T Straamijer et al. Clin Gastroenterol Hepatol 2023; 21: 182-191. Open Access! Superior Effectiveness of Tofacitinib Compared to Vedolizumab in Anti-TNF-experienced Ulcerative Colitis Patients: A Nationwide Dutch Registry Study
Methods: Adults with ulcerative colitis (UC) previously who failed anti-TNF treatment and initiated vedolizumab (n=83) or tofacitinib (n=65) treatment were identified in the Initiative on Crohn and Colitis Registry in the Netherlands.
Key findings (Vedolizumab is in gray):
- There was no difference in infection rate or severe adverse events.
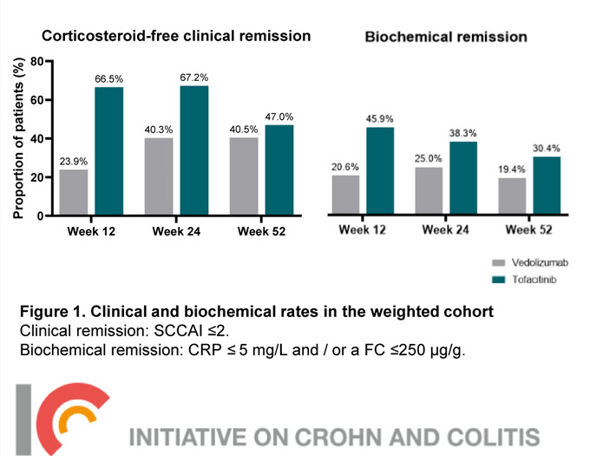

My take: Coupled with more recent reassuring safety data on JAK inhibitors, this study makes a strong case for positioning Tofacitinib (or other JAK inhibitor) earlier in patients with moderate-to-severe ulcerative colitis. Given that vedolizumab outperformed adalimumab in a head-to-head study, this indicates that tofacitinib is a very effective therapy.
Related article: B Chen et al. Gastroenterology 2022; 163: 1555-1568. Efficacy and Safety of Ivarmacitinib in Patients With Moderate-to-Severe, Active, Ulcerative Colitis: A Phase II Study This phase 2 study with 146 patients examined the effectiveness of the selective JAK inhibitor Ivarmacitinib found a week 8 clinical response in 46% of those receiving 8 mg per day. The week 8 clinical remission rate was 22%-24% in the treatment groups compared to 5% in the placebo group.
Related blog posts:
- Treatments for “Bad” Inflammatory Bowel Disease (Part 2) & Reassuring Data on Tofacitinib
- Vedolizumab vs Adalimumab: Histology Outcomes from Varsity Trial
- Vedolizumab versus Adalimumab for Ulcerative Colitis (part 2)
- Vedolizumab More Effective Than Adalimumab for Ulcerative Colitis
- IBD Updates: Probability of Needing a Stoma with Crohn’s Disease, “CEASE” anti-TNF study, Extending Tofacitinib Response Time
- Increased Risk, Increased Reward (possibly) with Tofacitinib
- A New FDA Warning for Tofacitinib
- “Tofacitinib: A Jak of All Trades”
- How Quickly Does Tofacitinib Work for Ulcerative Colitis?
- FDA Slaps Restrictions on JAK Inhibitors Over Serious Safety Risks (2021)
- A New FDA Warning for Tofacitinib (2021)
- AGA Guidelines: Moderate to Severe Ulcerative Colitis
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.
