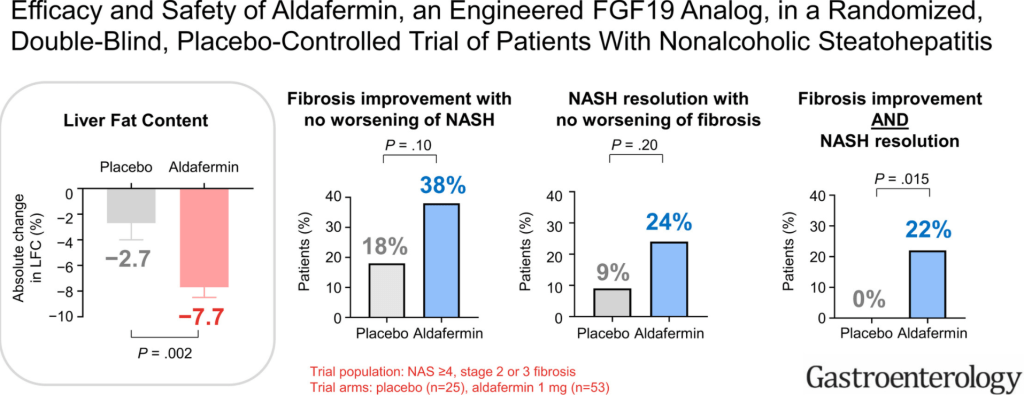
SA Harrison et al. Gastroenterol 2021: 160: 219-231. Full text PDF: Efficacy and Safety of Aldafermin, an Engineered FGF19 Analog, in a Randomized, Double-Blind, Placebo-Controlled Trial of Patients With Nonalcoholic Steatohepatitis
In this phase 2 double-blind study with 78 patients with NASH, at week 24, the aldafermin group had a significant reduction in absolute liver fat content (reduction of 7.7%) compared with placebo (reduction of 2.7%) (P=.002). Fibrosis improvement (1 stage) with no worsening of NASH was achieved in 38% of patients receiving aldafermin vs 18% of
patients receiving placebo (P = .10). And, NASH resolution with no worsening of fibrosis was observed in 24% of patients given aldafermin vs 9% of patients given placebo (P = .20)
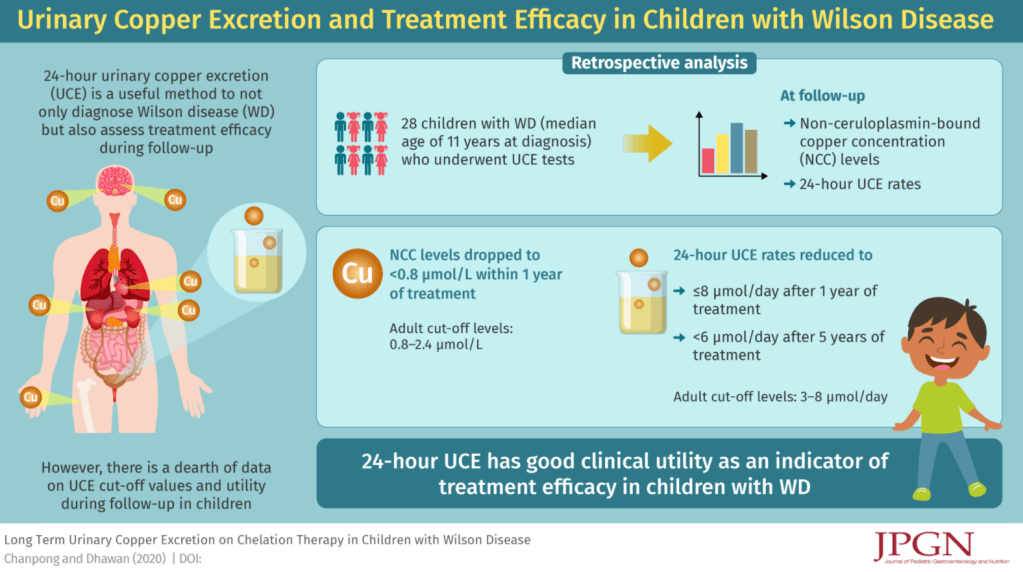
A Chanpong, A Dhawan. JPGN; 2021: 72: 210-215. Long-Term Urinary Copper Excretion on Chelation Therapy in Children with Wilson Disease Key finding: 24-hour UCE decreases to ≤8 μmol/day and <6 μmol/day after 1 and 5 years of treatment, respectively.
Related blog posts:
- Wilson’s Disease -Pediatric Guideline
- Finding the Right Specialist | gutsandgrowth This post has link to AASLD guidelines for Wilson disease.
- Data on Chelators for Wilson Disease | gutsandgrowth
- How Effective is Zinc Therapy for Wilson’s Disease
- Liver Shorts March 2020 (A1AT Heterozygosity worsens NAFLD/contributes to cirrhosis)
- Alpha-1-Antitrypsin Deficiency (review May 2020)
