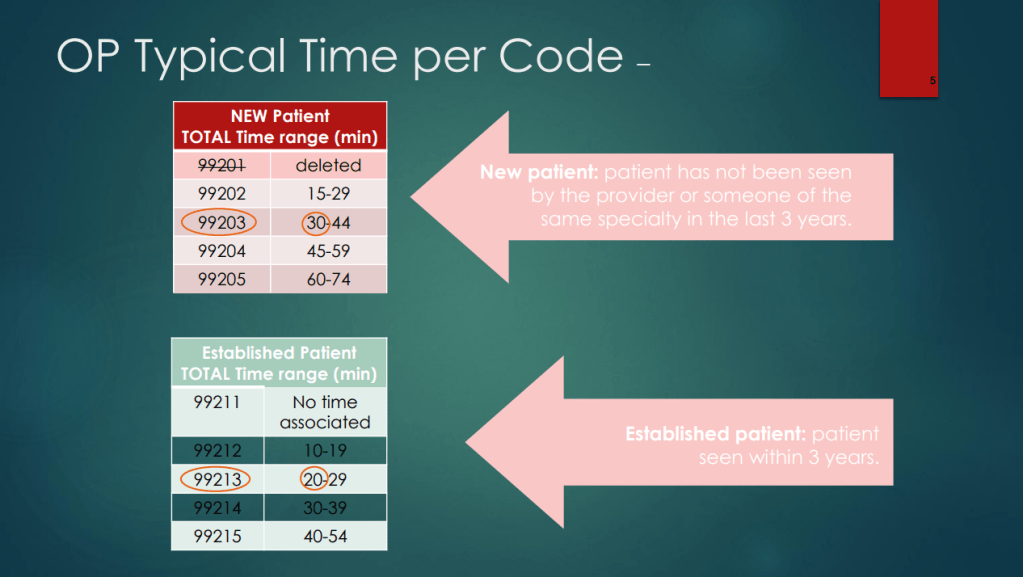
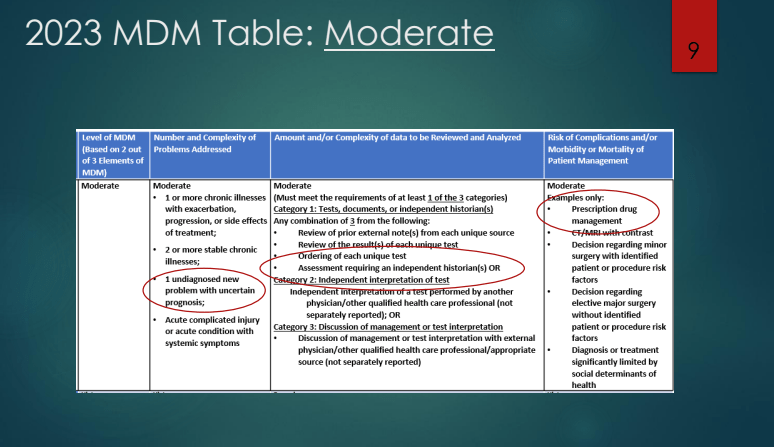
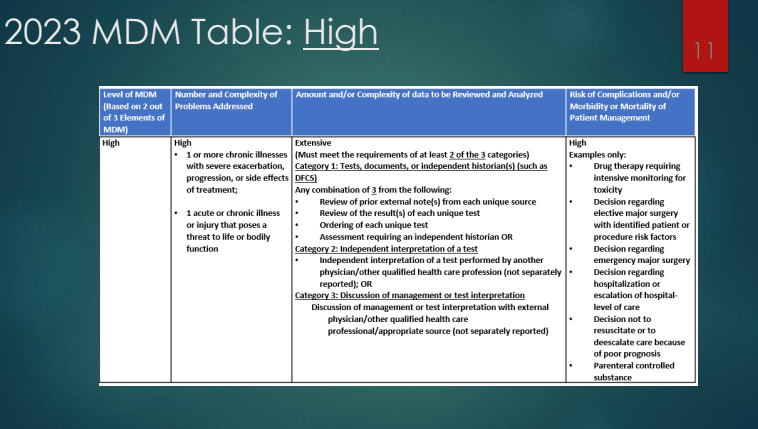
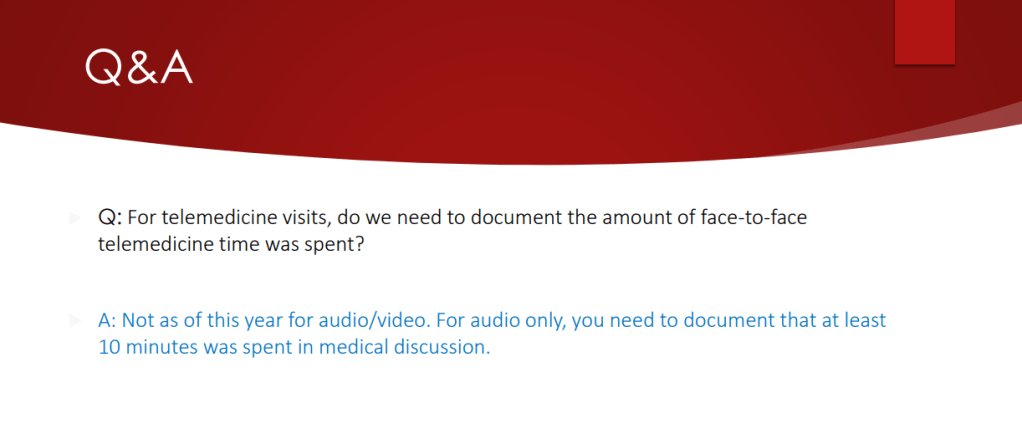
Zack Cooper, NY Times, 5/4/26. This Is the Biggest Culprit for High Health Care Spending.
An excerpt:
Responding to the wrongdoing of insurers is imperative, but it won’t do much to address the unsustainable cost of health care. We are directing our anger at the part of the system that is most visible and frustrating (insurers’ restrictions on care) while ignoring the part of the health system that is most responsible for high costs and economic pain: hospital prices…
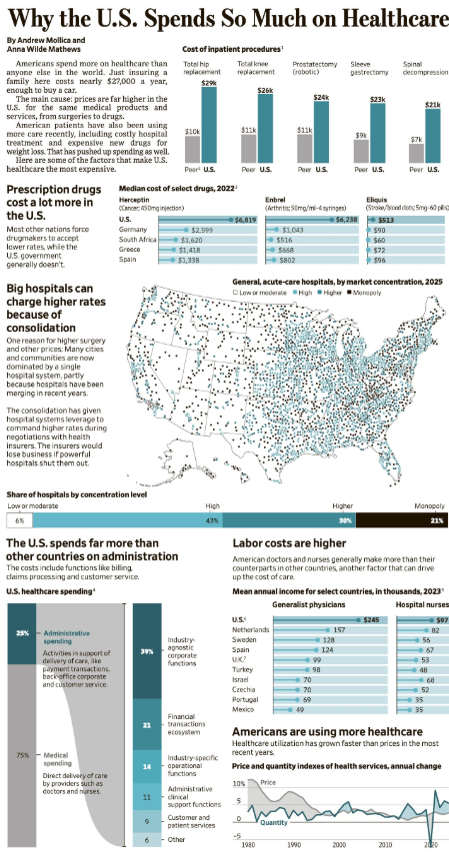
Hospitals in the United States earn $29,000 on average for a replacement covered by private insurance and $16,000 for one covered by Medicare. In Germany, the public system of nonprofit insurers, which covers 90 percent of the population, pays hospitals $9,400.
Hospital prices are the leading driver of the 320 percent increase in insurance premiums that Americans have experienced over the past 25 years. Since 2000, prices at hospitals have grown faster than prices in virtually any other sector of the economy. They have grown three times as fast as inflation and twice as fast as prescription drugs and doctor visits.
The reason hospital prices are so high: hospitals’ accumulation of market power, which brings them more bargaining heft when they negotiate prices with insurers. Since 2000, there have been more than 1,300 hospital mergers among the nation’s approximately 5,000 hospitals. When hospitals that were once competitors merge, prices go up, often by double-digit percentages, with no measurable improvement in patient outcomes. Even though we rely on competition to determine hospital prices, 21 percent of hospitals are effectively monopolies — they have no competitor within a 30-minute drive — and an additional 24 percent face only one competitor…
If hospital prices are such a key driver of rising costs, why aren’t elected officials doing more about them? Partly the answer is politics. Hospitals are the largest or second-largest employer in many counties in America, and a formidable lobbying force — spending more than $100 million annually in Washington, often more than health insurers spend, to protect their interests. Politicians who represent places with dominant hospital systems are not eager to pick a fight with these institutions. Moreover, when an insurer denies your claim, you know it immediately. When a hospital merges and its prices go up, the harms — slower overall economic growth and job losses outside the hospital sector — are real but diffuse…
Holding that tension between the immense good that hospitals do and the economic harm their market power creates is what it will take to address the rising cost of health care.
In reaction to this editorial, the American Hospital Association Blog responded on May 6, 2026: Setting the Record Straight: Three Ways the Hospital‑blame Narrative Gets it Wrong
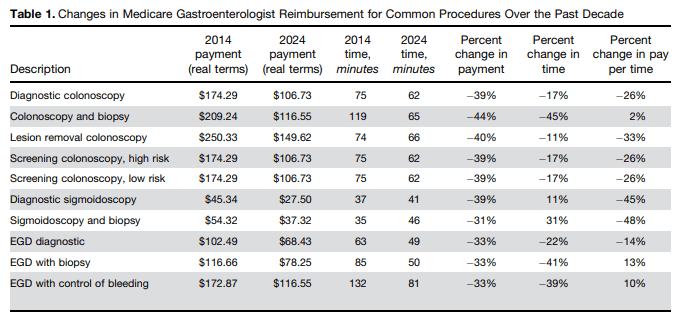
“Let’s start with a basic point — the entire concept of hospital “prices” is flawed. Hospitals are largely price takers, not price setters. Government programs like Medicare and Medicaid set rates administratively. But Medicare reimbursement continues to lag behind inflation — covering just 83 cents for every dollar spent by hospitals in 2023 — resulting in over $100 billion in underpayments. Needless to say, government rates fall far short of covering hospitals’ actual costs. Likewise, commercial insurers aggressively negotiate payment terms with hospitals. In many cases, these are large, vertically-integrated companies that hold significant shares in their markets. With that kind of market power, commercial insurers do not simply accept the numbers that hospitals offer and then sign on the dotted line. Thus, the notion of hospital “prices” does not reflect how those purposed “prices” are set in the real world…But here’s the reality: Over the same time frame, commercial insurance premiums have increased more than hospital prices…premiums are primarily growing due to an increased need for services: People are sicker and they need more care.”
“The essay also … appears to excuse commercial insurers’ role in deciding to raise their own premiums because they are in the “business of making money…The essay argues that policymakers must be harder on hospitals.”
“The essay also fails to ask another important question: Why are hospitals forced to raise their “prices”?…In 2025, total hospital expenses grew 7.5%, more than twice the rate of growth in hospital prices. Costs increased in every major category — workforce, drugs, medical supplies, and more. Hospital expenses also increased because of increased patient complexity; growing uncompensated care; continued government underfunding; changes in the policy landscape; and the many commercial insurer tactics like prior authorization and improper denials that the essay observes.”
My take: There are lots of reasons for high healthcare costs. There are not simple fixes nor enough incentives to make changes.
Related blog posts:
- When Hospitals Look Like The Ritz (But Cost Even More)
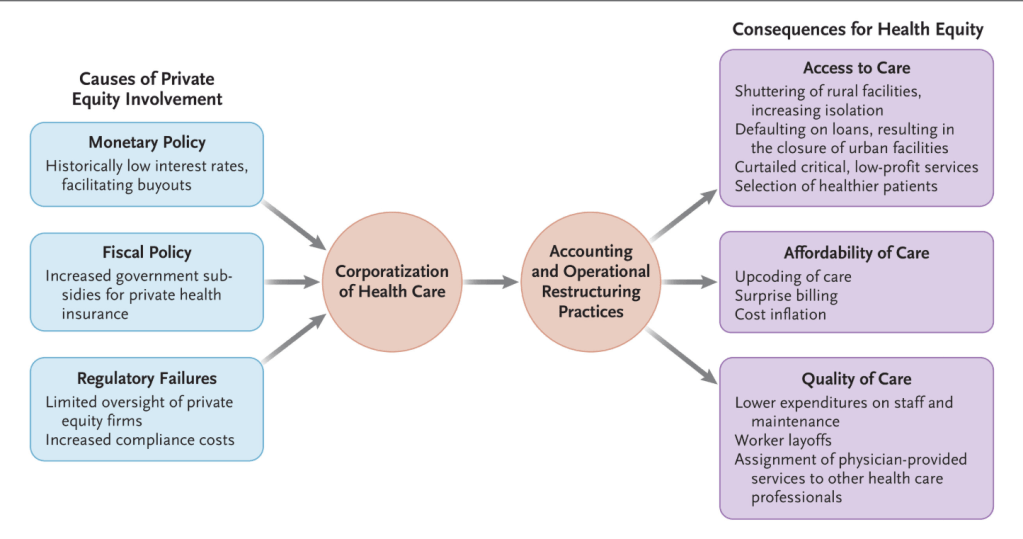
- Why Corporatization Occurs in Health Care -What Motivates Hospitals
- Understanding the Business of Medicine and Downstream Consequences
- Medical Billing Trap: Hospital Pricing for Urgent Care Visits and Outpatient Departments
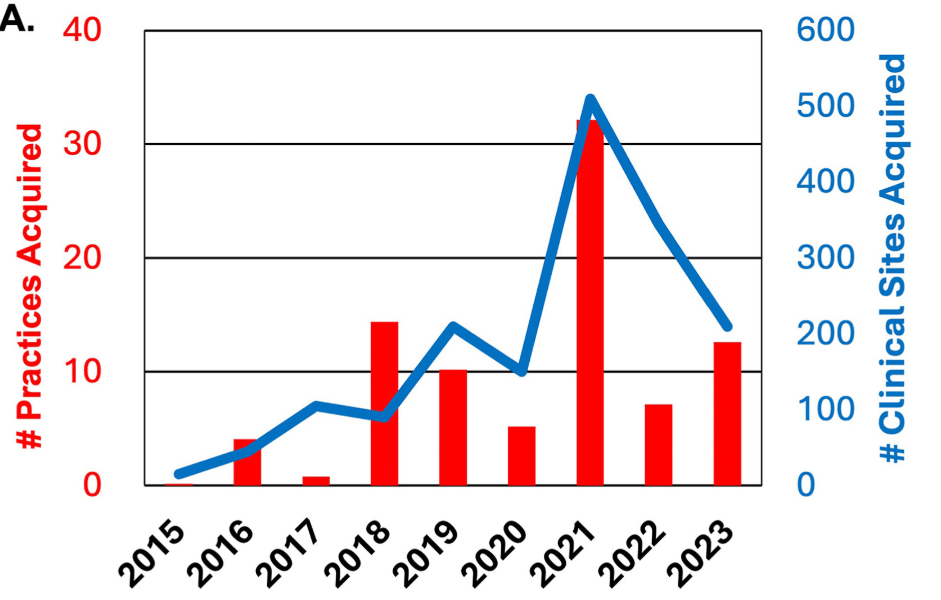
- What Is Driving Hospitals’ Acqui$ition of Physician Practices?
- Healthcare: “Where the Frauds Are Legal”
- Dr. John Barnard: Trends in Pediatric Workforce — A Growing Concern One of the slides shows a 3000% growth in the number of adminstrators/managers in health care from 1970 to 2015.