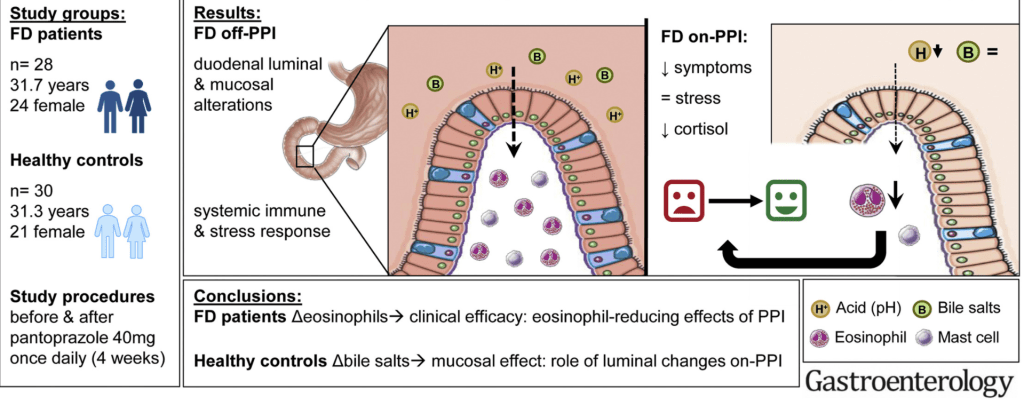
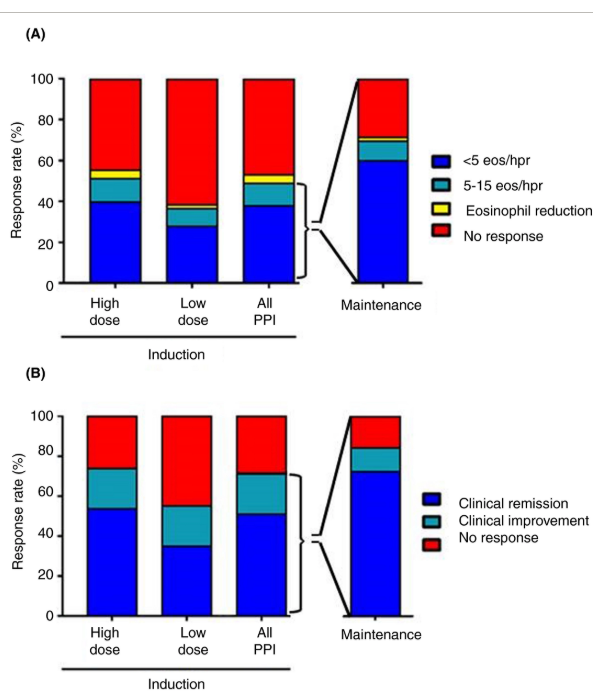
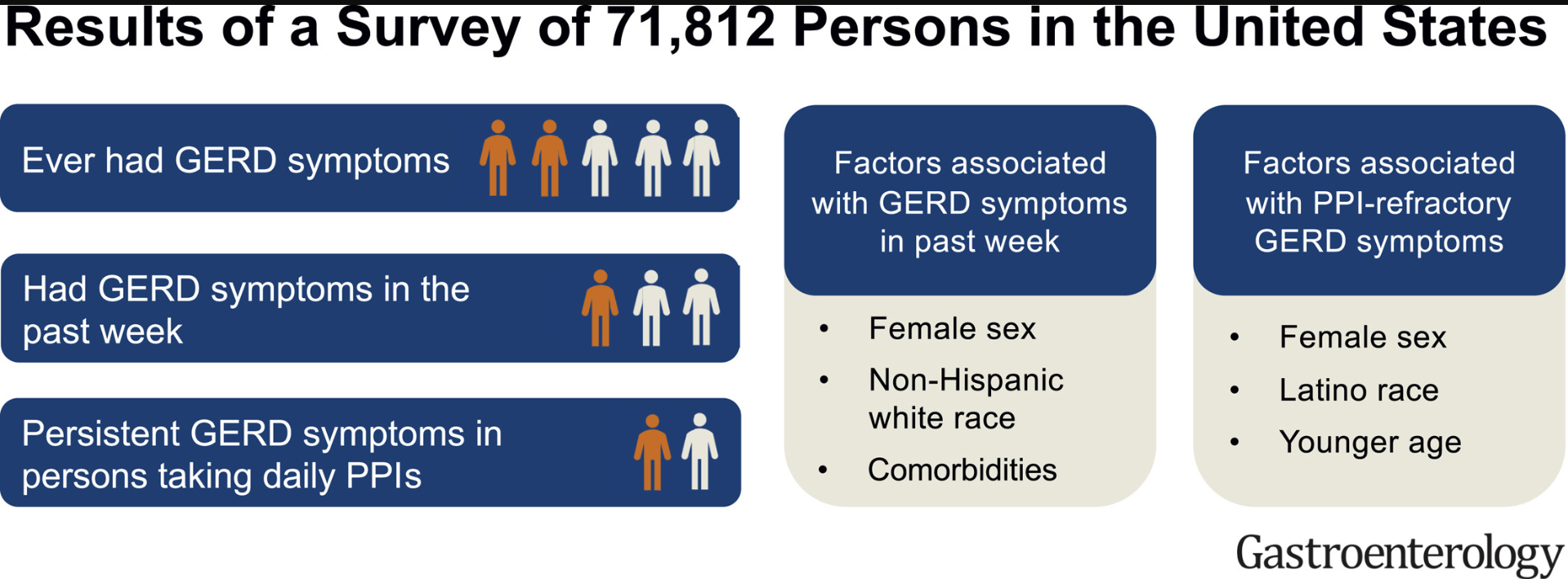
D Lin et atl. Clin Gastroenterol Hepatol 2020; 18: 763-6. In a retrospective chart review, the authors examined pharmacy data from patients in the Harris Health System (Harris county -Houston, TX) which had more than 1.9 million outpatient clinic visits in 2017.
In January 2018, multiple efforts were made to try to reduce inappropriate proton pump inhibitor (PPI) usage. This included grand rounds and system-wide emails to providers. In addition, a suggested tapering algorithm (order in EPIC) was given to reduce the likelihood of rebound acid hypersecretion which could undermine the goal of stopping PPI.
Key points:
- Taper: When ready to taper, start with “a PPI every other day for 2 weeks, followed by a PPI every 4 days for 2 additional weeks before discontinuation.”
- De-escalation: Before educational intervention, in 2017, there were 66,261 unique PPI prescriptions. After educational intervention, in 2018, there were 55,322 unique PPI prescriptions (16.5% decrease). This equates to ~800,000 fewer capsules or pills dispensed in 1 calendar year
- The most “important driver” for de-escalation was the initiation of the discussion by the ambulatory primary care provider
- The authors recommend clinic followup within a month after starting de-escalation and gastroenterology evaluation for patients with severe symptoms or those refractory to PPI treatment
My take: This study indicates that 1 in 6 PPI users were able to de-escalate off treatment. Physician initiative is crucial to improve appropriate medication use.
Related blog posts:

Recent study from JAMA Pediatrics (5/11/20) -Full text: Characteristics and Outcomes of Children With Coronavirus Disease 2019 (COVID-19) Infection Admitted to US and Canadian Pediatric Intensive Care Units
Of the 48 children with COVID-19 admitted to participating PICUs (14 hospitals)… Forty patients (83%) had significant preexisting comorbidities; 35 (73%) presented with respiratory symptoms and 18 (38%) required invasive ventilation….At the completion of the follow-up period, 2 patients (4%) had died and 15 (31%) were still hospitalized, with 3 still requiring ventilatory support and 1 receiving extracorporeal membrane oxygenation. The median (range) PICU and hospital lengths of stay for those who had been discharged were 5 (3-9) days and 7 (4-13) days, respectively.
NY Times Summary of Study: Details of U.S. Children Severely Affected by Coronavirus

Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition