M Zvuloni et al. JPGN 2021; 73: 717-721. Open Access PDF: High anti-TNFa Concentrations Are Not Associated With More Adverse Events in Pediatric Inflammatory
Bowel Disease
Key findings (retrospective study):
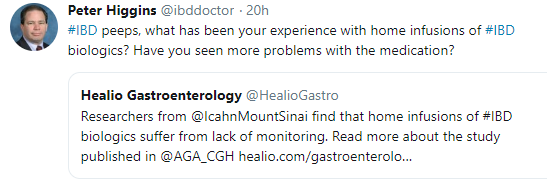
- Higher trough concentrations (TCs) (>10 mcg/mL) of anti-TNFa were not associated with higher rate of anti-TNFa-related adverse events in 135 patients & >1500 TC measurements
- Out of the 30 patients who presented with elevated transaminases, 27 (90%) patients had normalized transaminases values by the end of the follow-up
- Adverse events were noted in 68 of 135 patients (see below)
OH Nielsen et al. Clin Gastroenterol Hepatol 2022; 20: 74-87. Open Access: Biologics for Inflammatory Bowel Disease and Their Safety in Pregnancy: A Systematic Review and Meta-analysis
Forty-eight studies were included in the meta-analysis comprising 6963 patients. Key findings:
- Biologic therapy in IBD pregnancies was associated with a pooled prevalence of 8% for early pregnancy loss, 9% for preterm birth, 0% for stillbirth, 8% for low birth weight, and 1% for congenital malformations.
- These rates are comparable with those published in the general population.
- Importantly, studies with newer biologics (eg. vedolizumab, ustekinumab) had small sample sizes. In addition, ongoing prospective multicenter registries are ongoing.
EA Spencer et al. JPGN 2021; 73: 710-716. Open Access PDF: Outcomes of Primary Ileocolic Resection for Pediatric Crohn Disease in the Biologic Era
Key findings (n=78, retrospective study, 2/3rds received biologic postoperative prophylactic therapy):
- Endoscopic recurrence was 46% at 2 years (median time to recurrence: 10 months).
- Histologic recurrence was present in 44% in endoscopic remission
- At diagnosis and surgery, over a quarter met the criteria for growth failure.. Following surgery, height, weight and BMI z scores improved significantly both at 1 year and last followup