ME Rinella et al. Hepatology 2023; 77: 1797-1835. Open Access! AASLD Practice Guidance on the clinical assessment and management of nonalcoholic fatty liver disease
This 38 page report has a ton of updated recommendations and useful advice –geared to adults with fatty liver disease. The last ~dozen pages are the 491 references.
Some of the useful points:
- CVD and nonhepatic malignancies are the most common causes of mortality in patients with NAFLD without advanced fibrosis; death from liver disease predominates in patients with advanced fibrosis.
- Initial lab evaluation in adults:

- Statins are safe and recommended for CVD risk reduction in patients with NAFLD across the disease spectrum, including compensated cirrhosis.
- Patients with NAFLD should be screened for the presence of T2DM. T2DM is the most impactful risk factor for the development of NAFLD, fibrosis progression, and HCC.108–111 Given the central pathogenic role that insulin resistance plays in the pathogenesis of both T2DM and NAFLD, it is not surprising that patients with T2DM have a higher prevalence of NAFLD (ranging from 30% to 75%)10,112,113 and a higher risk of developing NASH with fibrosis.93,114–117
- Other important comorbidities: dyslipidemia, obstructive sleep apnea, cardiovascular disease, and chronic kidney disease

Lifestyle factors that can be beneficial:

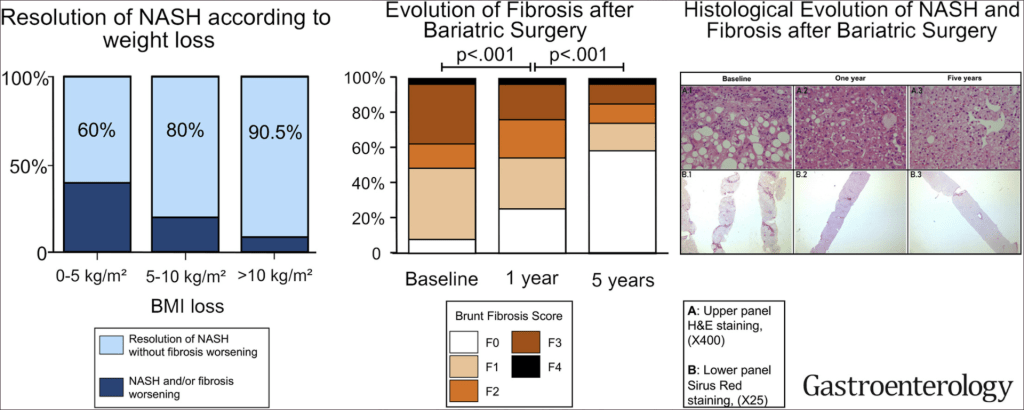
- Table 6 lists potential medications though there are no FDA approved treatments for fatty liver disease. Bariatric surgery is also a beneficial treatment option “in patients who meet criteria for metabolic weight loss surgery, as it effectively resolves NAFLD or NASH in the majority of patients without cirrhosis and reduces mortality from CVD and malignancy.”
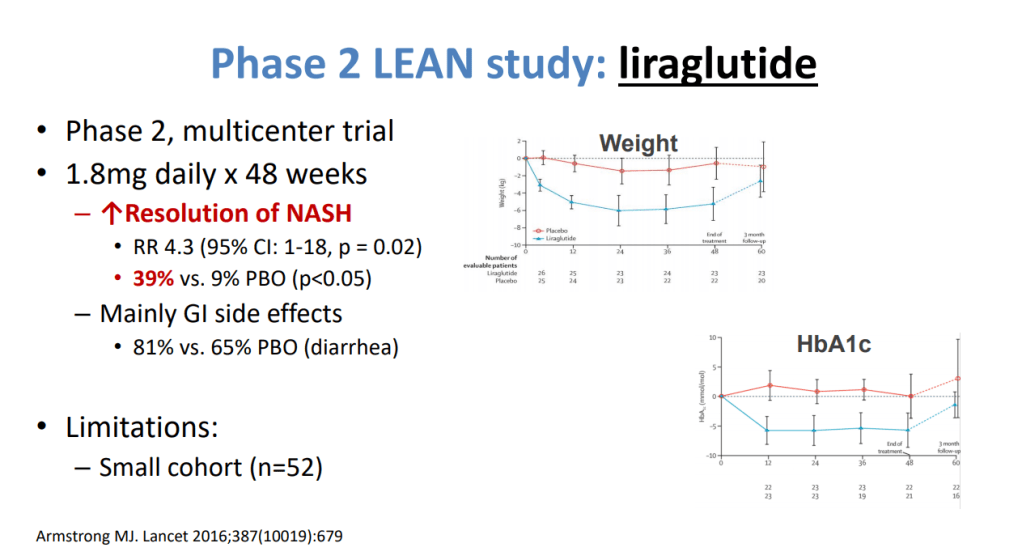
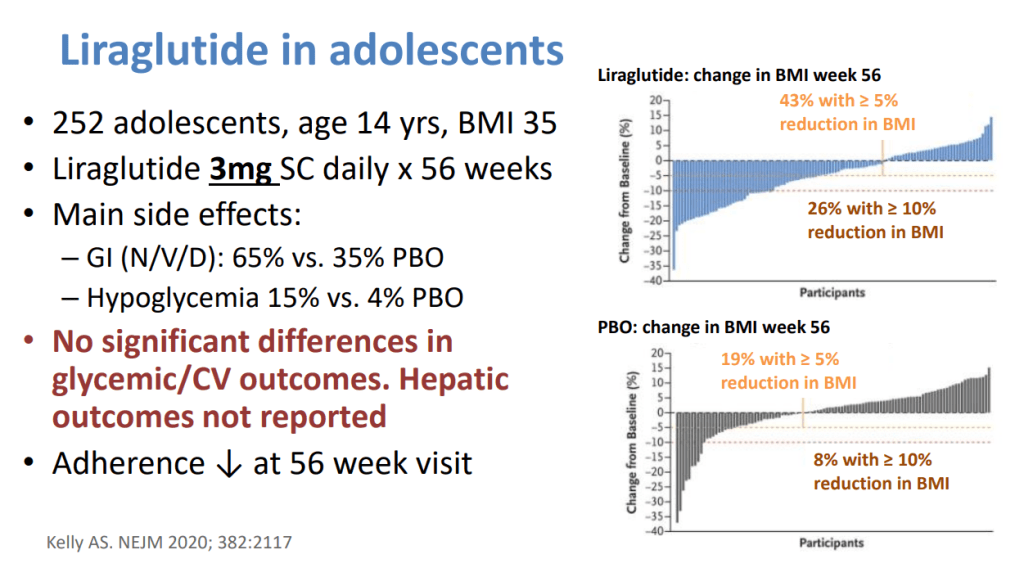
- Potentially useful medications include Vitamin E, Pioglitazone, Liraglutide, Semaglutide, Tirzepatide and SGLT-2i. “Semaglutide can be considered for its approved indications (T2DM/obesity) in patients with NASH, as it confers a cardiovascular benefit and improves NASH. Pioglitazone improves NASH and can be considered for patients with NASH in the context of patients with T2DM . Available data on semaglutide, pioglitazone, and vitamin E do not demonstrate an antifibrotic benefit, and none has been carefully studied in patients with cirrhosis.”
- Treatments NOT Recommended: “Metformin, ursodeoxycholic acid, dipeptidyl peptidase-4, statins, and silymarin are well studied in NASH and should not be used as a treatment for NASH as they do not offer a meaningful histological benefit.”
Related blog posts:
- Aspen Webinar 2021 Part 2 -Nonalcoholic Steatohepatitis
- Pediatric Fatty Liver Disease is a Reversible Disease
- Reversing Pediatric Nonalcoholic Fatty Liver Disease
- Type 2 Diabetes in Children with Nonalcoholic Fatty Liver Disease
- How Much Testing Is Needed In Children with Suspected Fatty Liver?
- What is the likelihood of an alternative liver etiology in children with obesity/overweight?
- Low Free Sugar Diet for Nonalcoholic Fatty Liver Disease in Adolescent Boys
- Statin Use May Help in Fatty Liver Disease
- Autoantibodies Significance in Pediatric Fatty Liver Disease
- NAFLD Guidance from American Association for the Study of Liver Diseases (2018)
- Pediatric NAFLD Guidelines 2017