JA Mark, RE Kramer. JPGN 2021; 73: 542-547. Ketorolac Is Safe and Associated With Lower Rate of Post-Endoscopic Retrograde Cholangiopancreatography Pancreatitis in Children With Pancreatic Duct Manipulation
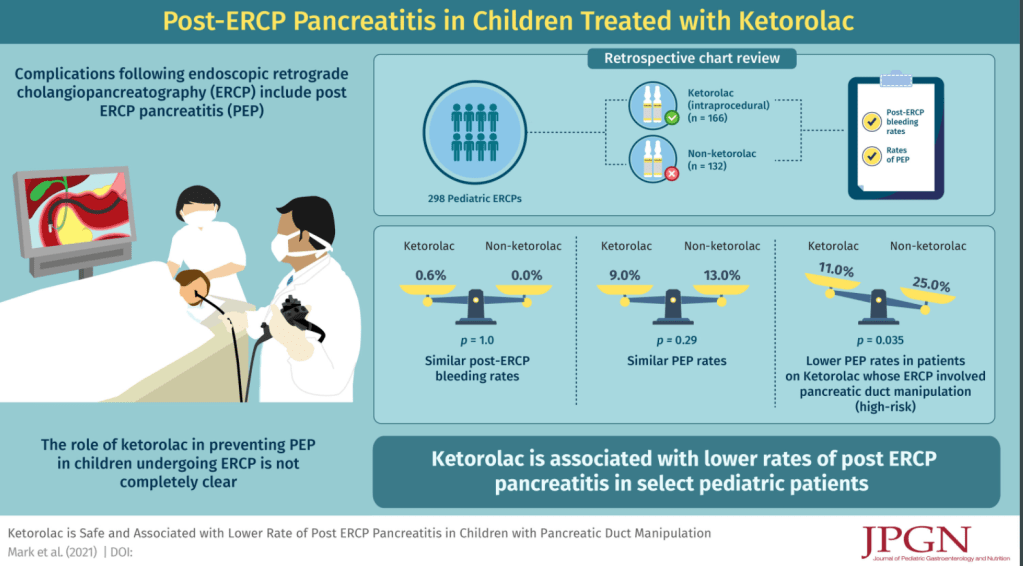
Methods: This retrospective chart review (n=298) examined the outcomes of pediatric patients who receivied ketorolac during ERCP compared to those who did not; ketorolac, was dosed using weight-based dosing (0.5 mg/kg/dose max 30 mg).
Key findings:
- Most common indications for ERCP were choledocholithiasis, biliary stricture, and chronic/recurrent pancreatitis
- Therapeutic ERCP: 91% of ketorolac group and 89% of non-ketorolac group; sphincterotomy was performed in 55% of both groups
- Post-ERCP bleeding rates were not significantly different between ketorolac and non-ketorolac groups (0.6% vs 0%, P = 1)
- PEP (post-ERCP pancreatitis) rates were not significantly different between the ketorolac and no ketorolac group 15/166 vs 17/132 (9% vs 13%, P = 0.29)
- Patients who had cannulation and/or injection of the PD had significantly higher rates of PEP (23/140 (16%) vs 9/158 (6%), P < 0.003)
- For high-risk pediatric patients with injection of contrast into and/or cannulation of the pancreatic duct, the rates of PEP were significantly lower for patients who received ketorolac (11% vs 25%, P = 0.035). In Table 2, the authors indicate that PEP in this high risk group occurred in 11/88 (12.5%) [mild discrepancy from abstract of 11%] of ketorolac group and 13/52 (25%)
It is possible that ketorolac (or other NSAIDs) in all patients may be beneficial but difficult to demonstrate without a larger cohort. In adults, “indomethacin reduces risk and severity of PEP in both high and average risk adults…and there is some evidence that NSAIDS given before ERCP may be more effective than those given later.” Thus, the authors state that use of ketorolac could be administered to all ERCP patients beforehand (w/o contraindication) or limited to the higher risk patients.
My take: In pediatric patients needing an ERCP, those with high-risk features (eg. injection of contrast into and/or cannulation of the pancreatic duct), use of ketorolac is likely to reduce the frequency of post-ERCP pancreatitis.
Related blog posts:
- Indomethacin to prevent post-ERCP pancreatitis | gutsandgrowth
- Why an ERCP Study Matters to Pediatric Care