RJ Colman et al. Crohn’s & Colitis 360, Volume 7, Issue 3, July 2025, otaf050. https://doi.org/10.1093/crocol/otaf050. Open Access! Therapeutic Drug Monitoring in Pediatric Inflammatory Bowel Disease: A Nationwide Survey of Anti-TNF Therapy Practices, Attitudes, and Barriers
Methods: A 28-item survey of therapeutic drug monitoring (TDM) was deployed through the ImproveCareNow Learning Health System Network between February and June 2023.
Key findings:
- Among 380 invitees, 256 (77%) completed the questionnaire. Among respondents, 67% (171) were academic-affiliates
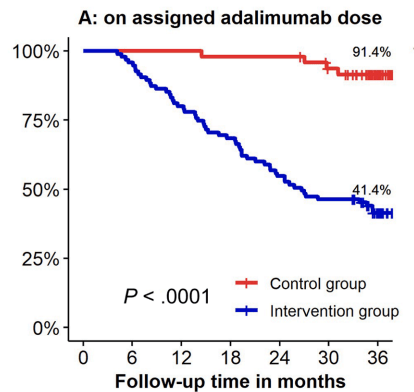
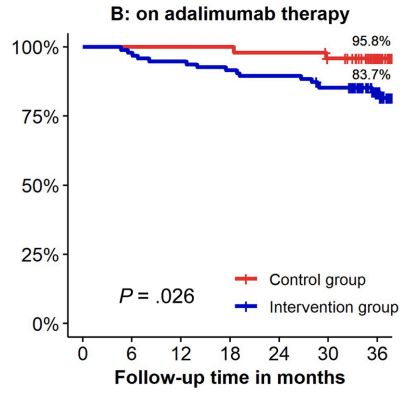
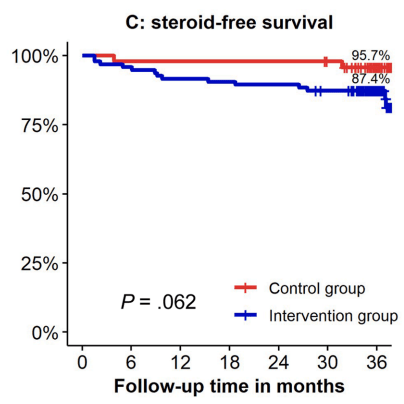
- In contrast to the adult literature, most pediatric gastroenterologists report undertaking proactive TDM for anti-TNF agents in IBD management
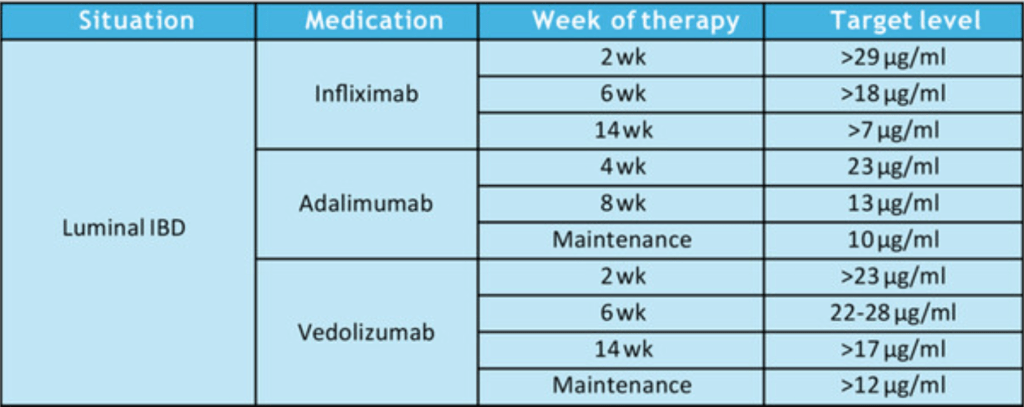
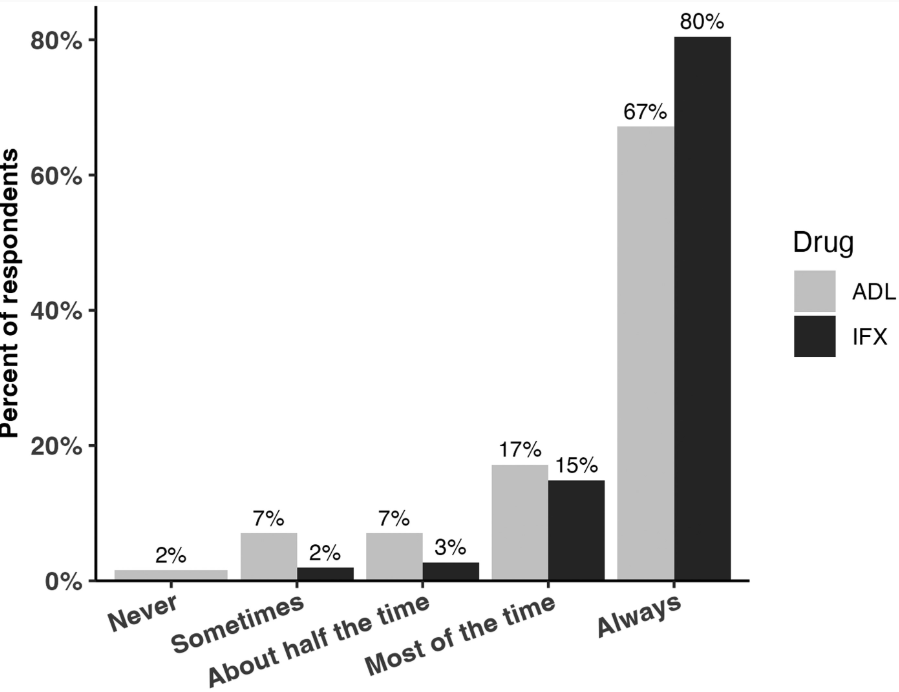
- TDM: All 256 respondents reported using TDM for infliximab, while 252 (98%) used TDM for adalimumab (ADL)
- Proactive TDM: Proactive TDM was more common for infliximab 98% (205/256) compared to ADL 92% (232/252; P = .0007)
- Overall, 61% (156/255) of respondents reported that they experienced barriers in undertaking TDM. Insurance denial (50%) and cost (31%) were the 2 most commonly reported barriers
Limitation: Only surveyed practitioners in ICN. However, “approximately 65% of US pediatric gastroenterologists practice within ICN-affiliated institutions, [thus] our findings likely reflect national trends.”
My take (borrowed from authors): “Proactive TDM, is a widely practiced strategy, and represents the current standard of care among pediatric gastroenterologists in the United States.”
How to explain a complication with Will Ferrell, YouTube Link: SNL Post-Op (5 minutes)
Related blog posts:
- NASPGHAN Pediatric Position Paper for Therapeutic Drug Monitoring Disease (2024)
- Proactive Drug Monitoring for Crohn’s Disease in Pediatrics
- Proactive Therapeutic Drug Monitoring and Better Outcomes in Pediatric Crohn’s
- Why Pediatric Patients Need Higher Dosing of Infliximab
- Another Study Justifying Higher Infliximab Dosing in Pediatrics
- Proactive Therapeutic Drug Monitoring in Pediatric Crohn’s disease -Better Outcomes (2020)
- Here’s The Proof That Proactive Drug Monitoring Improves Outcomes in Children With Crohn’s Disease (2019).
- Can Therapeutic Drug Monitoring with Monotherapy Achieve Similar Results as Combination Therapy for IBD? (2019)