- H Jaafari et al. Clin Gastroenterol Hepatol 2026; 24: 814-823. Open Access! Dietary Patterns Are Associated With Variations in the Global Prevalence and Severity of Rome IV Irritable Bowel Syndrome
- H Mikhael-Moussa et al. AJG 2026; 121: 461-471. Is Carbohydrate Intolerance Associated With Carbohydrate Malabsorption in Disorders of Gut-Brain Interaction? Thanks to Ben Gold for this article.
In the first article by Jaafari et al, the authors identified four predominant food cluster patterns from 54,127 participants from 26 countries who completed online questionnaires. The highest prevalence of IBS was associated with cluster A (including Egypt, Brazil, and Colombia) and the lowest with cluster D from several Asian countries. Cluster A dietary pattern was “rich in FODMAPs.” Cluster D participants reported the highest frequencies of fish, rice and tofu consumption and lowest milk consumption (which could be due to higher rates of lactose intolerance). The limitations from this study include the reliance on a questionnaire, the small number of foods surveyed (10), and cultural differences in reporting symptoms.
The study by Mikhael-Moussa et al examined carbohydrate malabsorption, based on breath testing, in patients diagnosed with disorders of gut-brain interaction (DGBI). Patients completed questionnaires as well.
Key findings:
- Among the 301 patients with DGBI included in our analysis, 178 (59.1%) had carbohydrate intolerance
- Carbohydrate-intolerant patients were significantly more likely to be female (P value < 0.001), to have 2 or more DGBI (P value = 0.001) to have lactose maldigestion (P value< 0.001) and fructose malabsorption (P value = 0.023), higher irritable bowel syndrome and somatic symptom severity, and lower quality of life (P value < 0.001) compared with patients without carbohydrate intolerance
Discussion:
- There are multiple potential reasons why certain foods can contribute to GI symptoms including malabsorption, gastrointestinal mobility, alterations in intestinal microbiota, intestinal distention, fluid accumulation, and localized IgE-dependent reactions (noted with confocal laser endomicroscopy).
- “Carbohydrate-reduced diets have been found effective in reducing symptoms in patients, regardless of the presence of maldigestion/malabsorption”
My take: Dietary triggers are important for the majority of patients with IBS. In this study, patients with documented carbohydrate malabsorption had increased severity of their symptoms.
Related blog posts:
- How FODMAPS Contribute to Irritable Bowel Symptoms
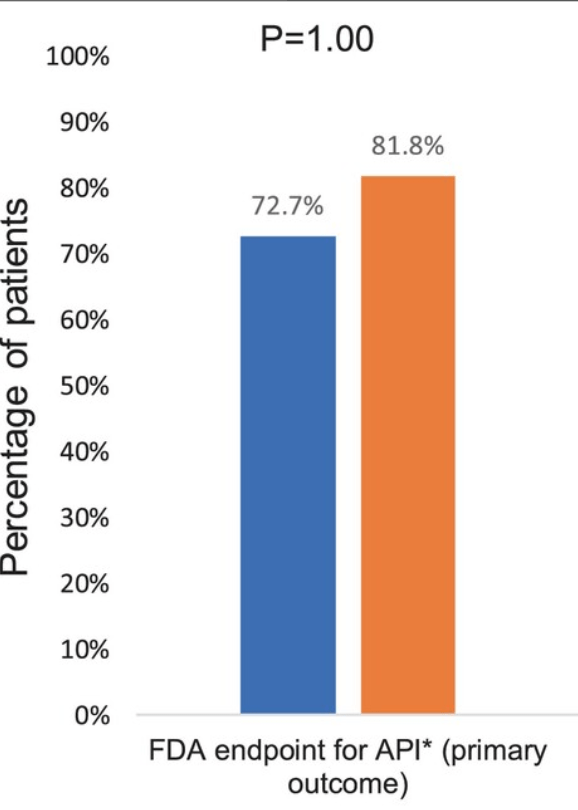
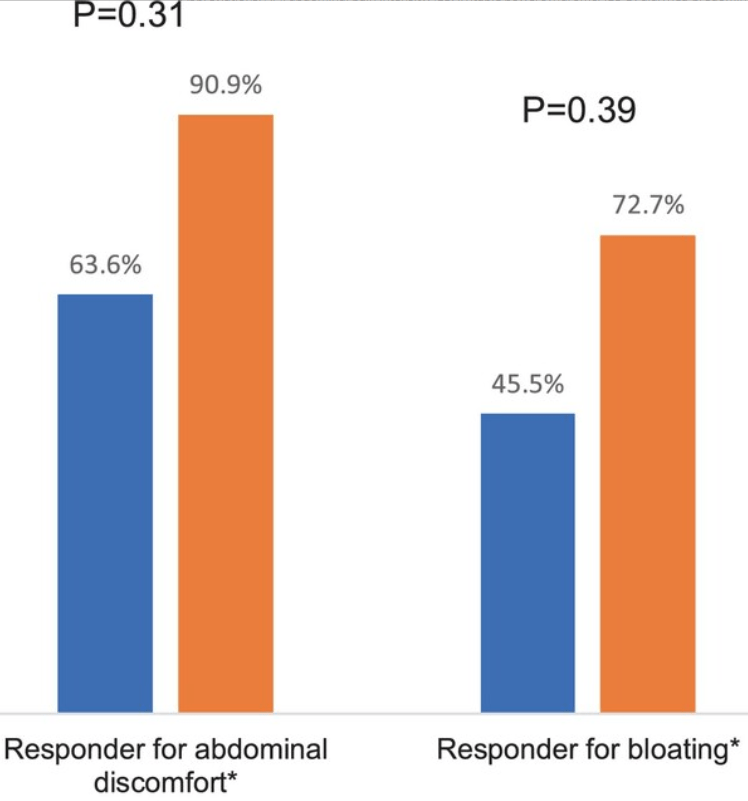
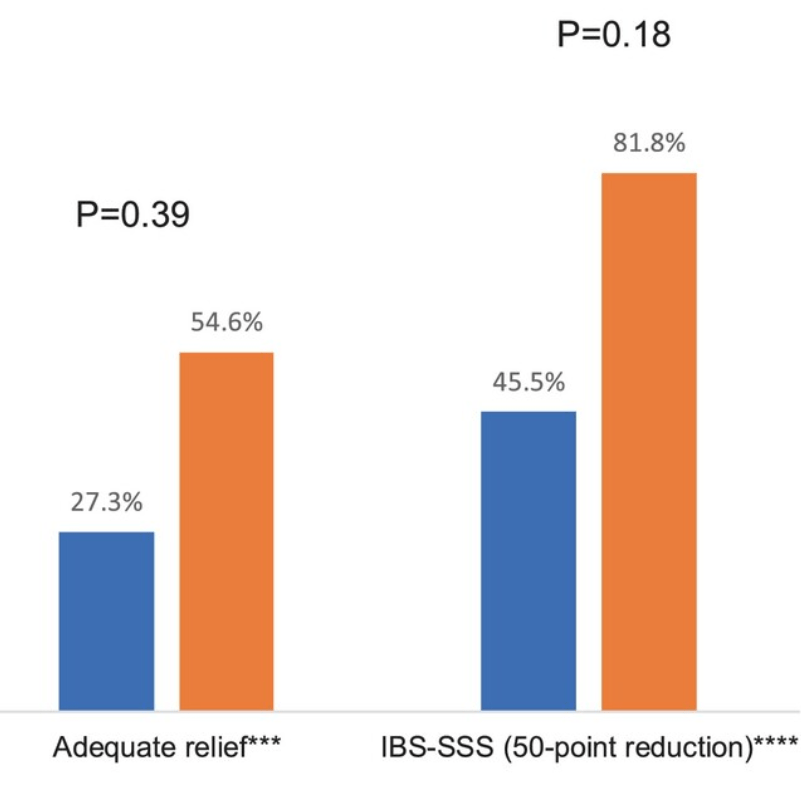
- Pilot Study: Mediterranean Diet vs Low FODMAP for Irritable Bowel Syndrome
- Mechanisms of irritable bowel syndrome
- Fructans and FODMAPs in Children with Irritable Bowel Syndrome
- Expert Advice on Bloating
- Which FODMAPs are Most Difficult to Reintroduce in Patients with Irritable Bowel Syndrome
- Low FODMAP –Real World Experience
- An Unexpected Twist for “Gluten Sensitivity”
- Tailoring IBS Dietary Therapy Based on Proprietary IgG-Based Blood Test –Does It Work?