E Vermeer et al. J Pediatr Gastroenterol Nutr. 2026;82:477–486. Open Access! Methotrexate toxicity and intolerance in paediatric inflammatory bowel disease: A retrospective cohort study
This was a a retrospective single-center cohort study, including pediatric IBD patients (n=207) initiating MTX between 2010 and 2023. The median follow-up time was 303 days.
Key findings:
- Methotrexate was used in combination with a biologic medication in 114 patients (55%)
- 157 patients (75.8%) experienced at least one MTX-induced AE, with hepatotoxicity occurring in 84 patients (53.5%), myelotoxicity in 43 patients (27.4%), and nausea in 95 patients (60.5%). Most hepato- and myelotoxicity cases were categorized as grade 1 or mild (60.7% and 81.4%, respectively). 10 patients had grade 3 hepatotoxicity (ALT 195-780 U/L)
- Nausea was reported in 46%. Fatigue was identified in 13, Headache in 6, and Alopecia in 6
- MTX was discontinued in 60 out of 157 cases with an AE (38%), including 27 following nausea, 27 and 4 following hepatotoxicity
- Sixty-five (43.0%) of all biochemical toxicities occurred within the first 3 months of MTX initiation
- Strategies to manage AEs included reduced dosage, use of antiemetics or PPIs, and change in route of administration
Discussion:
- The authors recommend biochemical testing after initiation “at 2, 4, 8, and 12 weeks, as most actionable toxicities occurred during this period. After 3 months, laboratory assessments could potentially be spaced out to every 4–6 months instead of every 3 months for stable patients, aligning with the new Dutch guideline for monitoring MTX toxicity in rheumatology.41 More frequent testing should be reserved for patients with risk factors such as renal dysfunction, hepatotoxic co-medications, or prior toxicity.42“
- A study limitation was “the frequent use of combination therapy, leading to a heterogeneous study population and possible overestimation of AE rates”
My take: Methotrexate remains an important part of treatment, especially combination treatment to prevent or overcome immunogenicity. Careful monitoring and prophylactic treatments of nausea may improve durability.
Related blog posts:
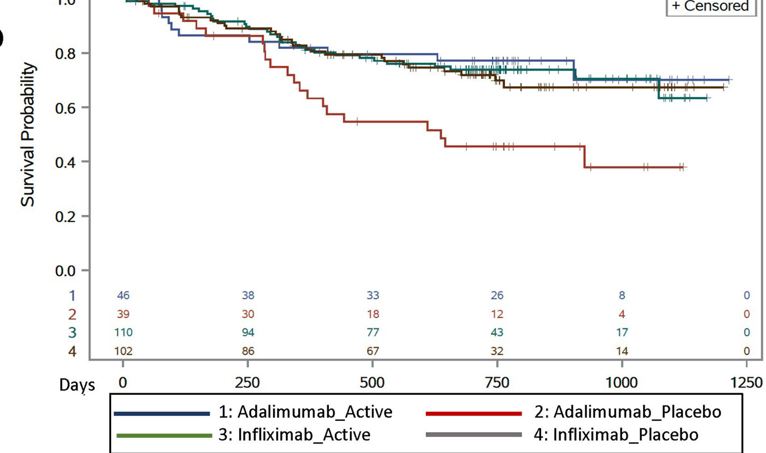
- Why Do Children Taking Adalimumab Benefit from Methotrexate Dual Therapy?
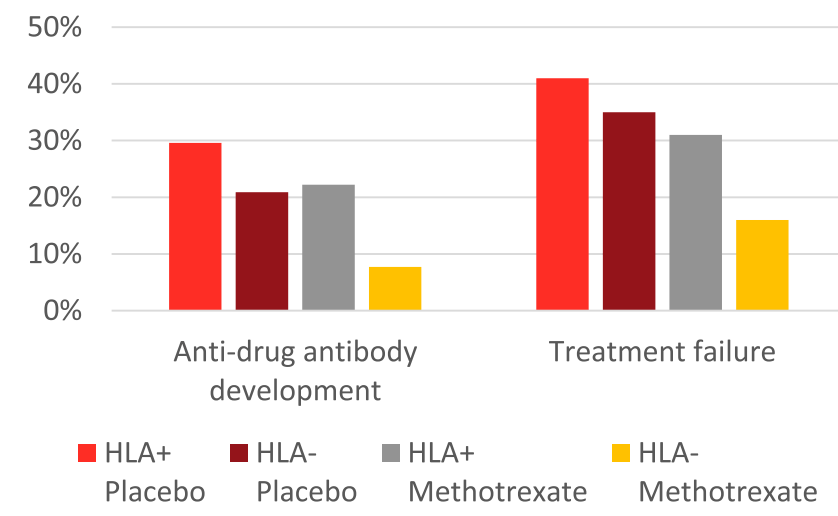
- How HLA DQA1*05 and Combination Therapy Modulate Treatment Outcomes in Children with Crohn’s Disease
- Combination Therapy Associated with Treatment Persistence
- Methotrexate -Not Effective as Monotherapy for Ulcerative Colitis
- Methotrexate Dosing in Dual Therapy
- Can Therapeutic Drug Monitoring with Monotherapy Achieve Similar Results as Combination Therapy for IBD?
- Combination Therapy Study Points to Central Role of Adequate Drug Levels
- COMBO-IBD Study -Combination Immunomodulator Use and Thresholds