A Yerushalmy-Feler et al. Inflammatory Bowel Diseases, Volume 30, Issue 2, February 2024, Pages 159–166. Open Access! Dual Biologic or Small Molecule Therapy in Refractory Pediatric Inflammatory Bowel Disease (DOUBLE-PIBD): A Multicenter Study from the Pediatric IBD Porto Group of ESPGHAN
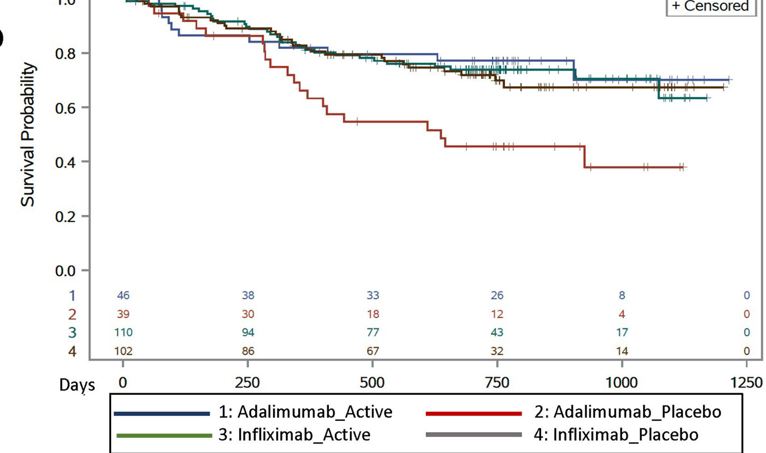
In this retrospective study with 62 children (35 Crohn’s disease, 27 ulcerative colitis) with extensive and severe IBD that was refractory to various therapies, the authors examined the outcomes of combination therapies: the dual therapy included an anti-tumor necrosis factor agent and vedolizumab in 30 children (48%), anti-tumor necrosis factor and ustekinumab in 21 (34%) children, vedolizumab and ustekinumab in 8 (13%) children, and tofacitinib with a biologic in 3 (5%) children.
Key findings:
- Clinical remission was observed in 21 (35%), 30 (50%), and 38 (63%) children at 3, 6, and 12 months, respectively.
- Normalization of C-reactive protein and decrease in fecal calprotectin to <250 µg/g were achieved in 75% and 64%, respectively, at 12 months
- Twenty-nine (47%) children sustained adverse events, 8 of which were regarded as serious and led to discontinuation of therapy in 6.
- Among the 43% that were receiving steroids at the start of dual therapy, twenty (74%) of them could be successfully weaned within 3 months after the initiation of dual therapy.
- Only 2 of 23 (8.7%) had endoscopic healing
My take (borrowed partly from authors):
- “Dual biologic therapy may be effective in children with refractory IBD. The potential efficacy should be weighed against the risk of serious adverse events” and affordability.
- “There are currently no data for identifying the patients that are more likely to benefit from dual therapy….The ideal selection of dual biologic regimens remains to be determined.”
A Al-Janabi et al. JAMA Dermatol. 2024;160(1):71-79. doi:10.1001/jamadermatol.2023.4846 Open Access! Risk of Paradoxical Eczema in Patients Receiving Biologics for Psoriasis
This study examined more than 13,000 patients enrolled in a prospective cohort study from the British Association of Dermatologists Biologics and Immunomodulators Register for adults treated with biologics for plaque psoriasis.
Key findings:
- A total of 273 exposures (1%) were associated with paradoxical eczema.
- The adjusted incidence rates were 0.94 per 100 000 person-years for TNF inhibitors, 0.80 per 100 000 person-years for IL-12/23 inhibitors, and 0.56 per 100 000 person-years for IL-23 inhibitors. IL-23 inhibitors were associated with a lower risk of paradoxical eczema (hazard ratio [HR], 0.39)
My take (from authors): The overall incidence of paradoxical eczema was low in biologic-treated patients with psoriasis. The risk was lowest in patients receiving IL-23 inhibitors. Increasing age, female sex, and history of AD or hay fever were associated with higher risk of paradoxical eczema.