In a previous post, this blog highlighted SERENE-CD which showed that higher induction doses of adalimumab did not improve outcomes compared to standard dosing (SERENE Study: Does a Higher Induction Dose of Adalimumab Help for Crohn’s Disease?)
However, there was a 2nd SERENE study: SERENE-UC: J Panes et al. Gastroenterol 2022; 162: 1891-1910. Open Access: Higher vs Standard Adalimumab Induction and Maintenance Dosing Regimens for Treatment of Ulcerative Colitis: SERENE UC Trial Results The online version includes supplementary material (link: supplement) which is needed to understand the response rate more fully.
The main component of this double-blind, randomized (no placebo) study allocated 512 patients with ulcerative colitis to a higher induction regimen (HIR) of adalimumab and 340 patients to a standard induction regimen (SIR). A maintenance phase continued with 374 main patients who were clinical responders at week 8 (n=757 who completed induction). The study results are presented in a confusing manner, in part because of a subgroup from Japan as well as a great deal of data from both the induction phase and the maintenance phase.
Key findings:
- In the main study, 13.3% vs 10.9% of patients receiving the higher induction regimen (HIR) vs standard induction regimen (SIR) achieved clinical remission (full Mayo score ≤2 with no subscore >1) at week 8 (induction primary end point; P = .265)
- Among week-8 responders, 39.5% vs 29.0% receiving 40 mg ew vs 40 mg eow achieved clinical remission at week 52 (maintenance primary end point; P = .069).
- Figure S2 below shows that approximately 50% of patients treated with adalimumab had a clinical response at week 8
My takes on this study:
- Fairly low response to adalimumab: the clinical remission rate for adalimumab is low at week 8 (10-13%) and the 8-week response rate is less than 50%
- Higher doses during induction were not helpful & did not result in significantly better responses at week 8
- Therapeutic drug monitoring was not beneficial in this study
- Higher doses during maintenance were associated with improved responses: patients receiving weekly adalimumab during maintenance treatment had improved week 52 remission. The editorial (pages 1831-1832) note that this effect was demonstrated in those with “elevated C-reactive protein, low albumin, extensive UC or long disease burden”
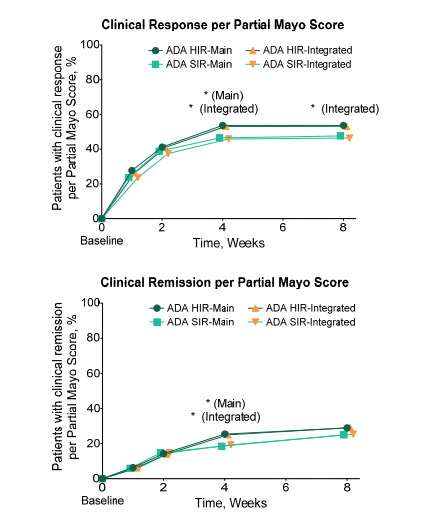
decrease from baseline in rectal bleeding subscore or absolute rectal bleeding subscore of 0 or 1.
Clinical remission was defined as Partial Mayo Score ≤ 2 with no subscore > 1.
ADA, adalimumab; HIR, higher induction regimen; SD, standard deviation; SIR, standard induction regimen
Related blog posts:
- Ustekinumab for Refractory Pediatric Ulcerative Colitis and Updated Adalimumab Dosing
- SERENE-CD Study: Does a Higher Induction Dose of Adalimumab Help for Crohn’s Disease?
- AGA Guidelines: Moderate to Severe Ulcerative Colitis
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.
