We had a terrific lecture given to our group by Dr. Bonney Reed. She is a pediatric psychologist with a clinical and research focus on children with inflammatory bowel disease. Our group has worked closely with Dr. Reed for many years. Many of her slides are included below along with my notes; my notes may contain errors in transcription and in omission.

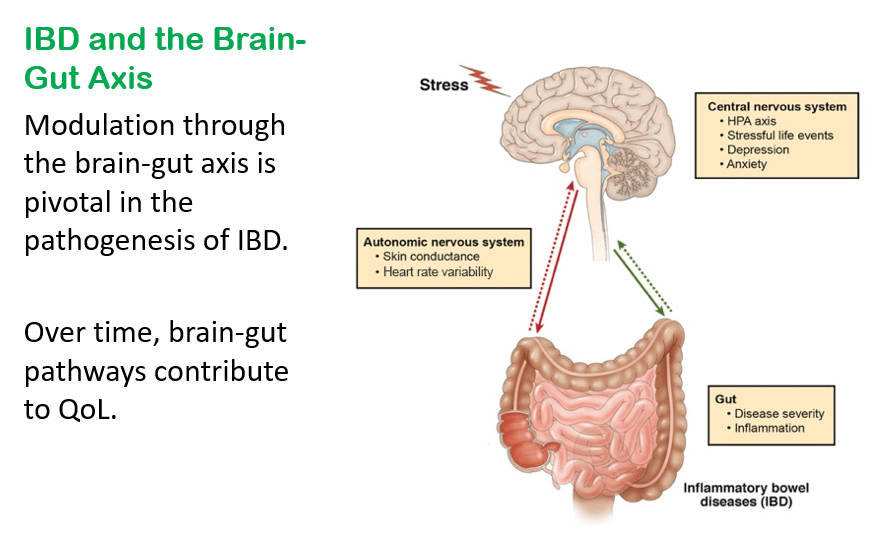
- GI symptoms may begin as the result of organic disease (e.g., IBD). Anxiety and chronic activation of the stress response system may lead to alterations in the brain, spinal cord, and gut increasing the load of GI symptoms. In turn, distress associated with GI symptoms may contribute to anxiety or depressed mood, creating a cycle of worsening GI symptoms and overall psychological distress.
- Consistent with a brain-gut axis model, individuals with IBD, compared to healthy controls, demonstrate dysfunction of the ANS indicative of a chronic stress response which is characterized by increased sympathetic nervous system (SNS) activity and reduced parasympathetic nervous system (PNS) activity
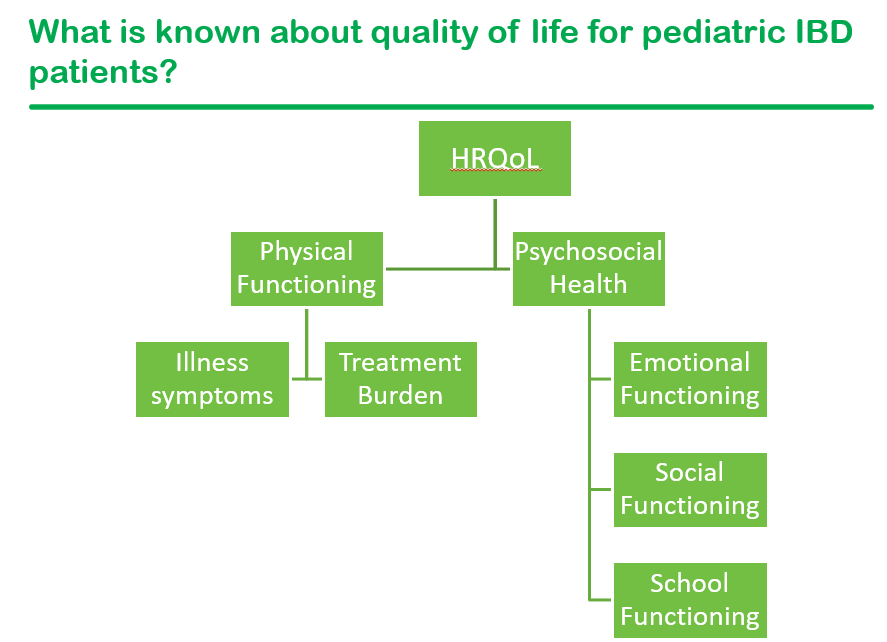

- Psychological factors are the key factor for pediatric patients with IBD when self-rating their global health
- Factors that contribute to an individual’s current QoL: symptom exacerbation, psychological functioning including stress, and family support.
- Health-related quality of life factors: major life transitions (eg. graduating high school and needing to manage IBD at college), fatigue ( persists despite controlled inflammation), poor body image (especially with weight changing rapidly), a diminished self-perception or seeing oneself as less capable, comorbid functional abdominal pain (about a quarter of youth with IBD), and food restrictions that can interfere with daily quality of life.
- Stress plays important role influencing (bidirectional) disorders of brain gut interaction (DBGI)
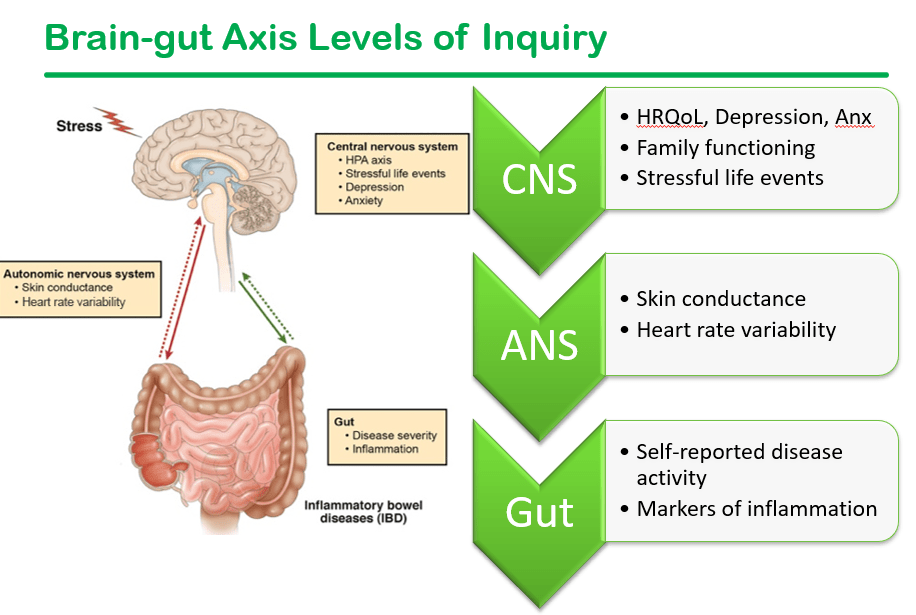


- Dr. Reed’s research includes a longitudinal cohort of newly-diagnosed (w/in 45 days) pediatric patients with IBD. This cohort undergoes psychosocial assessment along with ANS assessment
- Emotional reactivity indicates individuals with a ‘short fuse’ who take longer to return to normal. Those with emotional reactivity are at increased risk for anxiety/depression.
- Skin conductance response (SCR) can help determine autonomic nervous system (ANS) dysfunction. It is a measure of sympathetic arousal and stress
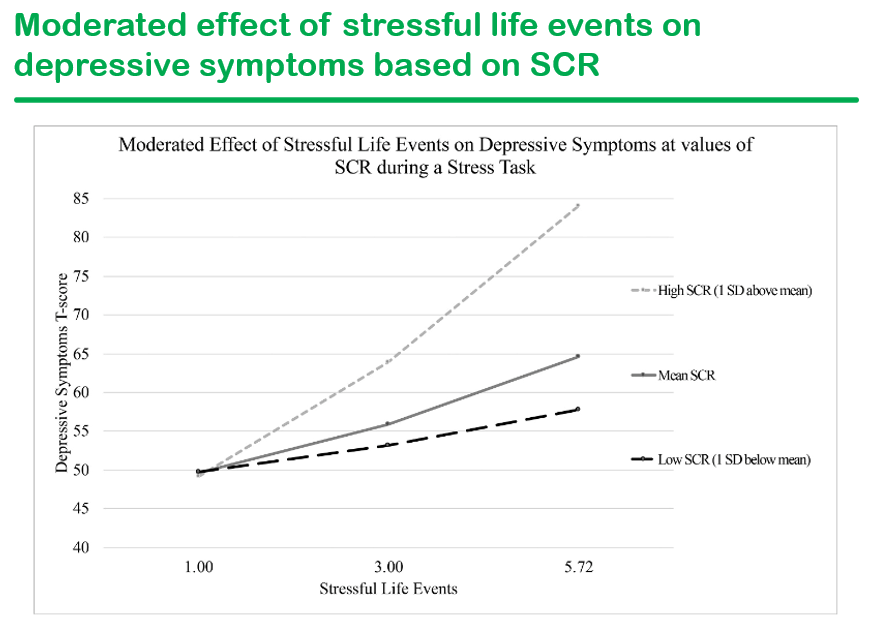
- Stressful life events increase the rates of depression and correlate with skin conductance at medium and high levels
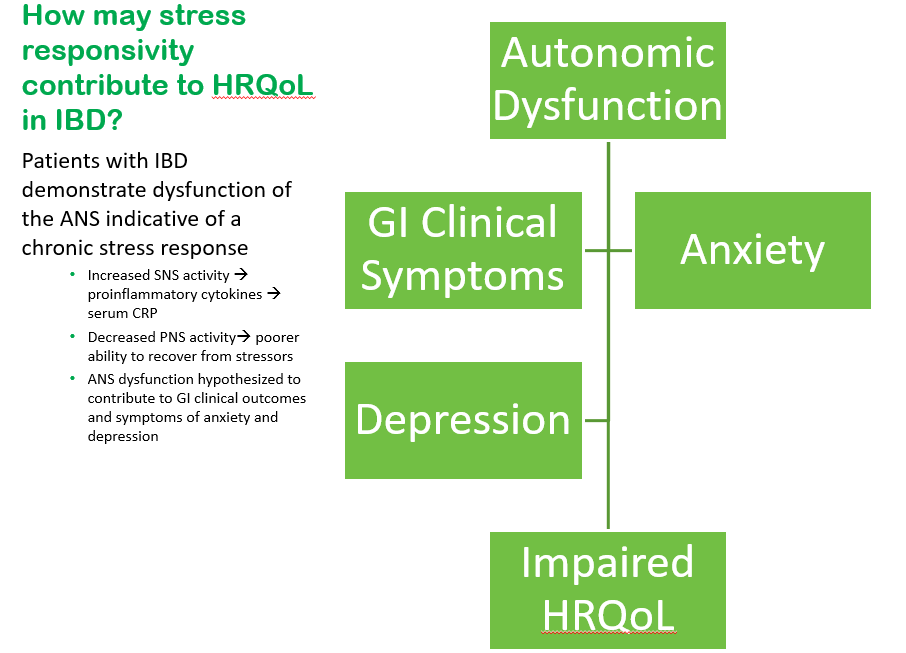
- Within this model, Dr. Reed’s research focuses on the hypothesis that autonomic dysfunction is indicative of a chronic stress response. This, in turn, contributes to increased sympathetic nerve activity and decreased parasympathetic activity. This contributes to symptoms of anxiety and depression as well as GI clinical symptoms, all of which lead to impairments in QoL. Addressing autonomic dysfunction may provide a mechanism by which to address all of these QoL drivers
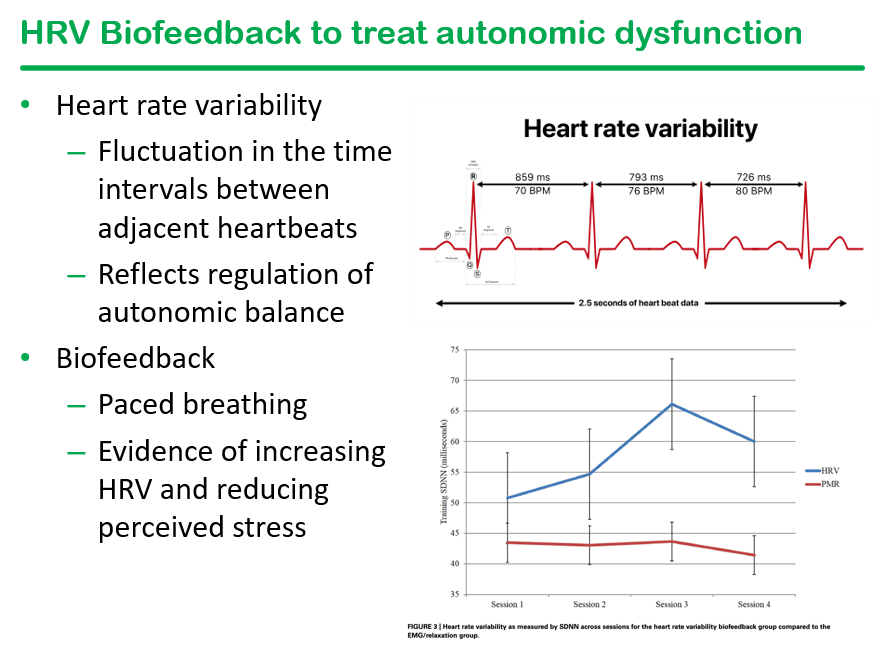
- ANS dysfunction (which is also seen in cyclic vomiting syndrome) can improve with biofeedback focused heart rate variability (HRV). HRV, in turn, is associated with increase inflammation

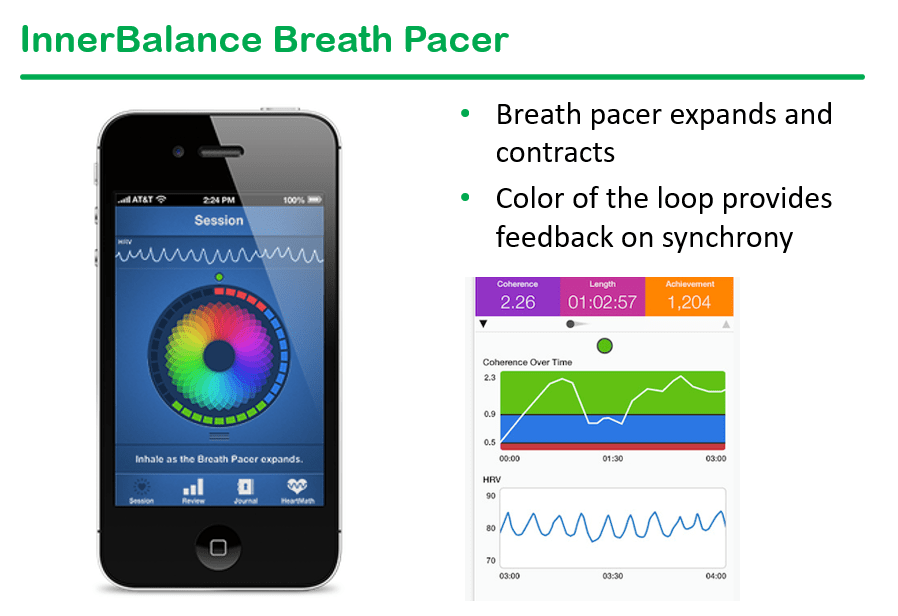
- Preliminary data from breath pacer intervention has shown in improvement in multiple variables

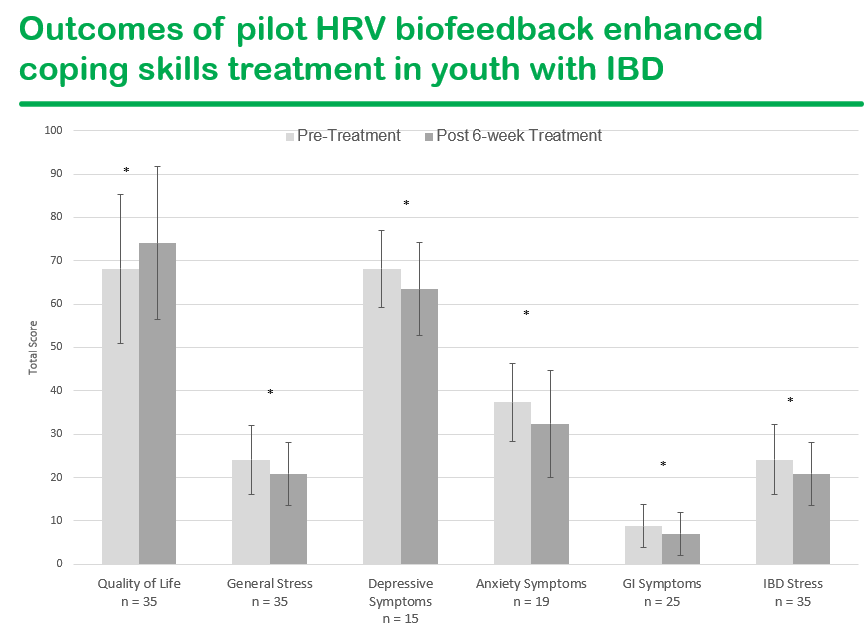
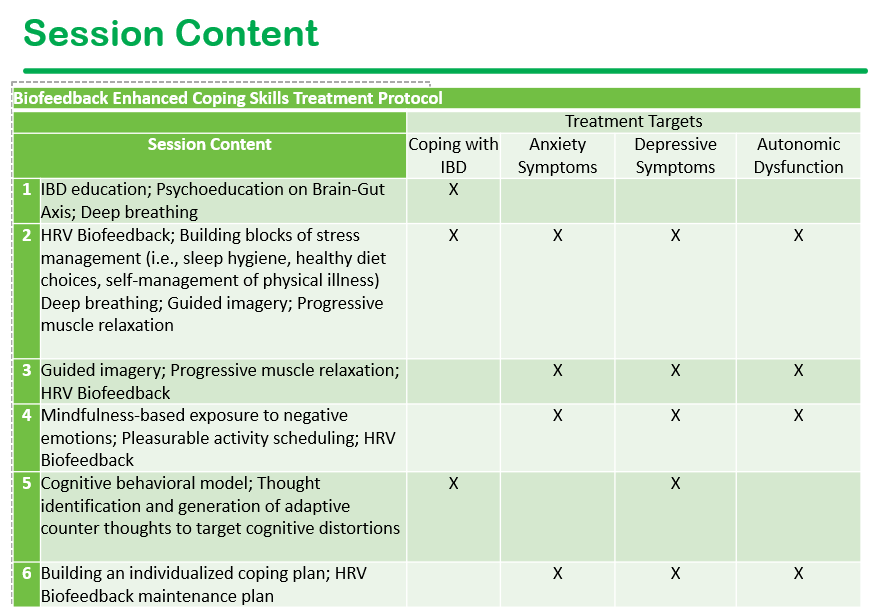
- Research published in past year — link: Biofeedback-Enhanced Cognitive-Behavioral Therapy Delivered Virtually to Youth With Inflammatory Bowel Disease: A Randomized Controlled Trial Inflamm Bowel Dis. 2025 Oct 1;31(10):2735–2745. Conclusion in study: “This pilot study supports the feasibility and acceptability of a virtual, group-based HRV biofeedback enhanced coping skills intervention for youth with IBD. Preliminary efficacy was demonstrated for reducing psychological and physical symptoms”

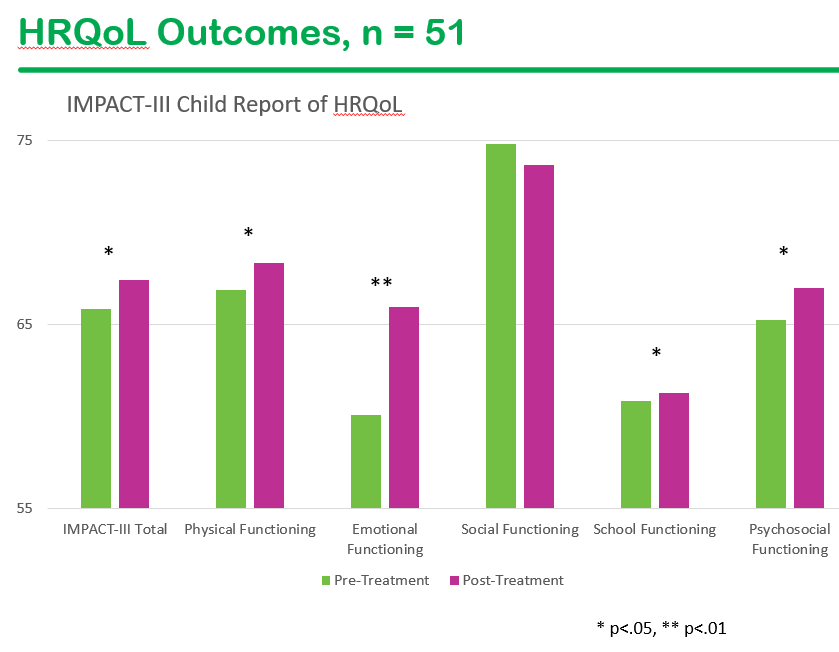
- Research link: Health-related quality of life in youth with chronic gastrointestinal disease following a biofeedback enhanced cognitive behavioral therapy intervention: A randomized controlled trial. J Pediatr Gastroenterol Nutr. 2026;82:672–683. Conclusion: “This study offers preliminary support for a biofeedback-enhanced, coping skill intervention for improving patient-reported HRQOL outcomes in youth with IBD.”
Related blog posts: