R Rexwinkel, NK Vermeijeden et al. Gastroenterol 2025; 169: 94-103. Open Access! Mebeverine and the Influence of Labeling in Adolescents With Irritable Bowel Syndrome or Functional Abdominal Pain Not Otherwise Specified: A 2 × 2 Randomized, Placebo-Controlled Trial
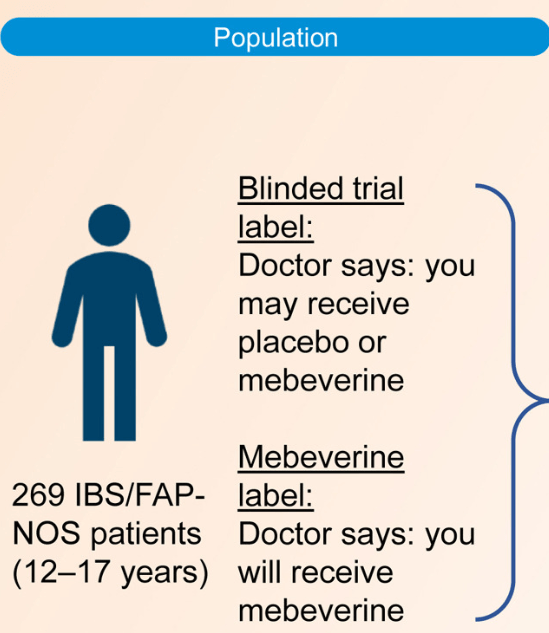
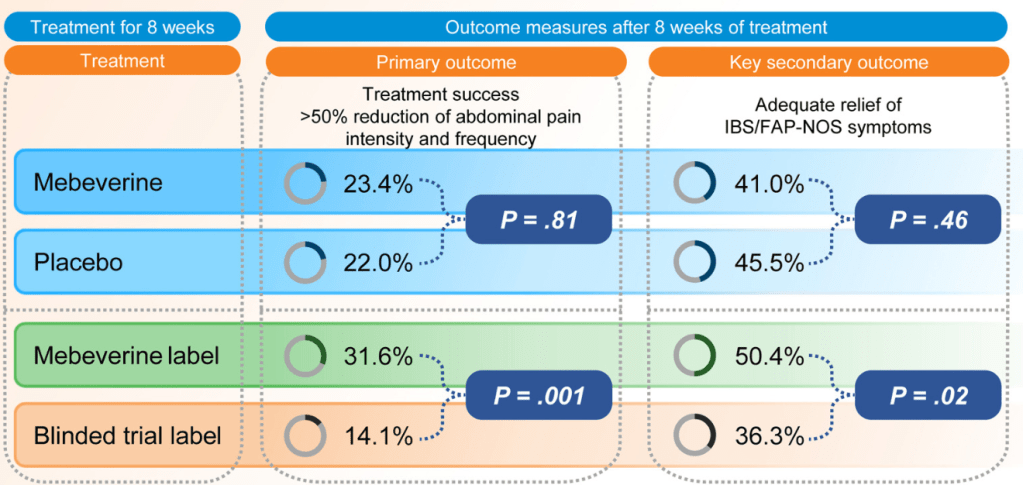
Methods: This was a randomized trial with 12-17 yr olds with irritable bowel syndrome or functional abdominal pain who received mebeverine (200 mg twice daily) or placebo for 8 weeks. Treatment was labeled as “mebeverine or placebo” (blinded trial label) or “mebeverine” (mebeverine label), creating the following 4 groups: (1) mebeverine-blinded trial label, (2) mebeverine-mebeverine label, (3) placebo-blinded trial label, and (4) placebo-mebeverine label. Randomization (1:1:1:1) was masked to physicians, except for drug labeling. Primary end point was treatment success (>50% reduction of abdominal pain intensity and frequency) after 8 weeks. The key secondary end point was adequate relief of symptoms.
Key findings:
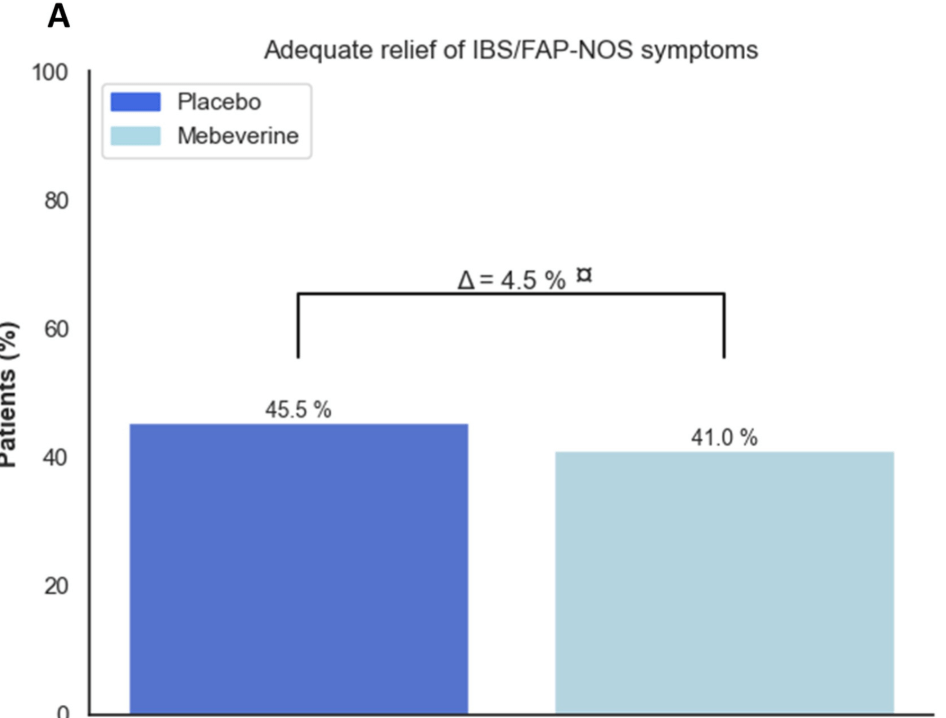
- Blinded Trial Label: Treatment success was similar between those receiving mebeverine (groups 1 and 2) (n = 31 [23.4%]) and placebo (groups 3 and 4) (n = 30 [22.0%]; odds ratio [OR], 1.08
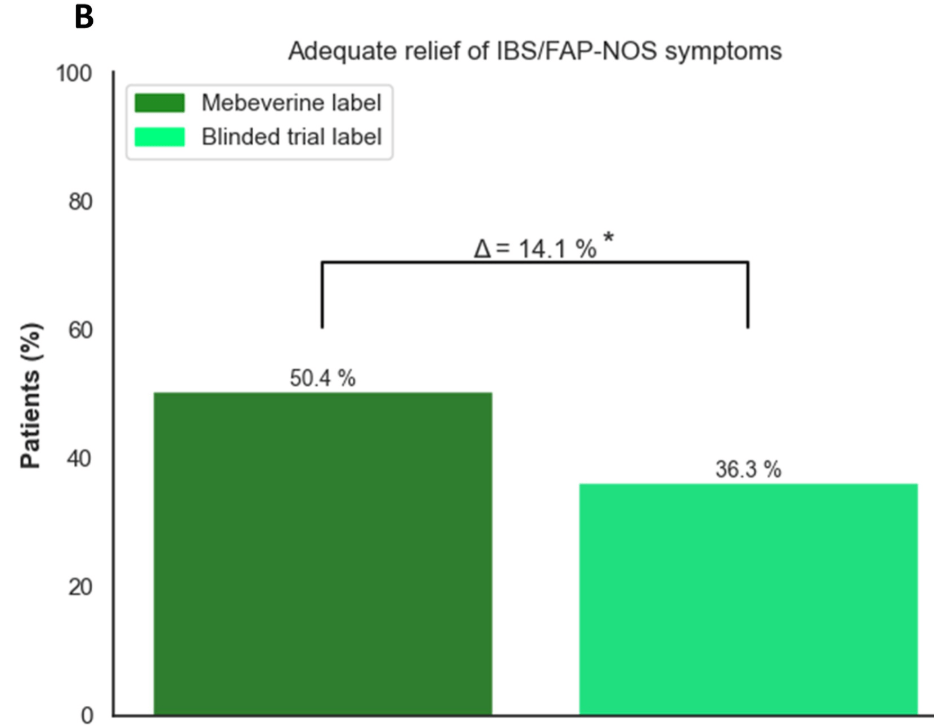
- Effect of Being Informed of Treatment with Active Medication: Treatment success was higher in groups with the mebeverine label (groups 2 and 4) (n = 42 [31.6%]) compared with the blinded trial label (groups 1 and 3) (n = 19 [14.1%]; OR, 2.84
Discussion Points:
- “This is the first study evaluating the impact of positive labeling on pain in a pediatric population. Our findings of a doubled treatment effect rate (32.9% vs 15.1%) when children were told they were receiving the active drug, are in line with adult studies showing higher pain reduction with positive labeling..14,19 It underscores the importance of positive expectations in pain management, which operates via multiple mechanisms.”
- “Results of previous research has shown that children with IBS can also report symptom relief when they know that they are receiving an inert compound, known as an ‘“’open-label placebo.’22“
- “Ethical norms state that ‘“’the use of a placebo without the patient’s knowledge may undermine trust, compromise patient–physician relationship, and result in medical harm to the patient.’26“
My take: This is a fascinating study showing how expectations for treatment can enhance the placebo effect. While the authors and the associated editorial mull over the ethical issues regarding deception of giving placebo without the family’s knowledge, in clinical practice many of the current drugs (eg. antispasmotics, neuromodulators, probiotics) have uncertain benefit and can be given without concern for deception.
Related blog posts:
- Good Study, Bad Practice: Placebo for IBS and Functional Abdominal Pain “It is a mistake to consider placebo as a treatment for functional abdominal pain. In many children, pain fluctuates and may improve with reassurance, distraction, healthier diets, and physical activity. However, we also need more effective therapies including pain psychology, dietary approaches and medications. The idea that placebo helps is misleading and undermines the fact that patients with functional disorders need effective treatment.”
- Treatment Guidelines for Pediatric Irritable Bowel Syndrome
- Dr. Carlo DiLorenzo: Advice for Managing DGBIs (Part 1)
- Dr. Carlo DiLorenzo: Advice for Managing DGBIs (Part 2)
- Pilot Study: Mediterranean Diet vs Low FODMAP for Irritable Bowel Syndrome
- AGA Guidelines for Pharmacologic Therapy of IBS-D and IBS-C (2022)
- ACG IBS Clinical Guidelines (2022)
- Does a Less Restrictive Low FODMAP Diet Work?
- “How to Approach a Patient with Difficult-to-Treat IBS”
- Gluten-Free Diet –Role in IBS?
- Dr. Neha Santucci: Management of DGBIs in the Post-Pandemic Era (Part 1)
