K Puri et al. J Pediatr Gastroenterol Nutr. 2025;81:1–4. Nutritional and growth concerns of vegetarian diets in children
Key points:
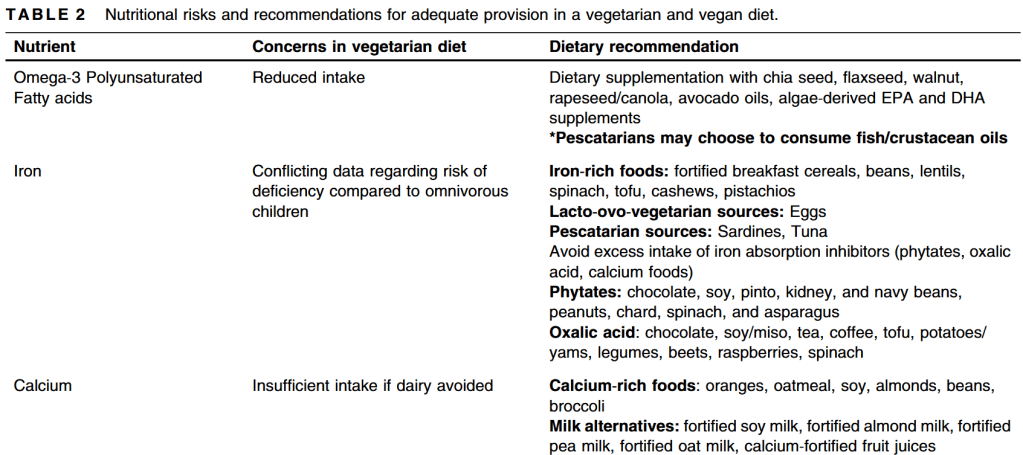
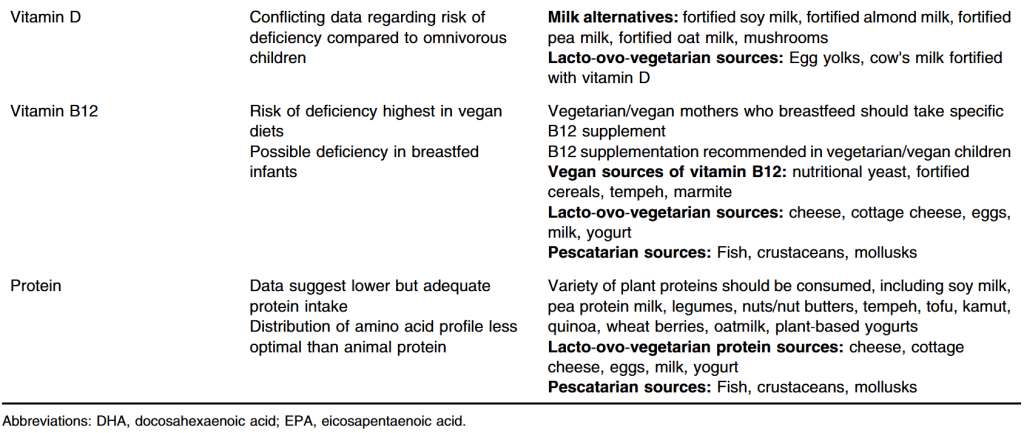
- “Although vegetarian diets have become popular due to their benefits on environment, they pose nutritional challenges due to the risk of developing nutrient or caloric deficiencies which may impact health or growth in children and adolescents.”
- “Any child or family who adheres to a strict vegetarian diet [needs to] work with a nutrition specialist or registered dietitian familiar with the nutritional concerns of a vegetarian diet. This includes calcium and iron intake as well as sufficient and diverse protein intake. B12 and vitamin D supplementation should be considered in vegetarian children”
- “While data suggest that average anthropometrics are similar between vegetarian/vegan and omnivorous children, there is some concern regarding the risk of being underweight while following a vegetarian diet…. a vegetarian diet can be a safe and complete diet for children when detailed and longitudinal attention is given to diversification of nutrient intake.”
My take: This article provides a useful review of the dietary issues that need to be monitored for pediatric vegetarians. I agree that a visit with a nutritionist is worthwhile for these children.
Related blog posts: