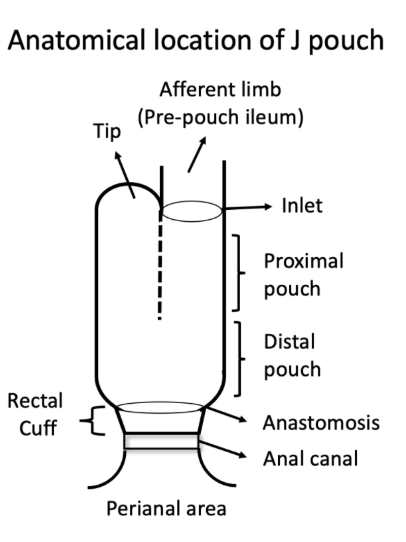
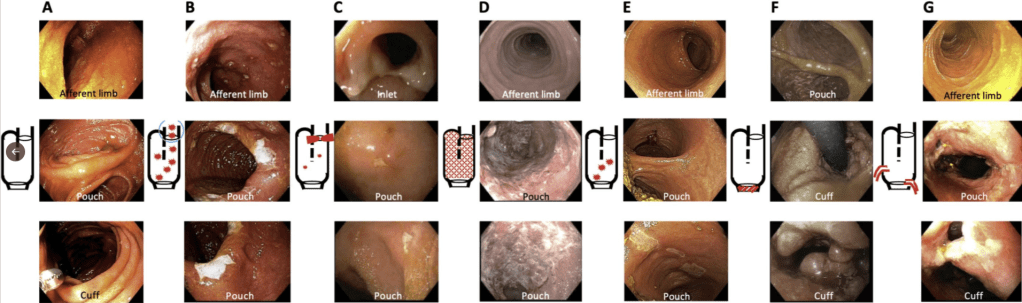
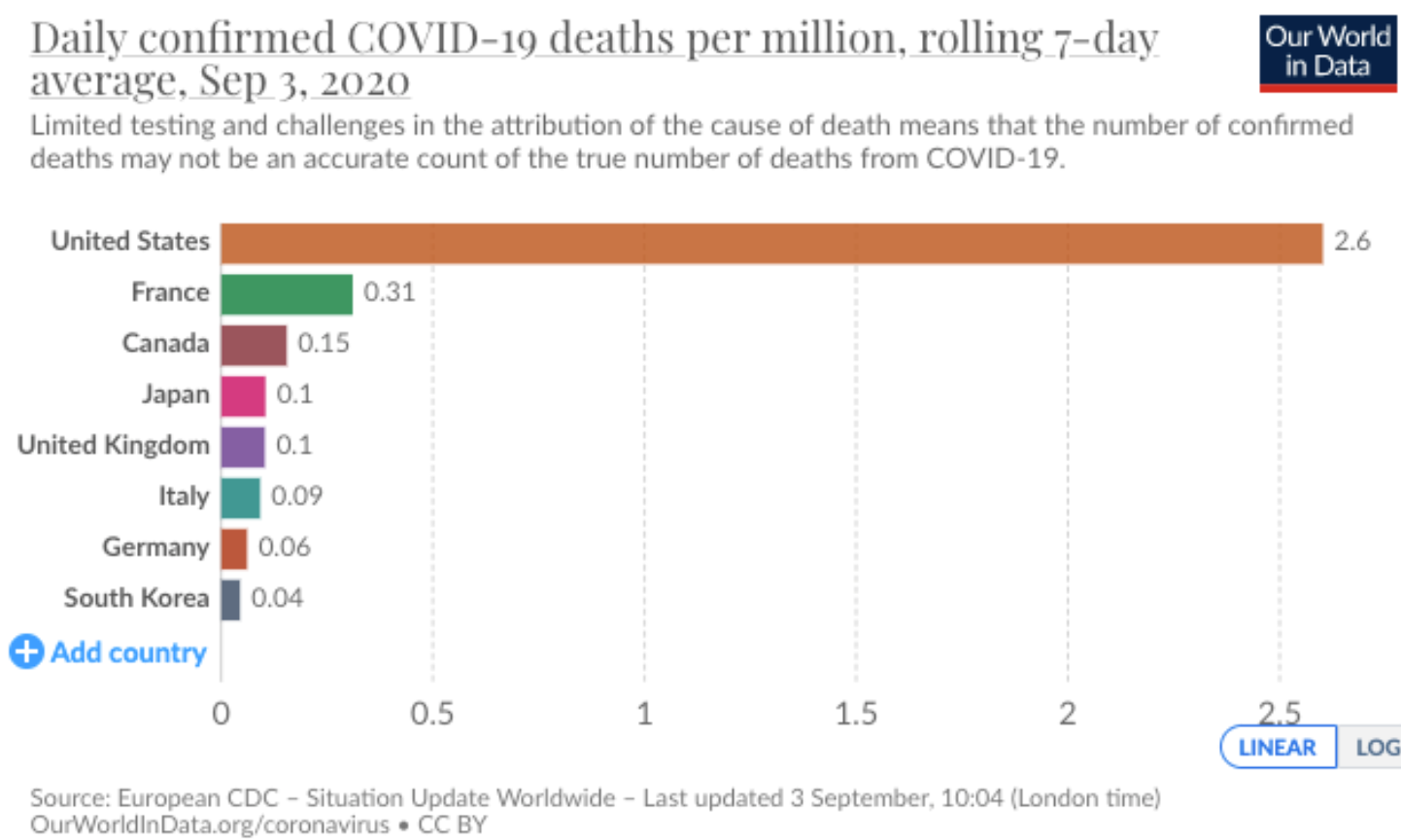
- EL Barnes, M Agrawal et al. Gastroenterol 2024; 166: 59-85. Open Access! AGA Clinical Practice Guideline on the Management of Pouchitis and Inflammatory Pouch Disorders
- Gastroenterol 2024; 166: 86. Open Access! Decision-support tool (1 page summary)
- Gastroenterol 2024; 166: 87. Open Access! Spotlight (1 page summary)
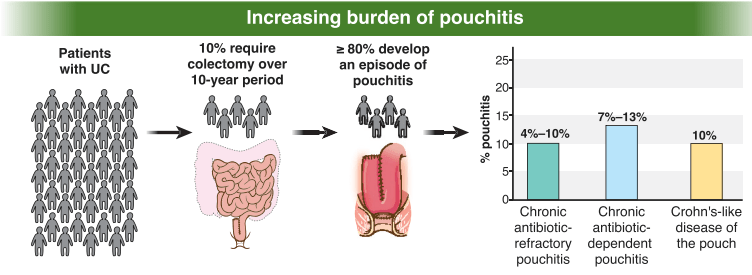
Key recommendations
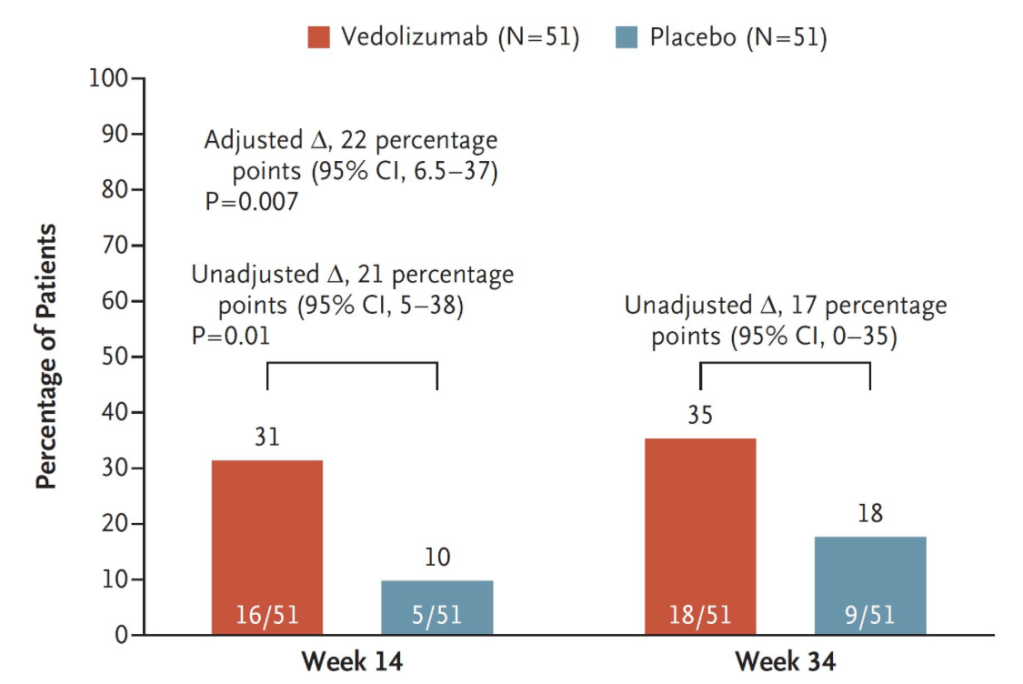
- AGA recommends metronidazole and/or ciprofloxacin as preferred treatment of pouchitis with duration of treatment 2-4 weeks.
- For Crohn’s-like disease of the pouch, AGA guideline recommends using either ileal-release budesonide or advanced immunosuppressive agents (eg. Biological therapies and small molecule therapies)
- “In patients with cuffitis, topical therapies should be the first-line therapy, such as mesalamine suppositories, corticosteroid suppositories, or corticosteroid ointment applied directly to the cuff. Biological therapies and small molecule therapies are recommended in refractory cases


Related blog posts: