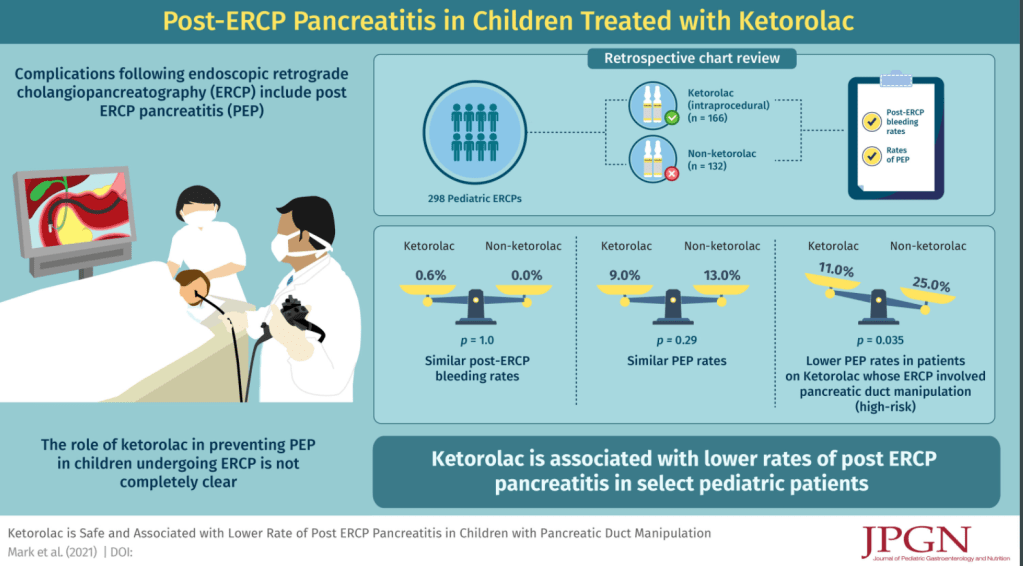
While there are pediatric patients who undergo endoscopic retrograde cholangiopancreatography (ERCP), this is a relatively infrequent occurrence. Nevertheless, a recent study has a couple useful clinical pearls that may have broader application.
- Clin Gastroenterol Hepatol 2014; 12: 303-07.
- Clin Gastroenterol Hepatol 2014; 12: 308-10 Associated editorial
- Gastroenterol 2014; 146: 581-82. Associated summary
Key points/Implications:
- Aggressive hydration may prevent post-ERCP pancreatitis. In the study, the treatment group received an average of 3290 mL over the 9-hour period compared with 945 mL in the standard infusion group.
- Implication: The speculation from the study and the editorials is that improved pancreatic perfusion will result in better oxygenation and reduce the likelihood of pancreatitis. In the 2nd reference, the author states that his practice is to administer “at least 3 L of crystalloid in recovery to young, healthy patients who have undergone high-risk ERCP and an additional 3 to 5 L within the first 12 hospital hours to those admitted with postprocedure pain”
- The best fluid (for post-ERCP and acute pancreatitis) may be lactated Ringer’s (LR).
- Implication: The lactate in LR may help reduce pancreatitis by avoiding acidosis which could promote zymogen activation and pancreatic inflammation. A previous small trial (n=40) of acute pancreatitits from any cause showed lesser degrees of systemic inflammatory response with LR in compared with normal saline (Clin Gastroenterol Hepatol 2011; 9: 710-17e1).
- This study adds aggressive IVFs as another intervention to prevent ERCP. Rectal indomethacin and prophylactic stent placement (in high-risk patients) are other accepted treatments.
Study details:
This pilot study randomly assigned 39 patients to aggressive hydration and 23 to standard hydration; all patients were inpatients who were not at risk for fluid overload. The aggressive group received 3 mL/kg/h during the procedure, a 20 mL/kg bolus after the procedure, and then continued on 3 mL/kg/hr for 8 hours. In contrast, the standard group received LR at 1.5 mL/kg/h during and for 8 hours afterwards.
Demographics: The average age was 43 years in the aggressive hydration group and 45 years in the standard group. 78% were hispanic. The ERCP procedures were mostly “average risk.” 74% had ERCP for choledocholithiasis. Only 2 subjects needed precut sphinterotomy (3%).
Results:
- No patients in the aggressive hydration group developed acute pancreatitis compared with 4 (17%) in the standard hydration group
- Elevated amylase (23% vs. 39%) and epigastric pain (8% vs 22%) were also less frequent in the aggressive hydration group.
Numerous Limitations: This was a small pilot study with an atypical population; thus, the findings are difficult to generalize. A false-positive (type 1 error) can easily occur due to the small numbers, especially as the standard hydration group had a rate of acute pancreatitis that was about double from previous studies. In addition, this study was not blinded and could have been susceptible to bias. Furthermore, the authors defined acute pancreatitis differently than in previous studies. In this study, the authors required enzyme increases 2 or 8 hours after ERCP with new abdominal pain; in previous studies, the definition of acute pancreatitis relied on enzyme increases for at least 24 hours after the ERCP.
Take-home message for those not doing ERCPs: Think about using lactated ringer’s and aggressive hydration in otherwise-well patients who present with acute pancreatitis.
Related blog entries: