L McIntyre et al. NEJM 2025; 393: 660-670. A Crossover Trial of Hospital-Wide Lactated Ringer’s Solution versus Normal Saline
Background: “A single-center, multiple-crossover trial involving noncritically ill patients in the emergency department found that balanced crystalloid fluids were associated with a lower incidence of major adverse kidney events at 30 days than normal saline. A systematic review of 13 randomized, controlled trials comparing balanced crystalloids with normal saline in a total of 35,884 critically ill participants showed no significant difference in mortality (17.4% with balanced crystalloids and 18.2% with saline; relative risk, 0.96; 95% confidence interval [CI], 0.91 to 1.01) or in the incidence of the use of renal replacement therapy (5.6% and 6.0%, respectively; relative risk, 0.95; 95% CI, 0.81 to 1.11) in trials with a low risk of bias.4 However, the investigators in that analysis and those in another patient-level meta-analysis involving a Bayesian approach concluded that there is a high probability that balanced crystalloids are associated with lower in-hospital mortality and a lower incidence of the use of renal replacement therapy than normal saline.5“
Methods of th “FLUID” trial: 3 hospitals used lactated ringer’s (LR) and 4 hospitals used normal saline throughout hospital setting for 12 weeks. Then after a 1-2 week washout period, the hospitals switched to the other fluid for 12 weeks.

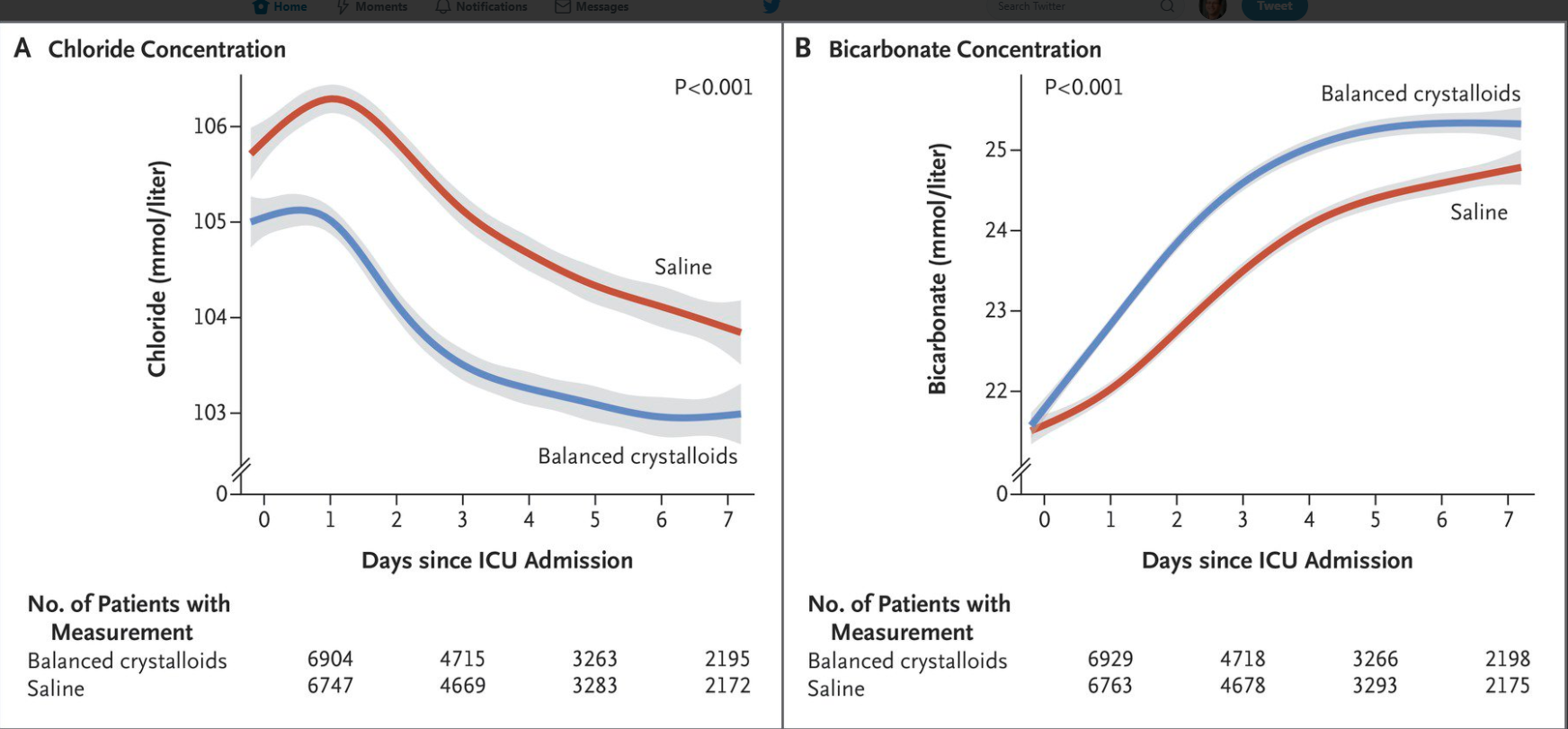
Key finding:

Discussion: “A limitation of this trial was the inability to recruit the total of 16 hospitals as originally planned owing to the Covid-19 pandemic. Hence, the trial had less power to detect differences that were small — but important to patients — at the level of the hospital or health care system…Our findings align with those of recent meta-analyses of randomized, controlled trials that suggest a small but clinically relevant reduction in mortality with balanced crystalloids as compared with normal saline.”
My take: This study did not show a significant difference in death or readmission at 90 days. Yet, lactated ringer’s is probably just a bit better fluid for most adult patients. In the pediatric population, more studies are needed.
Related blog posts:
- Choosing the Right IV Fluids -Risk of hypokalemia with isotonic fluids with low potassium content
- AAP Recommends Isotonic Maintenance IV Fluids
- Optimizing Fluid Resuscitation in Pediatric Acute Pancreatitis
- More Data Supporting Lactated Ringers for Acute Pancreatitis
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition