Recently, Dr. Stacy Kahn gave our group an excellent update on Clostridioides difficile. My notes below may contain errors in transcription and in omission. Along with my notes, I have included many of her slides. Dr. Kahn has been a leader on treatment and advocacy for C. difficille. In 2025, she received the Leadership Award from the Peggy Lillis Foundation recognizing her clinical, research and advocacy efforts related to C. difficile awareness and treatments.

Key points:
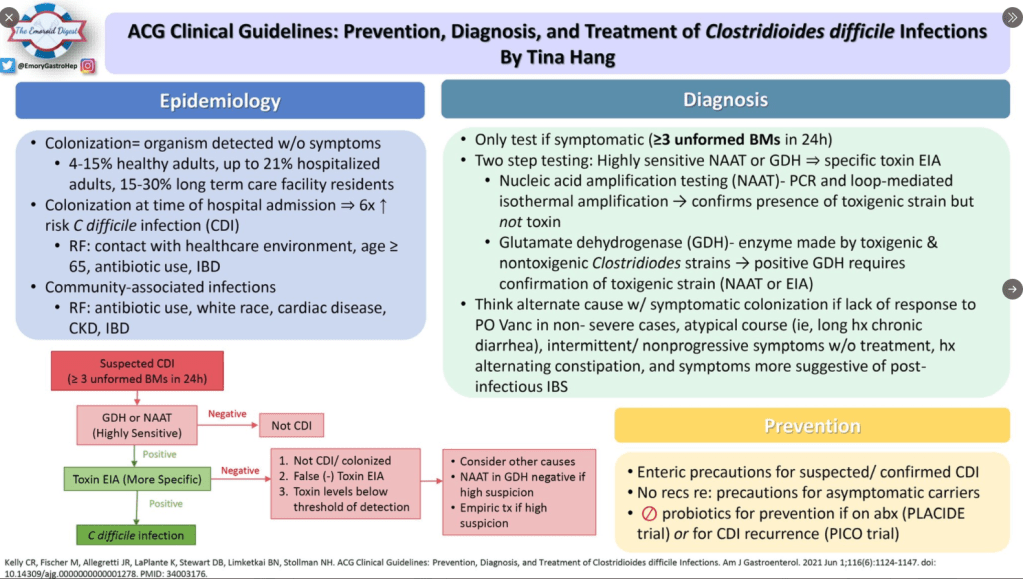
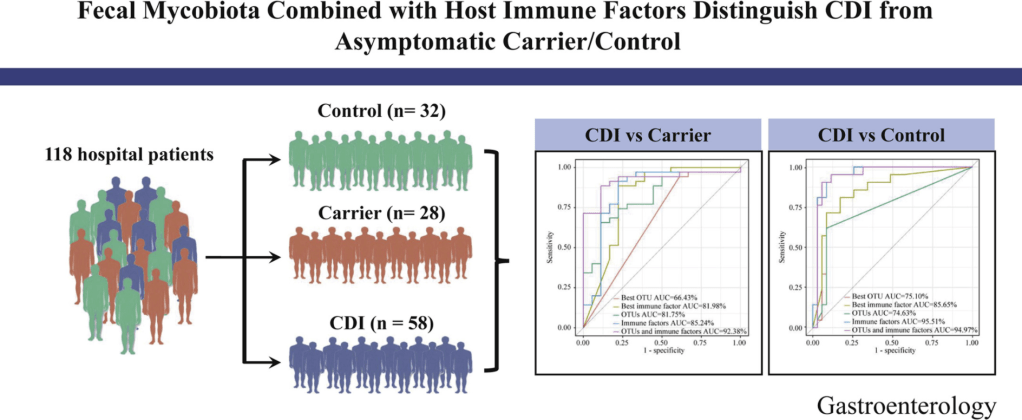
Diagnosis: C. diff is difficult to diagnose. The NAAT-based assays are highly sensitive but cannot readily distinguish active infection from colonization. ELISA toxin assays have higher specificity. However, there are many of these assays and their reliability in identifying active infection from colonization varies. In individuals with underlying diseases like IBD and IBS, this can create uncertainty about the diagnosis of C. diff.
Presentation: Symptoms are quite variable, from asymptomatic to bloody diarrhea to fulminant colitis (uncommon in kids). Profound urgency is a common feature.


Transmission: C. diff bacteria can survive on surfaces for 24 hrs. The spores can survive months to years. In addition, the (invisible) spores are highly resistant to heat, disinfection and antimicrobials. Thus, nursing homes and hospitals are frequent reservoirs.


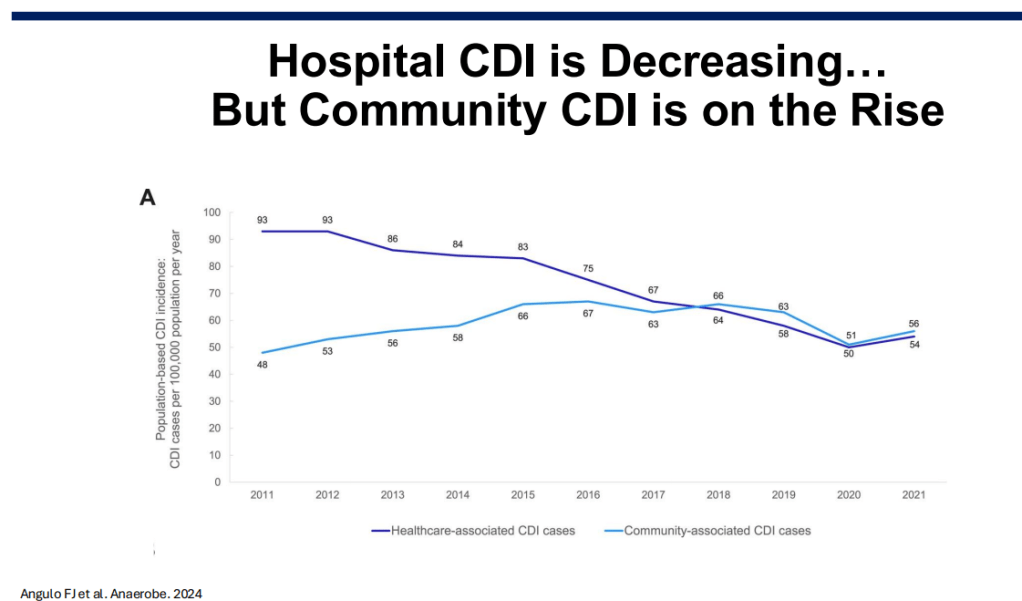
Epidemiology: C diff rates in the hospital setting have improved, likely due to antibiotic stewardship. Community rates have increased; though, precise estimates are problematic as the diagnostic testing is not straightforward.
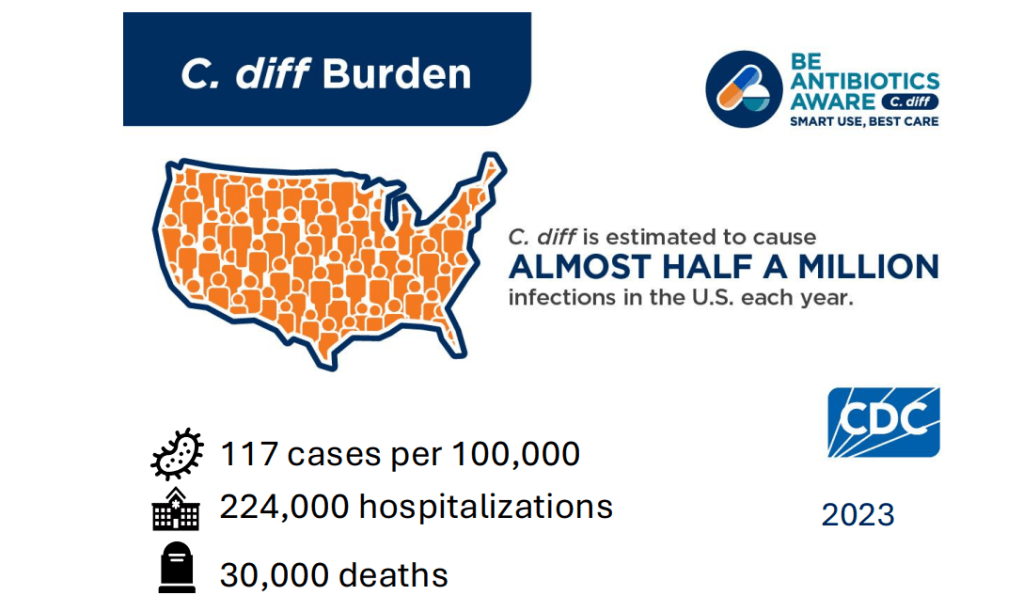
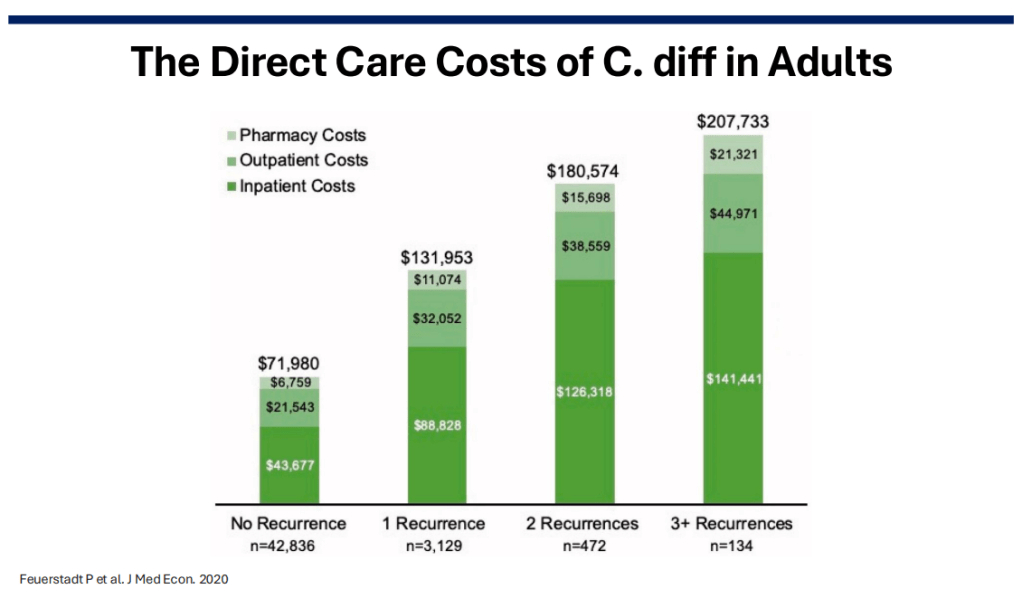
Costs: In 2016, the estimated annual costs due to C diff were $ 6.3 billion (Zhang S. et al. BMC Infect Dis. 2016).
Resources/Websites:
- CDC website has good information for families and clinicians.
- Peggy Lillis Foundation is another good website.
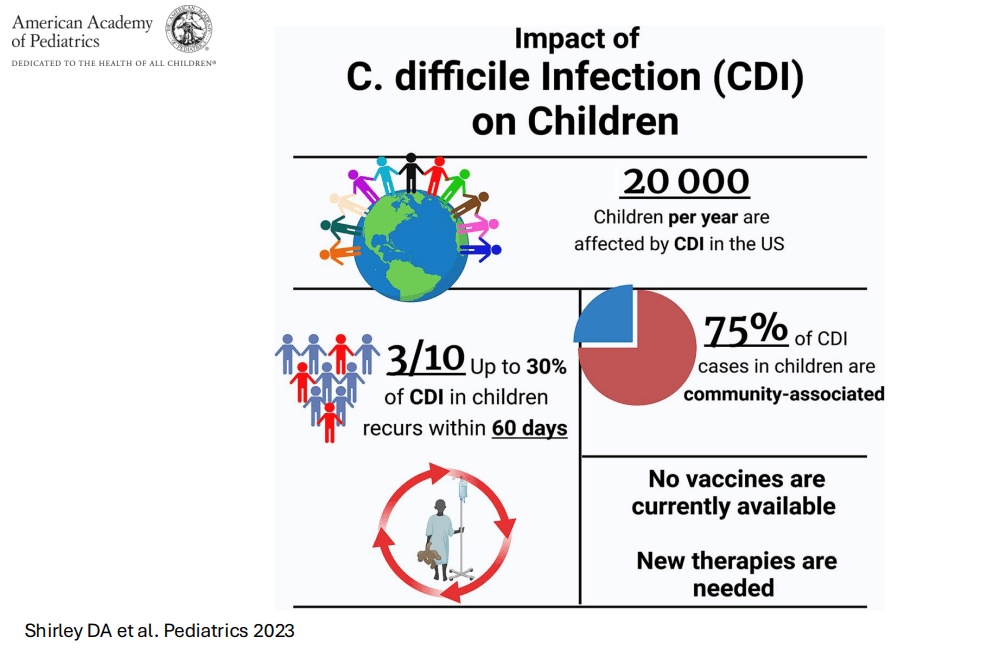
- For clinicians, a good pediatric review: DA Shirley et al. Pediatrics. 2023 Aug 10;152(3):e2023062307. doi: 10.1542/peds.2023-062307. Open Access! Clostridioides difficile Infection in Children: Recent Updates on Epidemiology, Diagnosis, Therapy
- AGA FMT resource page/frequently asked questions
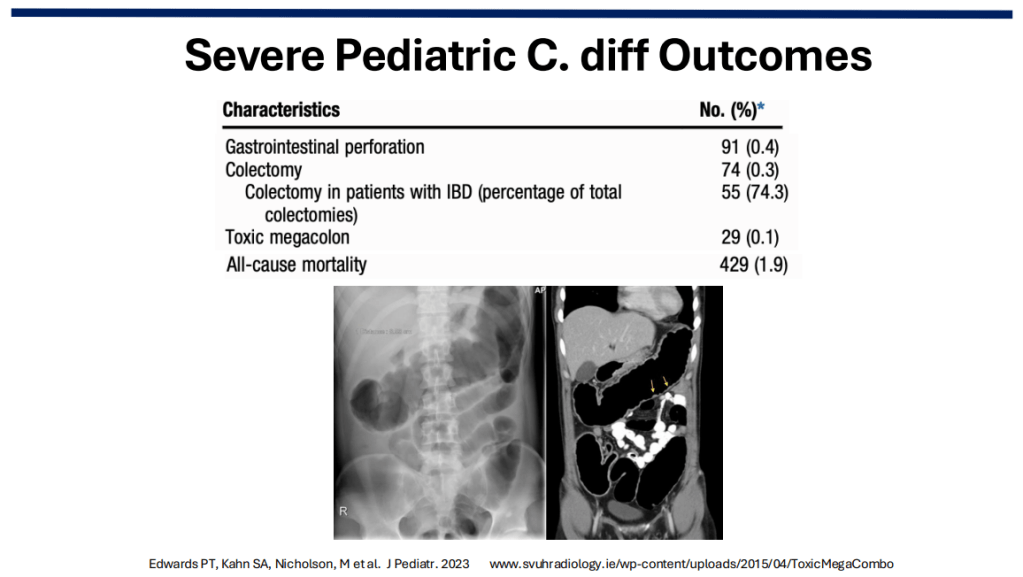
Severe C diff in Children: In a retrospective study of C diff in hospitalized children (2013-2019, n=17,142 children) showed that among 23,053 CDI admissions, 74 (0.3%) had a colectomy (55 in IBD patients), and 29 (0.1%) had toxic megacolon. All-cause mortality was noted in 429 (1.9%) (Reference: Edwards PT, Kahn SA, Nicholson, M et al. J Pediatr. 2023; 252:111-116.e1. Open Access! Clostridioides difficile Infection in Hospitalized Pediatric Patients: Comparisons of Epidemiology, Testing, and Treatment from 2013 to 2019).
Testing: Recommendations include avoiding testing in those taking laxatives; however, an exception to this would be patients with motility disorders. Even combination testing cannot always distinguish between colonization and active infection. In addition, there are numerous toxin tests with variable performance.
[From prior blog post: In a large adult study with 293 of 1416 hospitalized adults testing positive for C. diff, virtually all CDI-related complications and deaths occurred in patients with positive toxin immunoassay test results. Patients with a positive molecular test result and a negative toxin immunoassay test result had outcomes that were comparable to patients without C difficile by either method. (Overdiagnosis of Clostridium difficile with PCR Assays)]


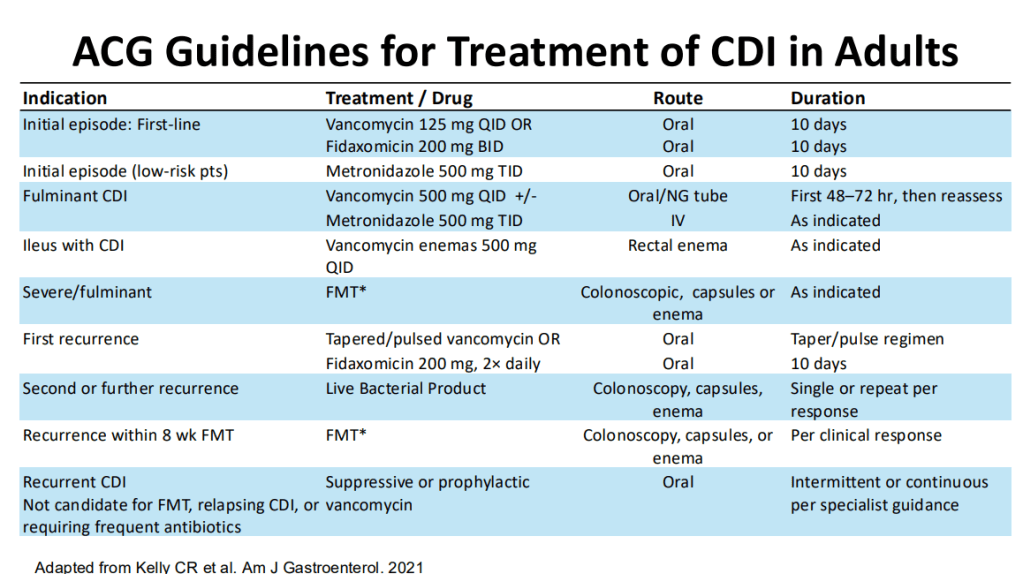
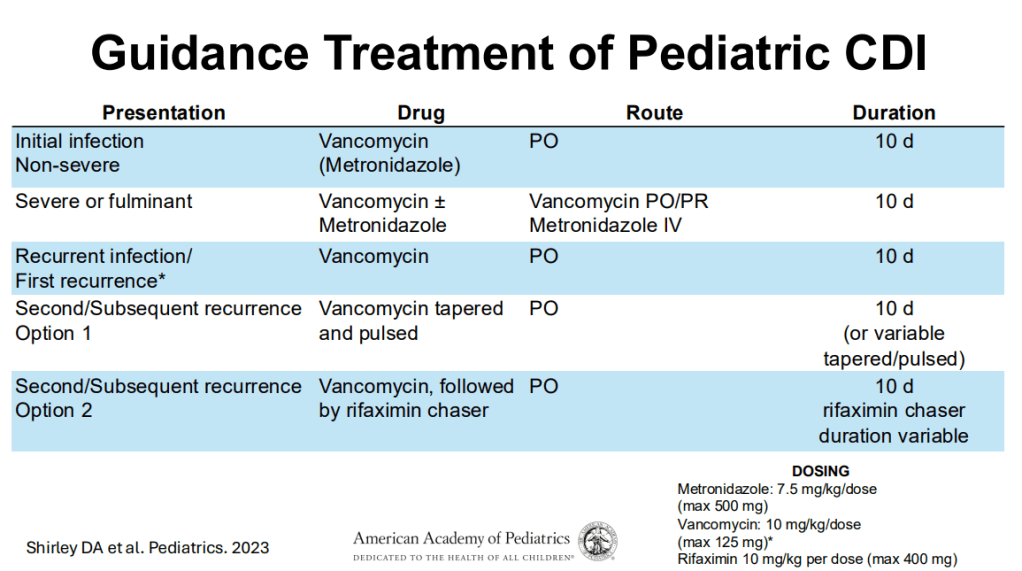
Treatment:
- 1st line treatment remains vancomycin.
- 10-day treatment course is recommended.
- Fidaxomicin, particularly for recurrrent C. diff could be helpful and easier to administer (2/day).
- Prophylactic treatment (low dose vancomycin) may be appropriate in high risk individuals needing to take antibiotics.
- FMT is no longer readily available from stool banks. Donor-directed FMT may be an option after appropriate screening. Given the lack of stool banks, urgent treatment with FMT for severe cases is not available.
- Probiotics have not been proven effective in reducing recurrence and increase the costs for families. A diet high in fruits and veggies (‘eat the rainbow‘) could help restore a more healthy microbiome.
- Newer treatments in adults, Vowst and Rebyota, are expensive and not readily available for children. Anecdotal reports suggest they may be beneficial in pediatric patients.







Conclusions: C diff research is difficult in pediatrics. Many of the patients who need treatment would be excluded from trials. There are very few treatment options in kids.

Related blog posts:
- Clostridioides difficile Treatment in 2026
- Fidaxomicin Treatment of Clostridioides difficile in Children and Adolescents
- C difficile three-fer: Overdiagnosis with Multiplex Testing, Fidaxomicin Pediatric Approval, & Changing Incidence
- Clostridium difficile and Inflammatory Bowel Disease
- NY Times: “Should We Bank Our Own Stool?” | gutsandgrowth
- Expert Advice on Clostridium difficile and Inflammatory Bowel Disease | gutsandgrowth
- 4 Points for C diff in Inflammatory Bowel Disease
- “Diagnostic Stewardship” –Reducing Unnecessary Clostridioides difficile Treatment by Changing Testing Approach (2024)
- OpenBiome Suspending FMT Shipments (2024)
- ACG Clostridium Difficile Guidelines Plus One (2021)

Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.