While proton pump inhibitors (PPIs) are used extensively for acid-related diseases and have been around for nearly 25 years, there have been a number of reports about potential side effects. As a drug class, PPIs have a very good safety profile. A recent article reviews some controversial adverse effects and summarizes the evidence for and against (Clin Gastroenterol Hepatol 2013; 11: 458-64).
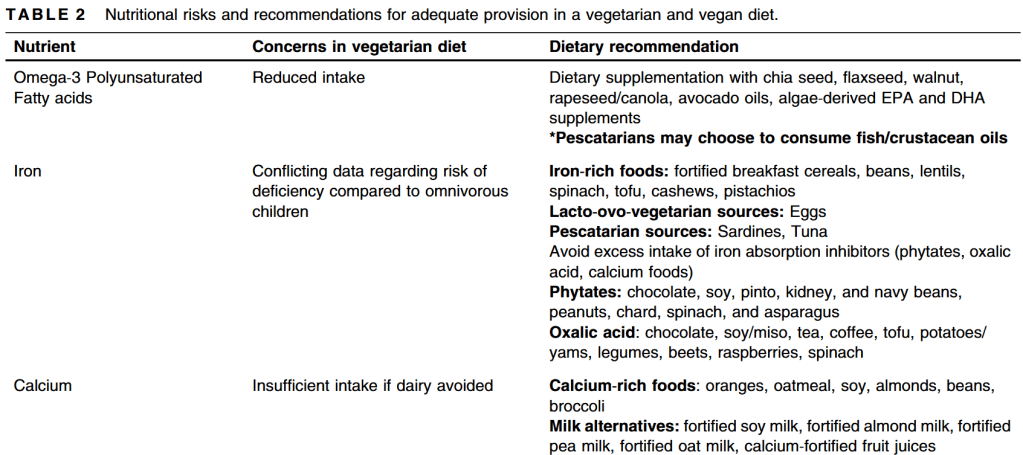
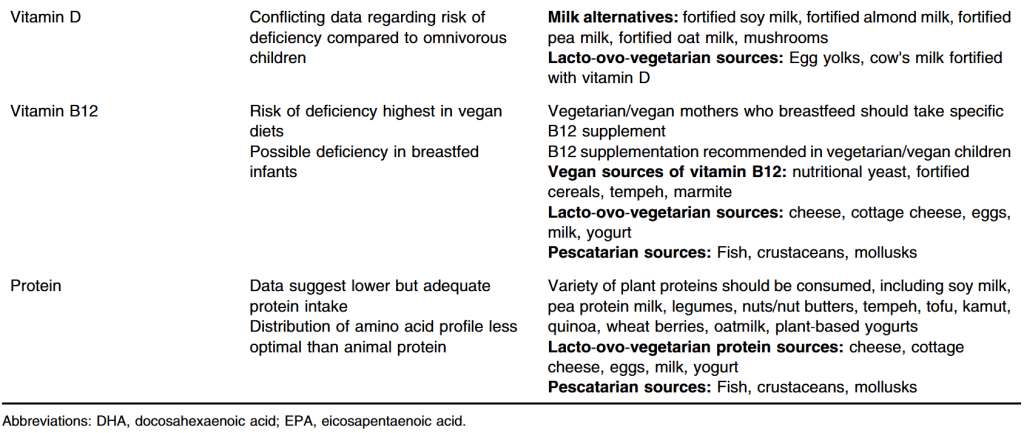
I. Calcium/bone effects. After reviewing a number of studies, the authors conclude: “There is no good evidence to establish that PPI use has a significant risk for bone density loss or osteroporotic-related fractures….Supplemental calcium is not recommended or justified solely because of PPI use.”
II. Iron. “Although it is conceivable that PPI therapy may reduce absorption of nonheme iron and retard iron pool replenishment, this effect has not been well-studied or evident from widespread use in clinical practice.
III. Magnesium. “The FDA recommendation to consider checking magnesium levels before starting is not practical, in particular for the over-the-counter market. In patients who may be predisposed to …ongoing magnesium loss…it may be reasonable to follow…Given the rarity of the reports and no controlled studies to delineate the mechanisms, it is important for health care providers to be aware of this” (rare reports of profound hypomagnesemia).
IV. Pneumonia. “Small relative risk associated with short-term and high-dose PPI use. These relationships, however, do not offer a definitive explanation for the relative risk” due to the studies and confounding factors.
V. Clostridium difficile. “To date, there is insufficient evidence to conclude that there is a definitive relationship between PPI use and C difficile infection…clinicians should be aware of this potential relationship.”
VI. Traveler’s diarrhea. “The data…were overall supportive of no associated risk, albeit there were a few specific case reports suggesting a remote causal association.”
VII. Small intestinal bacterial overgrowth. “The relationship between PPI use and the development of SIBO is still not understood.”
VIII. Interstitial nephritis. Extremely rare. “Investigators…did not find enough evidence to support a causative relationship.”
IX. Methotrexate. “Coadministration of PPIs with high-dose methotrexate appears to be correlated with delayed methotrexate elimination.”
Also discussed: Vitamin B12, Clopidogrel, Spontaneous bacterial peritonitis
The authors conclude that the above reported associations have received considerable attention. “Because PPIs are overprescribed in many patients, …the clinical effects always should be reviewed and attempts should be justified to stop any therapy that may not be needed.”
Related blog entries: