JG Hashash et al.Gastroenterology 166; 521-532. Open Access! AGA Clinical Practice Update on Diet and Nutritional Therapies in Patients With Inflammatory Bowel Disease: Expert Review
There are 12 “best practice” recommendations. Here are a few of them:
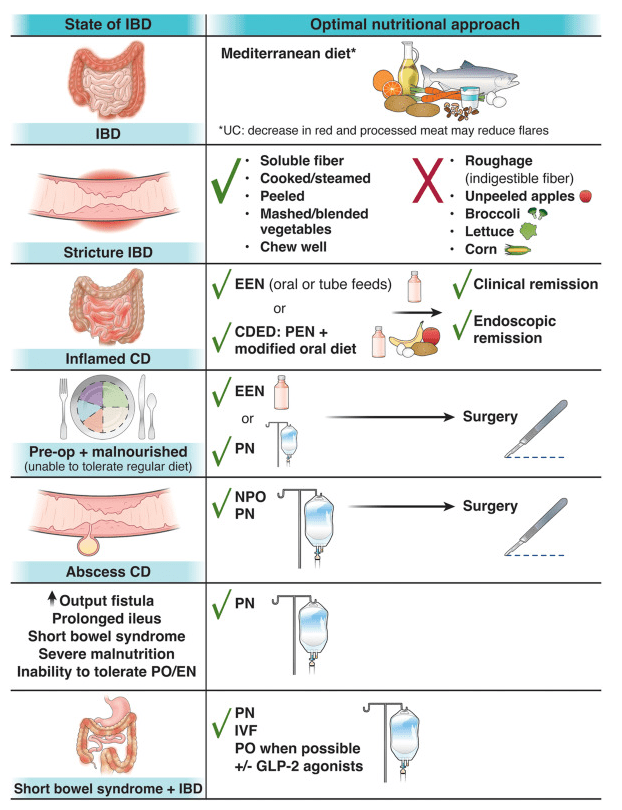
- Best Practice Advice 1: Unless there is a contraindication, all patients with IBD should be advised to follow a Mediterranean diet rich in a variety of fresh fruits and vegetables, monounsaturated fats, complex carbohydrates, and lean proteins and low in ultraprocessed foods, added sugar, and salt for their overall health and general well-being. No diet has consistently been found to decrease the rate of flares in adults with IBD. A diet low in red and processed meat may reduce ulcerative colitis flares, but has not been found to reduce relapse in Crohn’s disease.
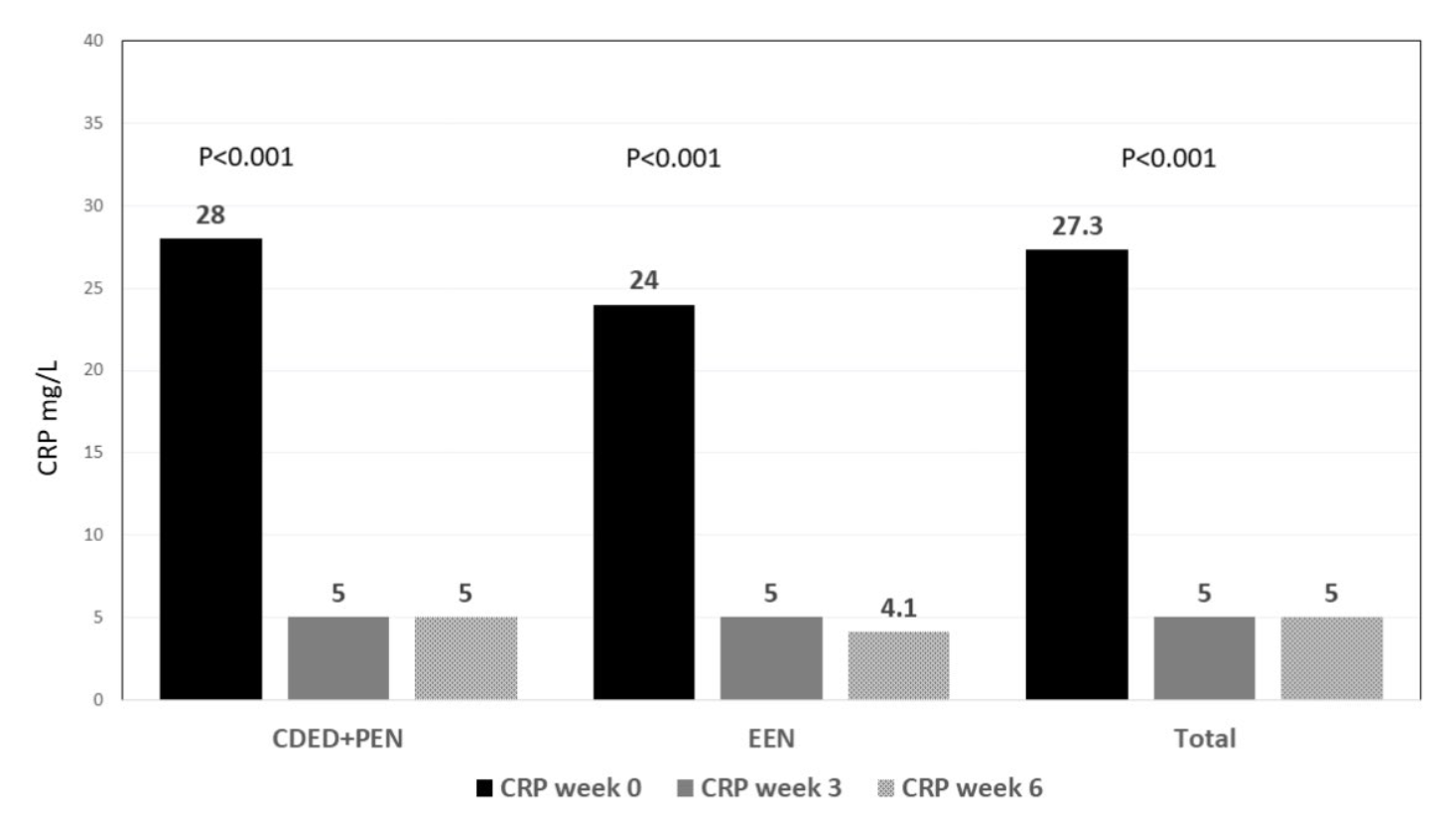
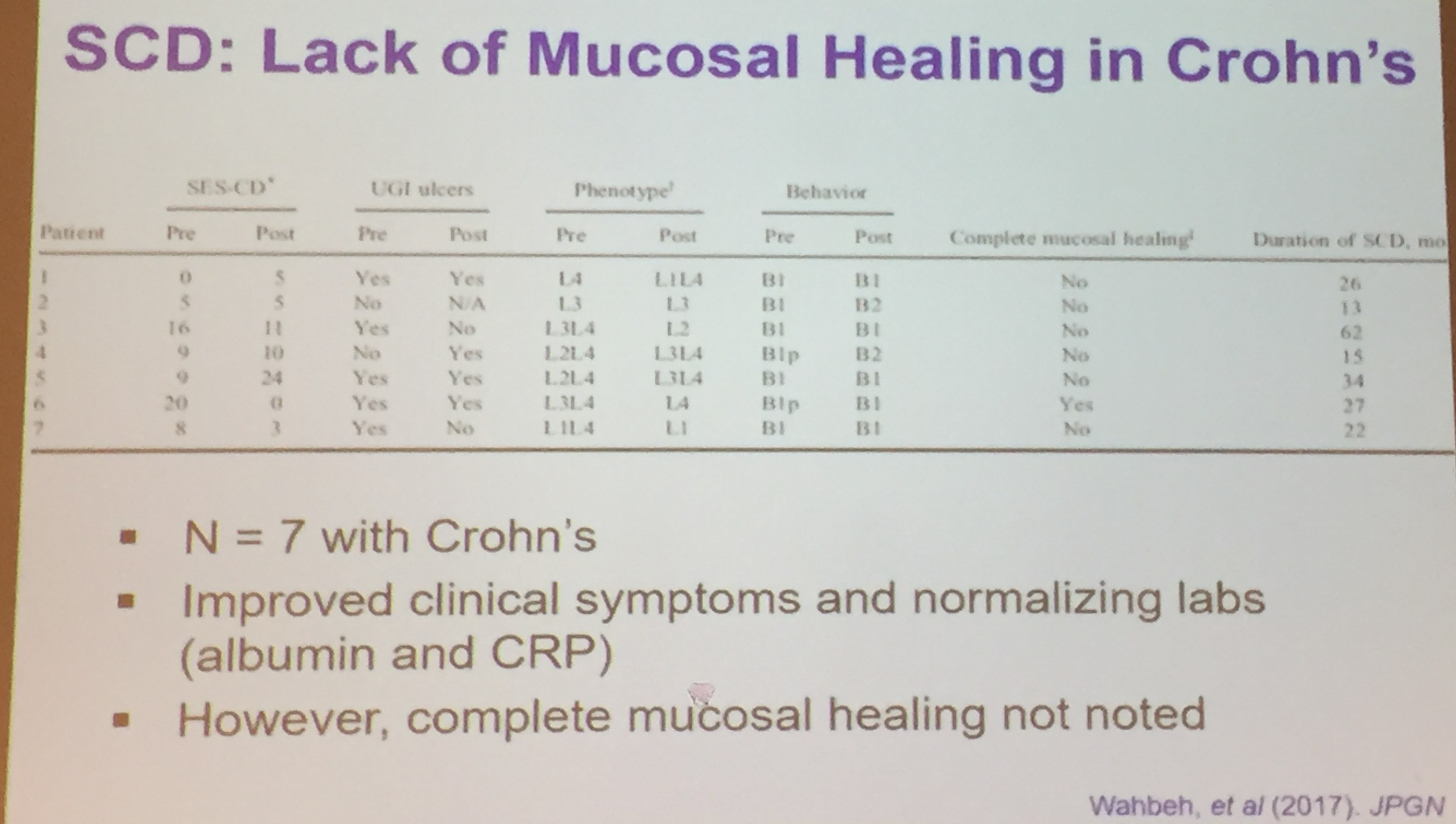
- Best Practice Advice 3: Exclusive enteral nutrition using liquid nutrition formulations is an effective therapy for induction of clinical remission and endoscopic response in Crohn’s disease, with stronger evidence in children than adults. Exclusive enteral nutrition may be considered as a steroid-sparing bridge therapy for patients with Crohn’s disease.
- Best Practice Advice 6: In patients with IBD who have an intra-abdominal abscess and/or phlegmonous inflammation that limits ability to achieve optimal nutrition via the digestive tract, short-term parenteral nutrition may be used to provide bowel rest in the preoperative phase to decrease infection and inflammation as a bridge to definitive surgical management and to optimize surgical outcomes.
- Best Practice Advice 7: We suggest the use of parenteral nutrition for high-output gastrointestinal fistula, prolonged ileus, short bowel syndrome, and for patients with IBD with severe malnutrition when oral and enteral nutrition has been trialed and failed or when enteral access is not feasible or contraindicated.
- Best Practice Advice 10: All patients with IBD should be monitored for vitamin D and iron deficiency. Patients with extensive ileal disease or prior ileal surgery (resection or ileal pouch) should be monitored for vitamin B12 deficiency.
- Best Practice Advice 12: Breastfeeding is associated with a lower risk for diagnosis of IBD during childhood. A healthy, balanced, Mediterranean diet rich in a variety of fruits and vegetables and decreased intake of ultraprocessed foods have been associated with a lower risk of developing IBD.
AGA has a summary and video here: What you need to know about diet and nutritional therapies for IBD patients
Related blog posts:
- The Quality of Evidence for Dietary Treatments in Inflammatory Bowel Disease
- Mediterranean Diet vs Specific Carbohydrate Diet for Crohn’s Disease
- Outcomes with Enteral Nutrition
- Can IBD Be Treated with Diet Alone?
- Dietary Therapy for Inflammatory Bowel Disease –Useful Update
- Top Lecture: Enteral Nutrition for Crohn’s Disease
- Head-to-Head: Nutritional Therapy versus Biological Therapy in Pediatric Crohn’s Disease | gutsandgrowth
- Ultraprocessed Food and the Risk of Inflammatory Bowel Disease
- Risk Factors for Inflammatory Bowel Disease: Ultra-Processed Food (Part 1)
- No Solid Conclusions” for Alternative/Complementary Therapies for Inflammatory Bowel
- More Evidence That A Proinflammatory Diet May Increase the Risk of Crohn’s Disease | gutsandgrowthDisease | gutsandgrowth
- Trial by Diet Approach for Crohn’s Disease in Children (this blog post has link to 16 other dietary therapy posts)