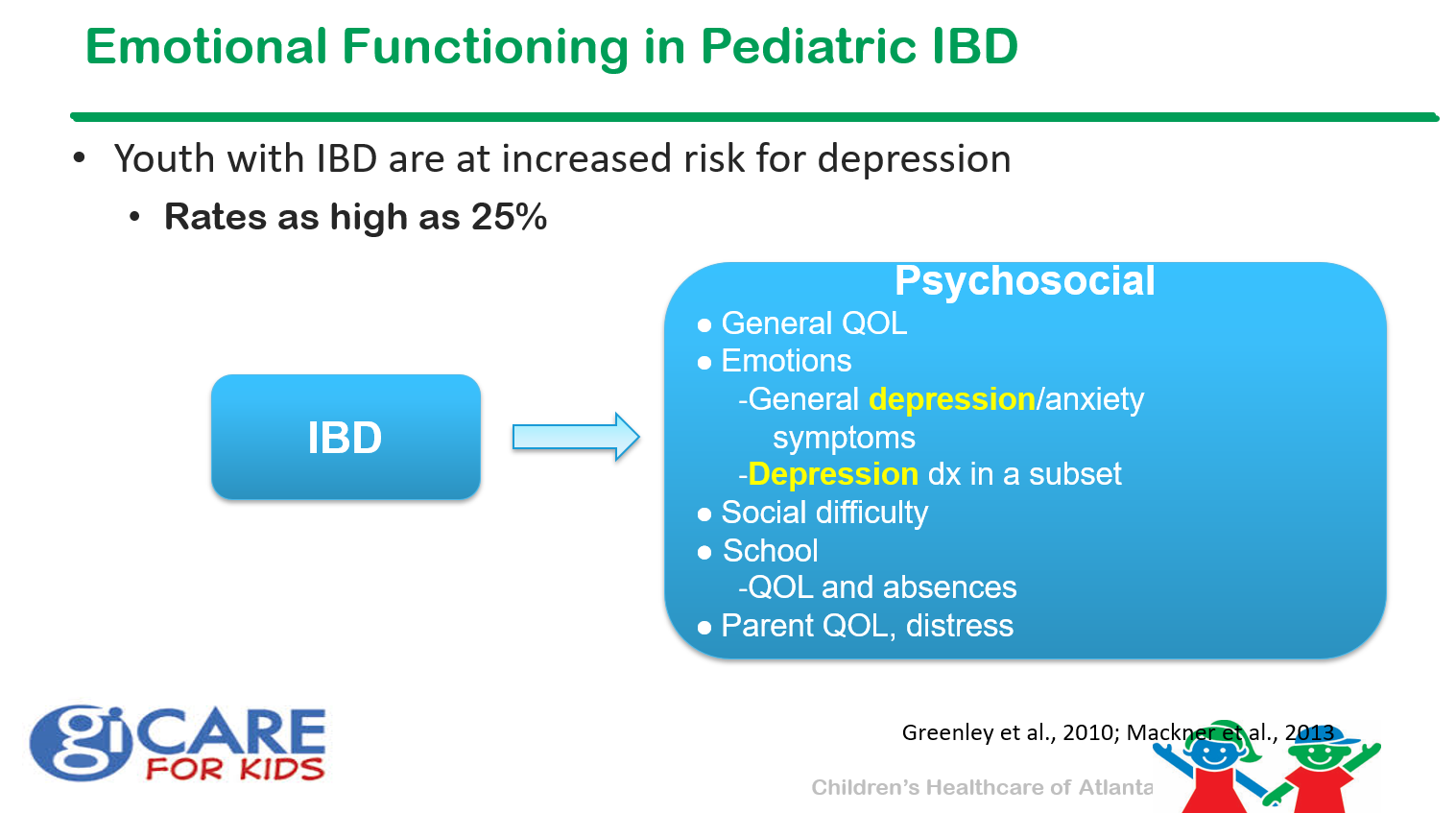
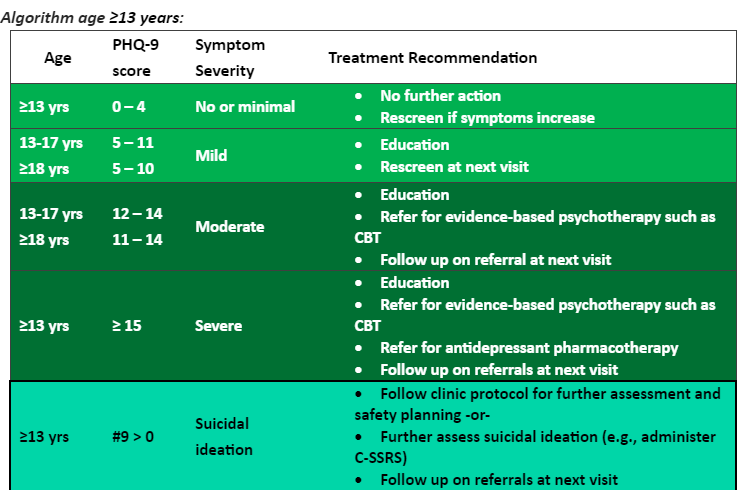
Patient education materials:
#1 Ulcerative Colitis For Dummies | UC Patient Resource This link connects to a free educational book promoted by Salix pharmaceuticals. In order to receive a free download, you have to register and include your email. I have not read this book but other similar books (eg. Colonoscopy for Dummies) by Salix have been well-written.
#2 This link, ow.ly/sPX95, is to the ImproveCareNow visit planner website. It poses of ~ 8 questions and a text box for “my list of things I’m concerned about and questions that I have.” Families that use this planner may help themselves achieve more comprehensive care.
Anyone who follows this blog knows that I really enjoy a good study acronym. The Program of Ulcerative Colitis Research Studies Utilizing an Investigational Treatment (PURSUIT) is responsible for two papers in the most recent Gastroenterology issue:
- Gastroenterol 2014; 146: 85-95
- Gastroenterol 2014; 146: 96-109
PURSUIT is composed of >200 sites from around the world. The first study was a combined phase 2 and phase 3 study. It enrolled 1064 adults with moderate to severe ulcerative colitis (UC) who were randomly assigned to either placebo, 200/100 mg or 400/200 mg of SC golimumab at 0 and 2 weeks. All patients were naive to previous anti-TNF therapies. The average duration of disease was 6 years among the participants. The primary endpoint of the phase 3 part of this study was the clinical response at 6 weeks. Clinical response was at least a 30% improvement and a ≥3-point improvement in the Mayo score. At baseline, the average Mayo score was 8.
- The golimumab groups had response rates of 51.8% and 55.0% respectively compared to 29.7% for placebo.
- Approximately 18% of patients achieved a clinical remission with treatment compared with 6% of placebo patients.
- Approximately 10% had healed mucosa compared with ~4% in the placebo group.
- Adverse events: Rates of serious infection were 1.8% for the active treatment group compared with 0.5% for placebo-treated patients. In the 400/200 mg dosing group, there was 1 death attributed to peritonitis and sepsis after multiple procedures for ischiorectal abscess repair. In addition, a single case of demyelination was noted in this group.
The second study, a phase 3 double-blind trial, evaluated the efficacy of maintenance treatment of 50 mg or 100 mg SC every 4 weeks in those with a successful induction (n=464). This study took place at 251 centers between 2007-2011. At 54 weeks, the actively-treated gourds had maintained a clinical response, using the Mayo score, in 47.1% and 50.6% respectively compared to 31.4% for placebo. Antibodies to golimumab developed in 2.9%, two-thirds of these antibodies were neutralizing. Antibody formation was lower in those receiving concomitant immunomodulators. 4 cases of tuberculosis were noted from endemic regions despite previous screening. Overall, infections occurred in 28% of those treated with placebo compared with 39% of those treated with golimumab.
During the course of the study, three deaths were reported, all in the 100 mg golimumab maintenance group. The causes were malnutrition/sepsis, cardiac failure/thrombosis, and disseminated tuberculosis (patient was receiving isoniazid). After the study, another 6 deaths were reported, including two in the placebo group. Three malignancies were reported through week 54 in patients receiving golimumab maintenance, two of these presented in the induction period while receiving placebo rectal cancer and thyroid cancer) and one (lung adenocarcinoma) occurred in a patient with a 40-year smoking history who received golimumab for induction and maintenance.
Taken together, about 25% of patients randomized to and maintained on golimumab achieved a clinical response lasting >1 year; similarly, about 17% had clinical remission at 1 year.
In the commentary (page 13-15), Stephen Hanauer notes that better response was noted with higher serum levels and there remains “a strong possibility that optimal dosing was not achieved.” He and the authors comment on the observation that less-severe patients had a better response, indicating that “greater disease severity may be correlated with more rapid clearance.”
Bottomline: These studies demonstrate that golimumab is an effective treatment for UC with a similar risk of adverse reactions as other anti-TNF agents. The published studies are complicated and take some time to analyze.
Plus more references:
Gastroenterol 2014; 146: 110-18. “Adalimumab therapy is associated with reduced risk of hospitalization in patients with ulcerative colitis.” Data for this studies was derived from ULTRA1 and ULTRA2 trials with 963 patients. Risk for hospitalization, whether due to UC or all-causes, was reduced between 40-50% compared to placebo within the first 8 weeks of adalimumab administration.
Clin Gastroenterol Hepatol 2013; 11: 1538-49. Excellent review on pouchitis. Figure 5 (pg 1545) provides a nice treatment algorithm. Initial approach is antibiotics (metronidazole or ciprofloxacin); in those responsive, either prn therapy or chronic treatment. In those not responsive, look for pathogens (eg. CMV and C difficile) or determine it is immune-mediated (PSC-associated, IgG4-associated, or autoimmune). The immune-mediated may respond to 5-ASA/budesonide or immunomodulators.
Clin Gastroenterol Hepatol 2013; 11: 1601-08. This case-control study with 141 UC controls and 59 patients who developed colorectal neoplasia found that increased inflammation was associated with colorectal neoplasia. Use of immune modulators reduced the risk of colorectal neoplasia.
Related blog post:
Simponi (Golimumab) Approved for Ulcerative Colitis | gutsandgrowth