MT Fioretti et al. J Pediatr Gastroenterol Nutr. 2025;80:300–307. A decade of real-world clinical experience with 8-week azithromycin–metronidazole combined therapy in pediatric Crohn’s disease
Methods: This retrospective study over 10 years examined the efficacy of azithromycin-metronidazole for induction treatment in 44 children. All patients were given metronidazole (15–20 mg/kg/day two times daily, maximum of 1000 mg/day) administered daily for 8 weeks and azithromycin (7.5 mg/kg to a maximum of 500 mg/once a day) administered 5 days per week for the first 4 weeks, followed by 3 days per week for the final 4 weeks as per the initial publications.17, 18
Key findings:
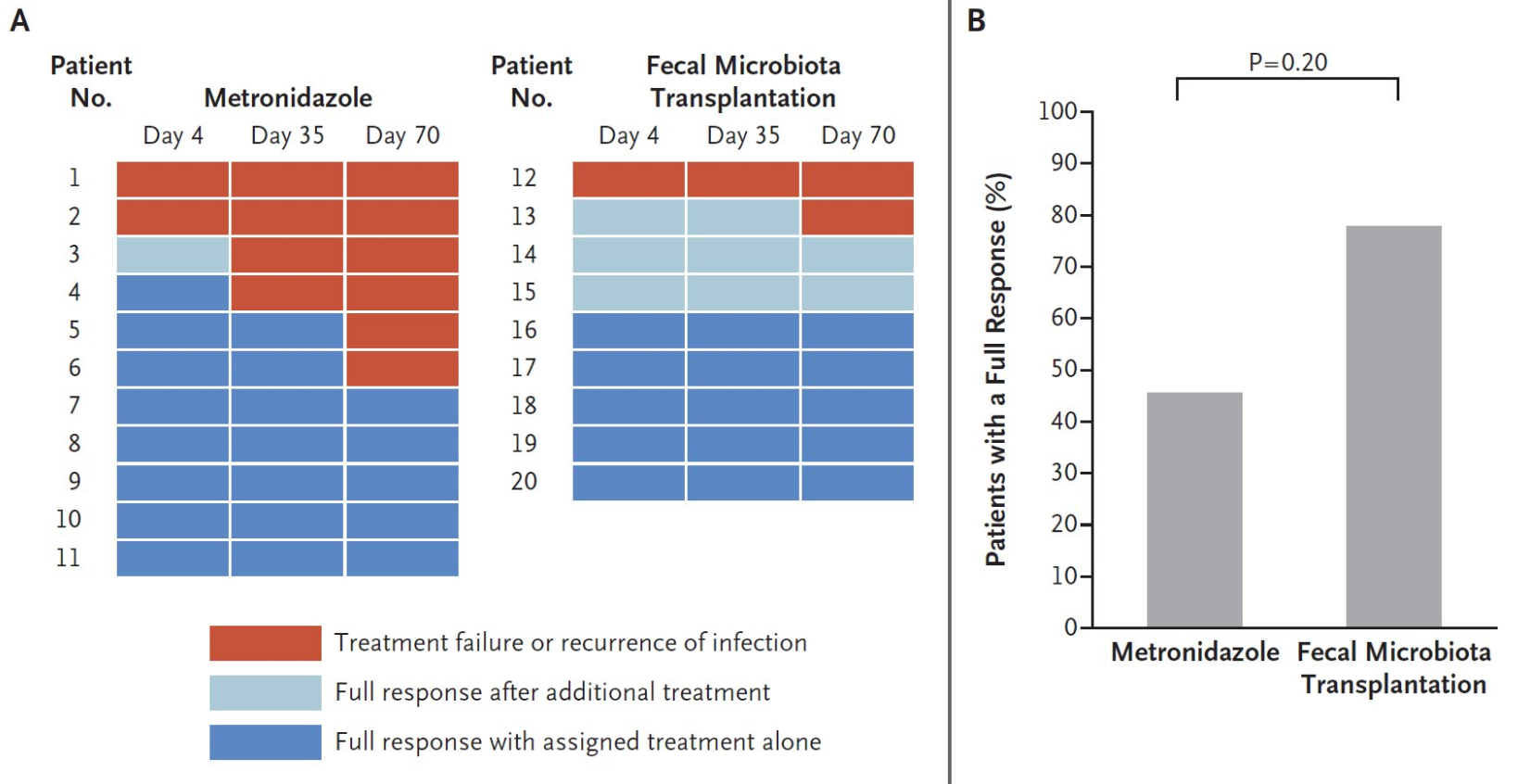
- After 8 weeks, the overall remission rate was 64%.
- Of the 38 patients who completed the CD AZCRO course, 28 patients (74%) entered remission (Group 1) and 10 (26%) did not (Group 2)
- After 8 weeks, Group 1 showed improved CRP levels and higher albumin and hemoglobin levels than Group 2. Median FC declined significantly from 650 mcg/g at baseline to 190 mcg/g at Week 8 in Group 1 (p < 0.001).
The authors conclude that “a combination treatment of azithromycin and metronidazole represents an alternative induction therapy for mild to moderate pediatric CD, offering benefits in terms of cost and practicalities compared to EEN and in side effects compared to steroids.”
My take: There are a small number of children with mild Crohn’s disease who could benefit from this induction regimen. An alternative would be the use of a more modest dietary approach (eg. Mediterranean diet)
Related blog posts:
- Mediterranean Diet vs Specific Carbohydrate Diet for Crohn’s Disease
- What is Mild Crohn’s Disease and How to Treat It
- Mesalamine in Pediatric Crohn’s Disease is Still Not Effective
- AGA Guidance: Nutritional Therapies for Inflammatory Bowel Disease
- What’s in Your Gut and How to Change It
- Oral Antibiotics For Refractory Inflammatory Bowel Disease
- A Little More Data on Antibiotic Cocktail for Pediatric Acute Severe Ulcerative Colitis

Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.