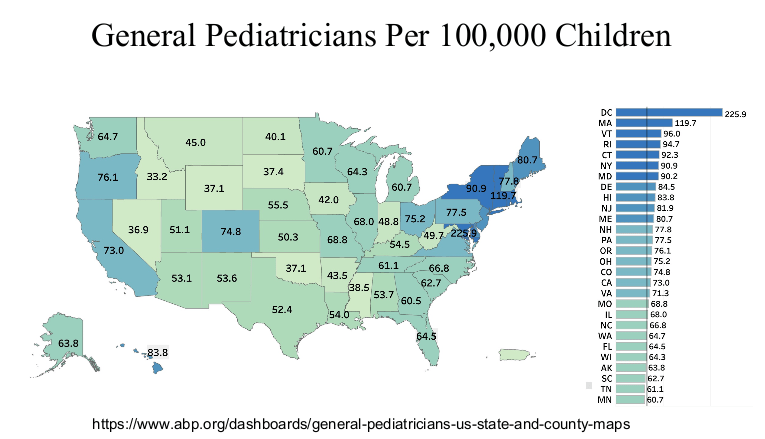
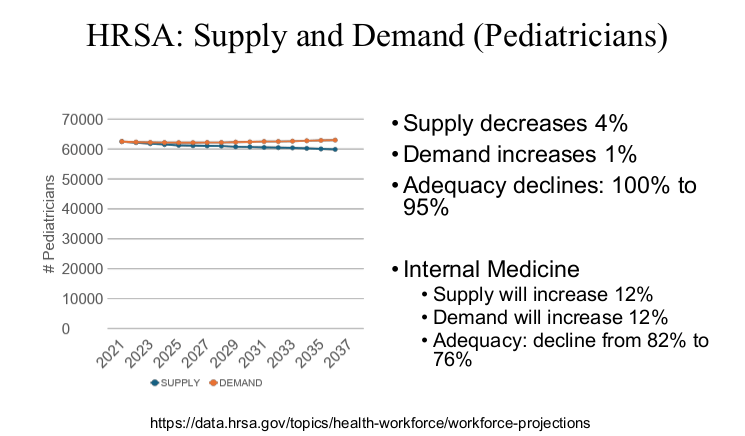
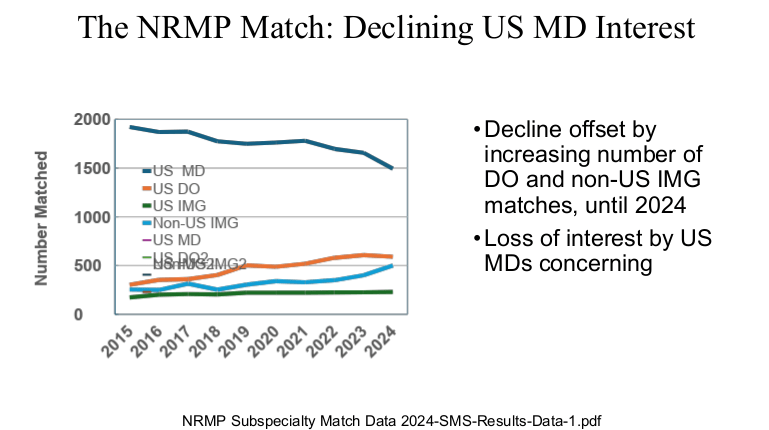
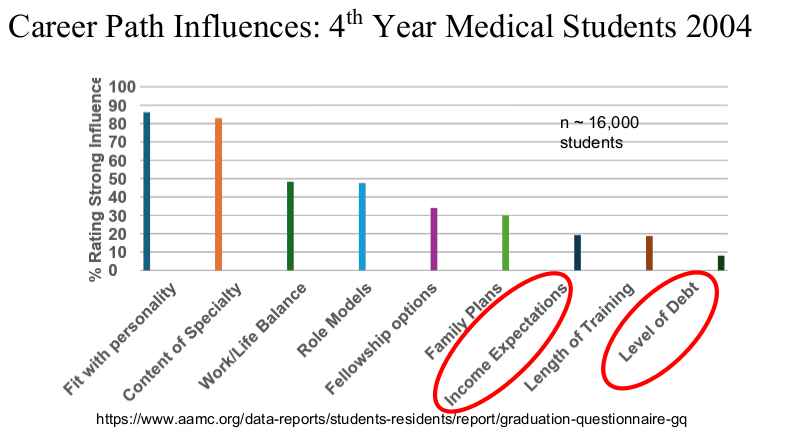
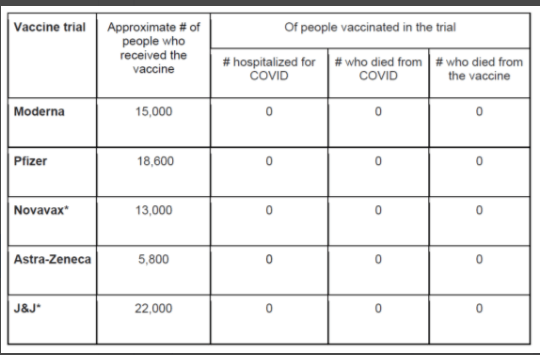
R Smith-Bindman et al. NEJM 2025; 393: 1269-1278. Medical Imaging and Pediatric and
Adolescent Hematologic Cancer Risk
Methods: This was a retrospective cohort of 3,724,623 children born between 1996 and
2016 in six U.S. health care systems and Ontario, Canada, until the earliest of cancer
or benign-tumor diagnosis, death, end of health care coverage, an age of 21 years, or December 31, 2017.
Key findings:
- During 35,715,325 person-years of follow-up (mean, 10.1 years per person), 2961 hematologic cancers were diagnosed, primarily lymphoid cancers (2349 [79.3%]), myeloid cancers or acute leukemia (460 [15.5%]), and histiocytic- or dendritic-cell cancers
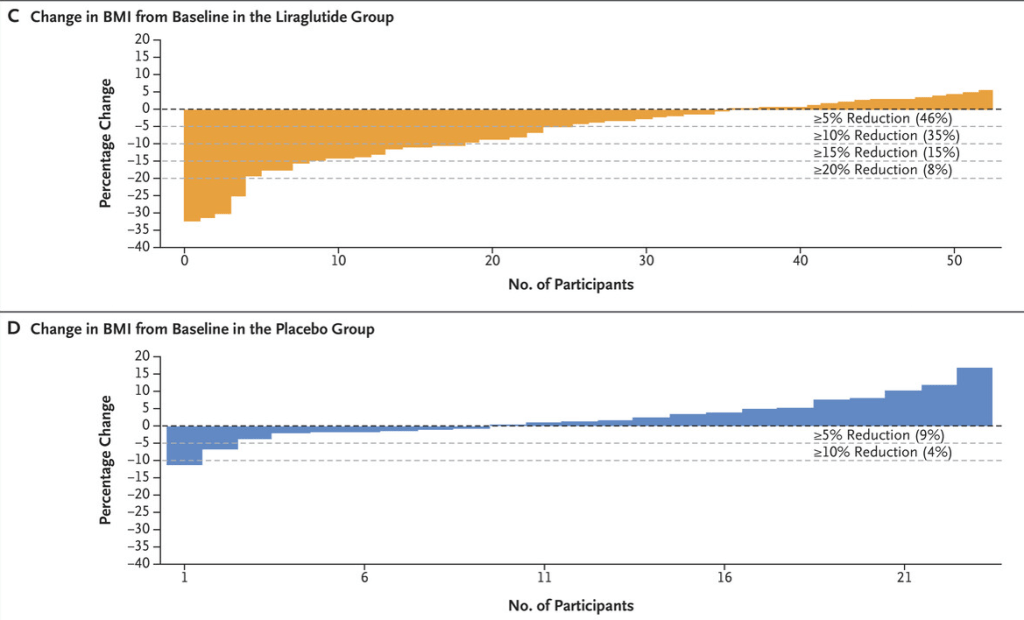
(129 [4.4%]). - The excess cumulative incidence of hematologic cancers by 21 years of age among children exposed to at least 30 mGy (mean, 57 mGy) was 25.6 per 10,000
- The authors estimated that 10.1% of hematologic cancers may have been attributable to radiation exposure from medical imaging, with higher risks from the higher-dose medical-imaging tests such as CT
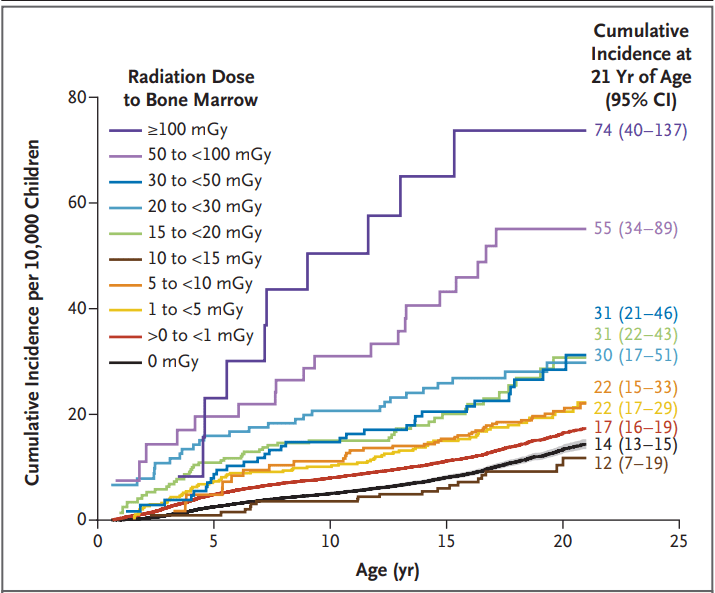
Radiation Dose to Bone Marrow among Children without Down’s Syndrome
Discussion Points:
- “A 15-to-30-mGy exposure equivalent to one to two CT scans of the head was associated with an increased risk by a factor of 1.8”
- “Although CT and other radiation-based imaging techniques may be lifesaving, our
findings underscore the importance of carefully considering and minimizing radiation exposure during pediatric imaging to protect children’s long-term health” - “Research on Japanese atomic-bombing survivors showed that leukemia rates peaked 6 to 8 years after exposure, with excess risk lasting for more than five decades, particularly for acute myeloid leukemia”
- This study tried to avoid concerns about reverse causation — in which imaging is performed because of existing cancer symptoms –by lagged exposures by 6 and 24 months
- “The increasing use of low-value imaging in children and excessive radiation doses in CT are well documented…In many cases, reducing the imaging dose or substituting magnetic resonance imaging or ultrasonography may be more feasible than avoiding imaging altogether”
While the risks in aggregate appear quite substantial, the editorial (L Morton. NEJM 2025; 393; 1337-1339.Studying Cancer Risks Associated with Diagnostic Procedures –Interpret Wisely) makes the point that the risks for the individual are very small. “Fewer than 1% of youths in this study accumulated doses of 30 mGy or more from medical imaging and even at this exposure level, the excess cumulative incidence of hematologic cancers was low (25.6 per 10,000)…we need to ensure that all involved in medical imaging…wisely interpret the results…to understand the balance of the very small risks and the notable benefits of necessary imaging examinations to provide optimal patient care.”
My take: This study is a reminder to carefully evaluate the benefits, risks and alternatives when using ionizing radiation studies.
Related blog posts: