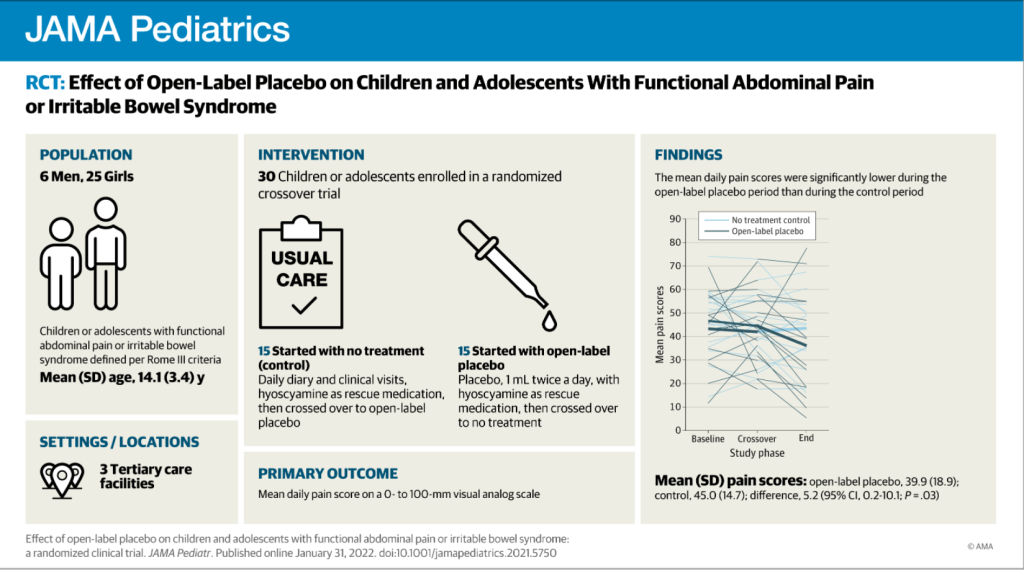
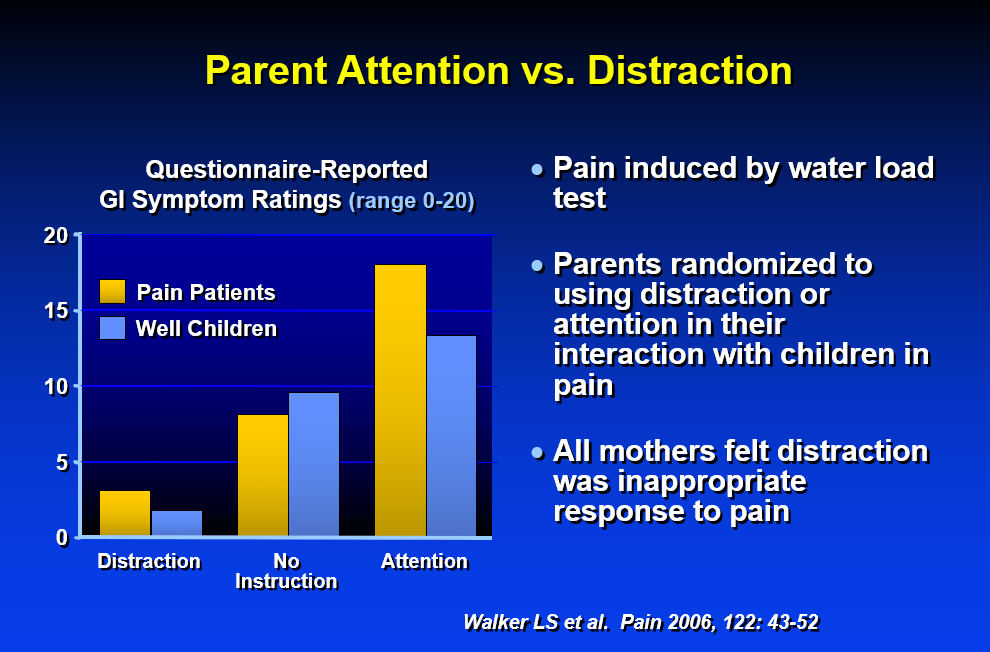
Recently, Dr. Neha Santucci gave our group an excellent update on disorders of gut-brain interaction. My notes below may contain errors in transcription and in omission. Along with my notes, I have included many of her slides.


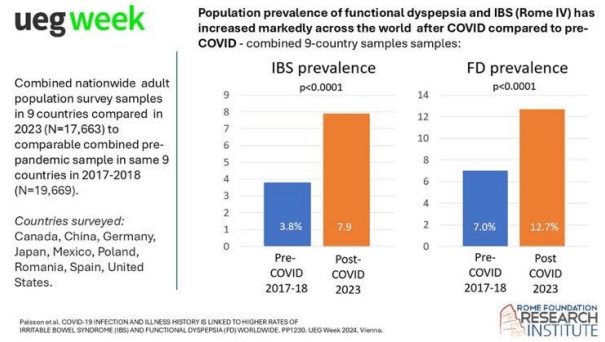
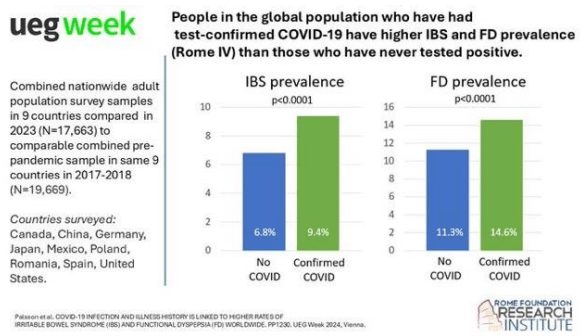
This study in adults showed a greater increase in functional dyspepsia compared to IBS.
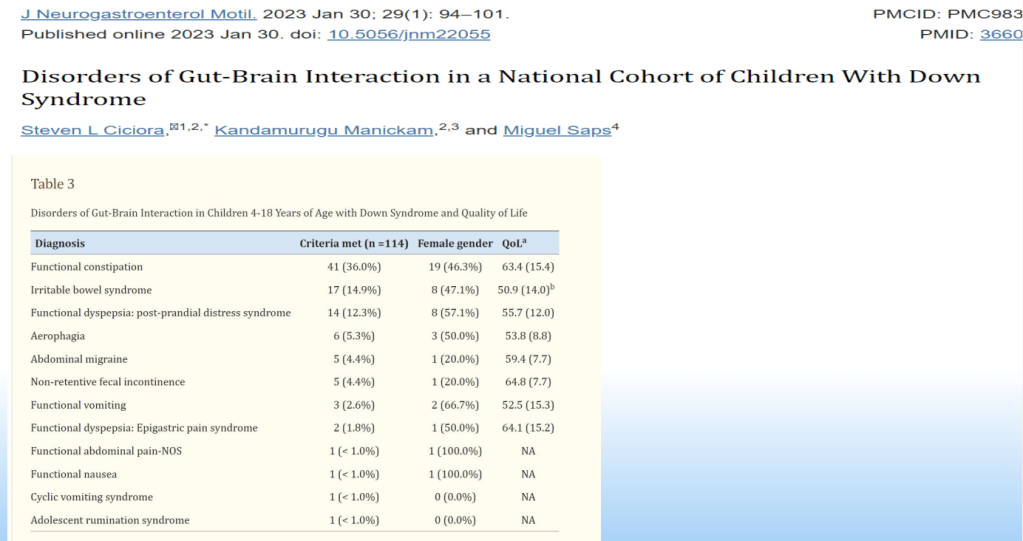
DGBIs occur in Children with Down syndrome. This cohort showed high rates of functional constipation (36%), irritable bowel syndrome (14.9%), functional dyspepsia (12.3%) and aerophagia (5.3%).

- Development of DGBIs is influenced by psychological factors, early life events, chronic stress, gut motility, inflammation, mucosal immune activation and altered gut microbiota
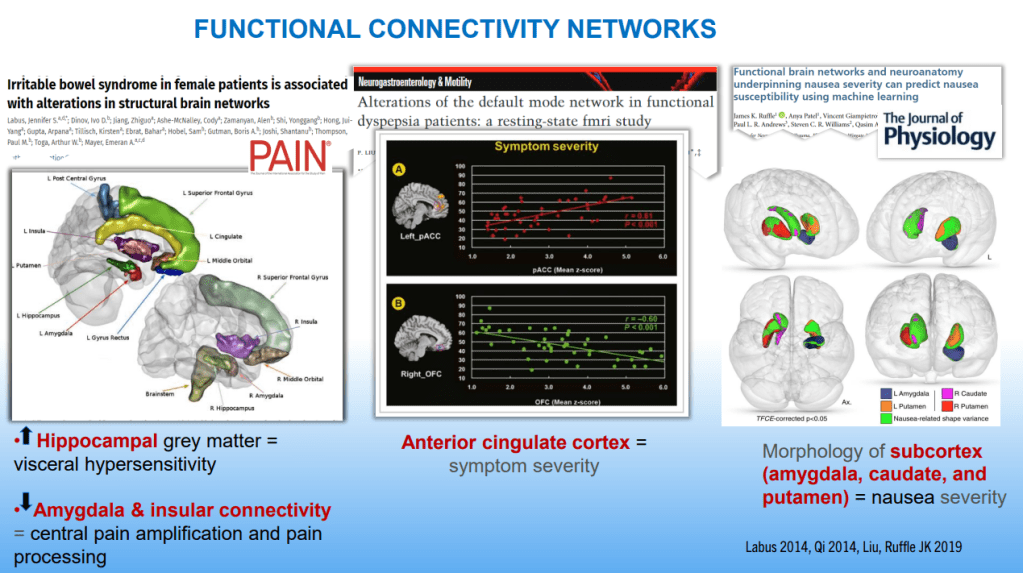
- DGBIs are associated with altered brain networks
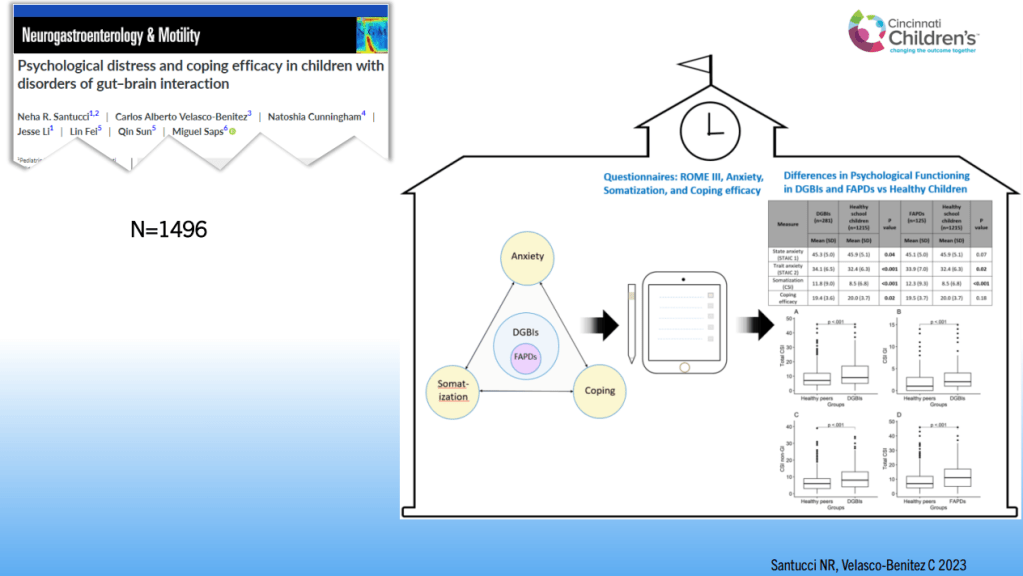
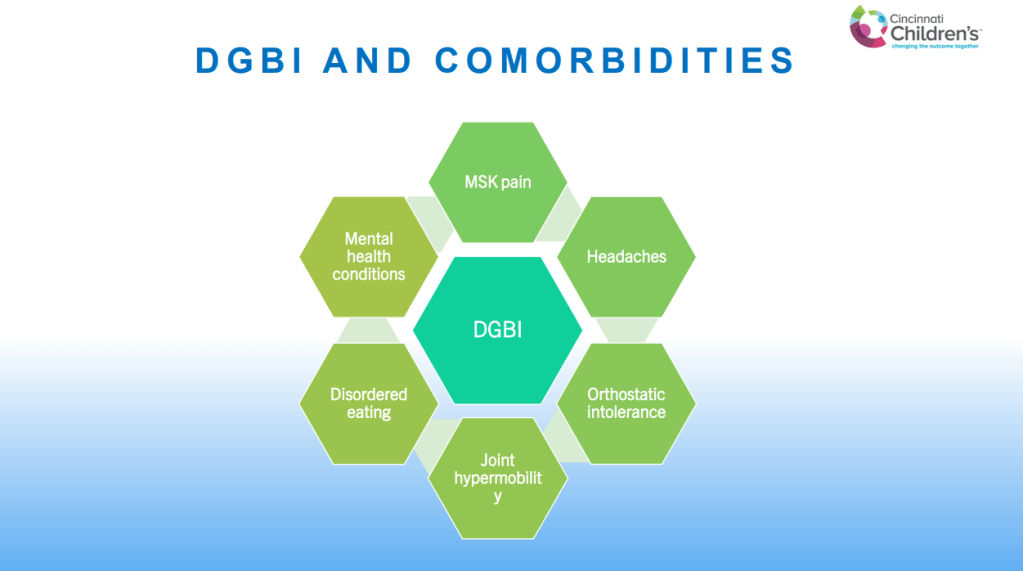
- DGBIs are associated with a number of comorbidities including mental health disorders, joint hypermobility, headaches, POTS, musculoskeletal pain, disordered eating, and poor sleep
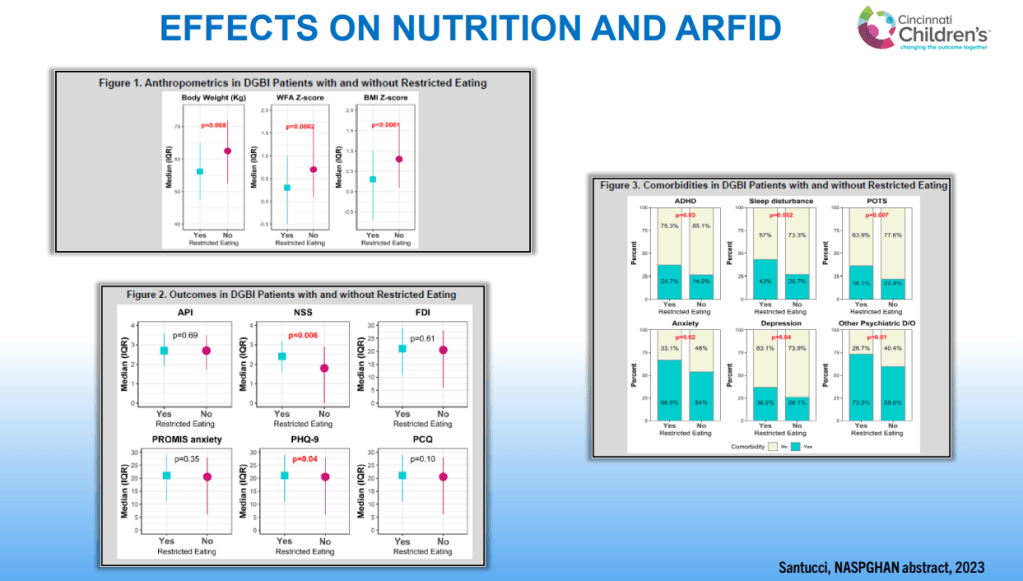
- Individuals with DGBIs are at increased risk of eating disorders including ARFID. Presence of ARFID with DGBIs has been associated with more anxiety, depression, ADHD and sleep disturbance
- Poor sleep in previous night is associated with increased pain the next day in individuals with DGBIs
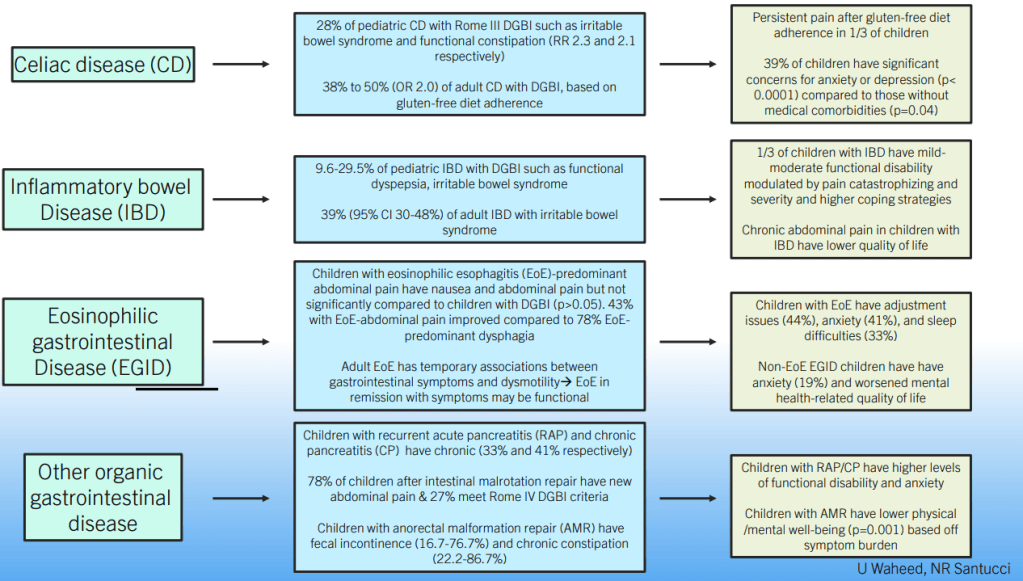
- DGBIs are common in children with organic diseases, including IBD, EoE, Celiac disease, Recurrent Pancreatitis, Malrotation and Anorectal disorders
- Up to 50% of pediatric GI visits are for functional disorders and ~25% of all children have DGBIs
- Strive to make a positive diagnosis (rather than simply a diagnosis of exclusion)
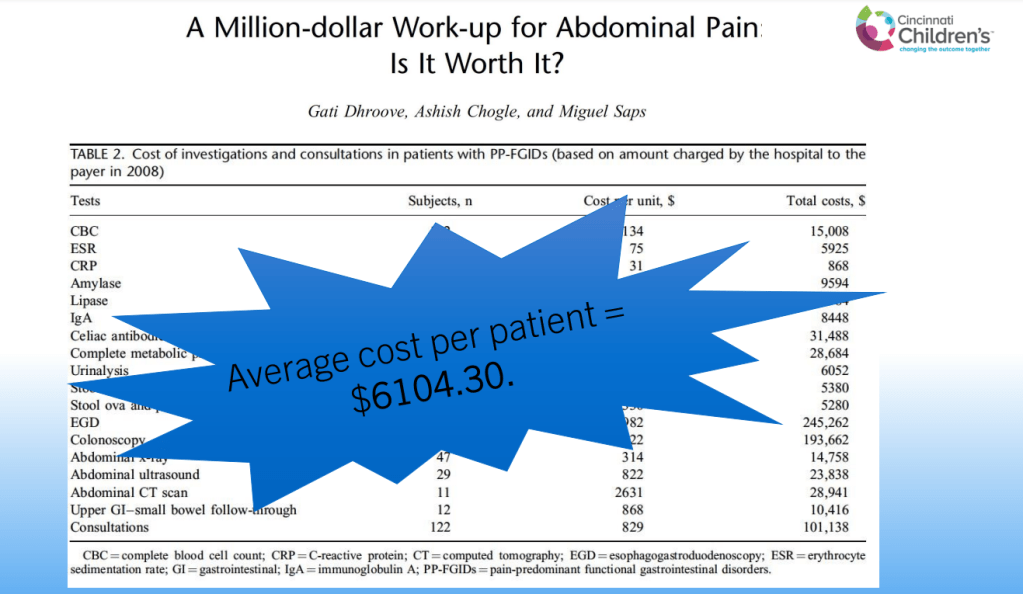
- Avoid excessive testing
- Dyspepsia and gastroparesis are not distinct disorders and likely exist on a spectrum (some of the same treatments for both)
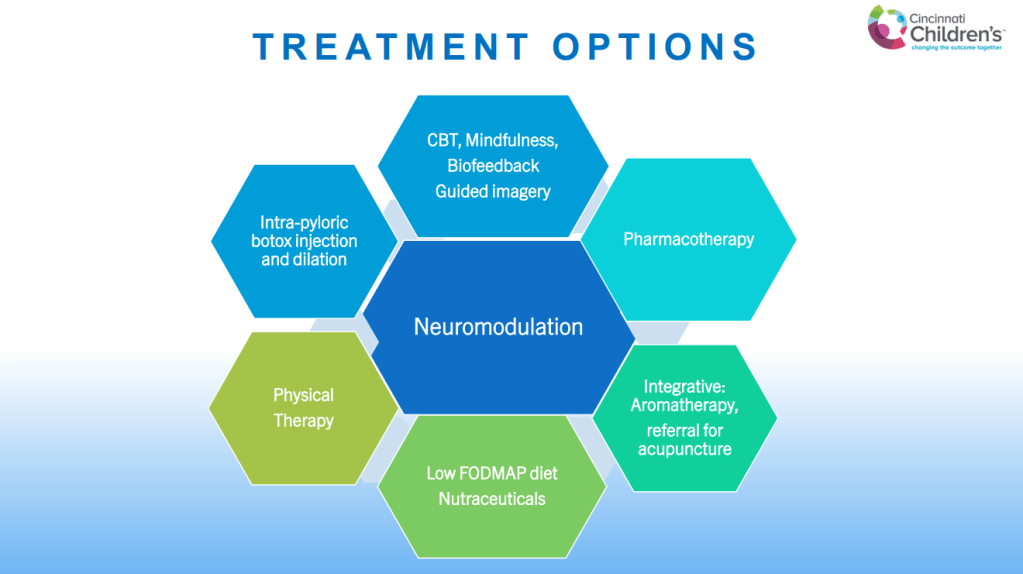
- First treatment goals: develop a good rapport with family and focus on improved functioning





Related blog posts:
- Advice on Abdominal Pain for Everyone Who Cares for Children In this post some of the pithy observations from John Apley:
-“The more time the doctor spends on the history, the less time he is likely to spend on treatment.”
-“Doctors who treat the symptoms tend to file a prescription. Doctors who treat the patient are more likely to offer guidance.”
-“It is a fallacy that a physical symptoms always has a physical cause and needs a physical treatment.”
-“Anxiety like courage is contagious.”
- #NASPGHAN19 Postgraduate Course -part 3
- Narrowing the Workup for Chronic Abdominal Pain –Carlo DiLorenzo Was Right!
- Pain changes brain
- Are Gastroparesis and Functional Dyspepsia Part of the Same Problem?
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.