From AGA Today (8/5/25): “Sterile Water is Unnecessary for Endoscopy”
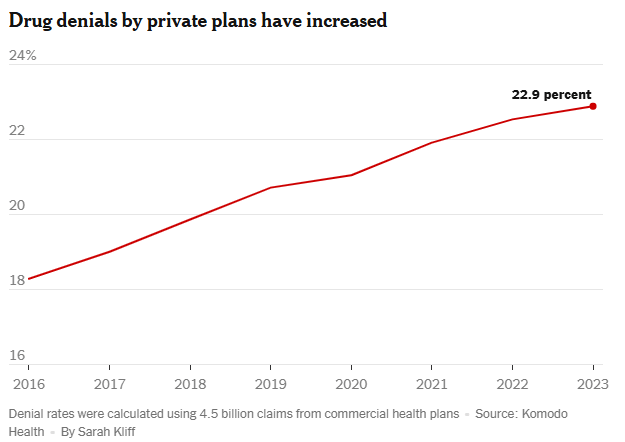
GI and Hepatology News (8/4, Pass) reports a review suggests that “endoscopists can safely forgo sterile water in favor of tap, reducing both environmental and financial costs.” Researchers found that only two studies since 1975 “directly compared sterile and tap water use in endoscopy,” and “neither showed an increased risk of infection from tap water. In fact, some cultures from allegedly sterile water bottles grew pathogenic bacteria, while no patient complications were reported in either study.” Current guidelines “recommend sterile water for procedures involving mucosal penetration but acknowledge low-quality supporting evidence.” However, they pointed out that “these recommendations are based on outdated studies, some unrelated to GI endoscopy.” Furthermore, the “review estimates that the production and transportation of sterile water bottles contributes over 6,000 metric tons of emissions per year from US endoscopy units alone.” The review was published in Gastro Hep Advances.
Referenced article: D Agrawal, et al. Gastro Hep Advances, Volume 4, Issue 5, 100625. Ripple Effect: Safety, Cost, and Environmental Concerns of Using Sterile Water in Endoscopy
Environmental Costs:
“With a conservative estimate of using half of a 1-L sterile bottle for irrigation per endoscopy, 22 million yearly endoscopies in the US could result in an additional 6000 tons of eCO2.”
Economic Costs:
“A 1-L bottle of sterile water costs $3–$10. For an endoscopy unit performing 30 procedures daily and a conservative estimate of half a water bottle per case, the average monthly direct costs could be $1000–$3000”
Discussion:
“There is no direct supporting evidence for using sterile water during endoscopy…a Cochrane review show no difference in infection risk when using tap or sterile water to irrigate wounds…Similarly, there is no benefit in using sterile water for enteral feeds in immunosuppressed patients, and tap water enemas are routinely acceptable for colon cleansing before sigmoidoscopies in all patients, irrespective of immune status.
My take: Plastic water bottles in endoscopy centers contribute to health-care waste, climate change and increased costs.
Related blog post: Environmental Impact of Endoscopy