Bowel Sounds Link: Joel Rosh talks small molecules and biologics
This is another good chat. Dr. Rosh provides a lot of information about the newest IBD agents. Overall, the episode indicates a very enthusiastic experience with IL-23 targeting agents like risankizumab (perhaps the ‘Michael Jordan’ of biologics) and with JAK agents like tofacitinib and upadacitinib. Dr. Rosh’s experience with regard to safety of these newer agents has been very positive. For tofacitinib, the typical dosing alluded to in the podcast was 10 mg twice a day (not three times a day). The potential adverse effects, though unlikely in the pediatric population, are carefully discussed with families and monitored.
So far, Dr. Rosh has not found a niche for ozanimod. In addition, he briefly discusses therapeutic drug monitoring. With regard to using vedolizumab as a first-line agent for ulcerative colitis, he often uses the VARSITY study (BE Sands et al NEJM 2019; 381: 1215-26) to justify this to payers. There is a sad element to the podcast though –Dr. Rosh admits to being a lifelong Mets fan!
Related blog posts:
- Vedolizumab versus Adalimumab for Ulcerative Colitis (part 2)
- Vedolizumab More Effective Than Adalimumab for Ulcerative Colitis
- Vedolizumab vs Adalimumab: Histology Outcomes from Varsity Trial
- Risankizumab Receives FDA Approval for Crohn’s Disease
- Upadacitinib Receives FDA Approval to Treat Adults with Ulcerative Colitis

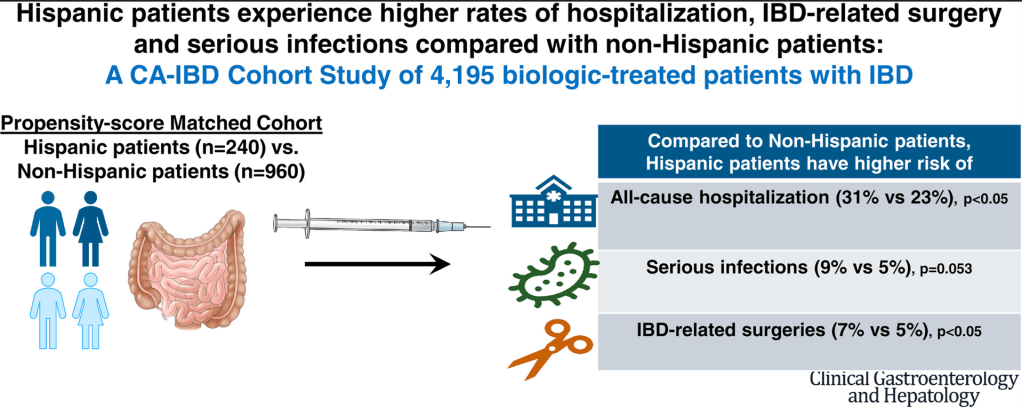
NH Nguyen et al. Clin Gastroenterol Hepatol 2023; 21: 173-181. Open Access! Effectiveness and Safety of Biologic Therapy in Hispanic Vs Non-Hispanic Patients With Inflammatory Bowel Diseases: A CA-IBD Cohort Study
Key findings in this retrospective study with 240 Hispanic patients:
- Within 1 year of biologic initiation, Hispanic patients had higher rates of hospitalizations (31% vs 23%; adjusted hazard ratio [aHR], 1.32; 95% CI, 1.01–1.74) and IBD-related surgery (7.1% vs 4.6%; aHR, 2.00; 95% CI, 1.07–3.72), with a trend toward higher risk of serious infections (8.8% vs 4.9%; aHR, 1.74; 95% CI, 0.99–3.05).
- The authors state “these findings suggest that biologic agents may not be as effective or safe in Hispanic patients as they are in non-Hispanic Caucasians… Besides biological factors, socioeconomic factors related to costs and access to care, which contribute to delayed initiation of biologics, and/or limited postinitiation monitoring, leading to higher rates of unplanned health care utilization.”
T Kucharzik et al. Clin Gastroenterol Hepatol 2023; 21: 153-163. Open Access! Early Ultrasound Response and Progressive Transmural Remission After Treatment With Ustekinumab in Crohn’s Disease (STARDUST study)
Key findings:
- IUS showed that ustekinumab-treated CD patients achieved progressive IUS response (46.3%) and transmural remission (24.1%) through week 48, with a more robust response in the colon and biologic-naive patients
- Fair/moderate reliability (κ = 0.21–0.51) was observed between week 4 IUS response and week 48 overall endoscopic response and fecal calprotectin/complete biomarker outcomes.
