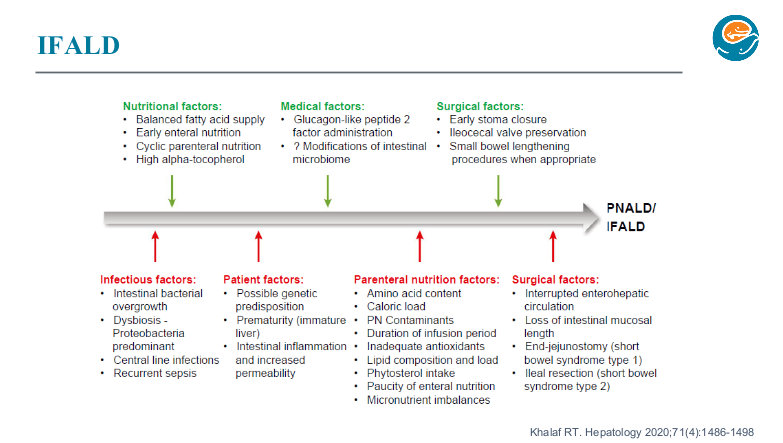
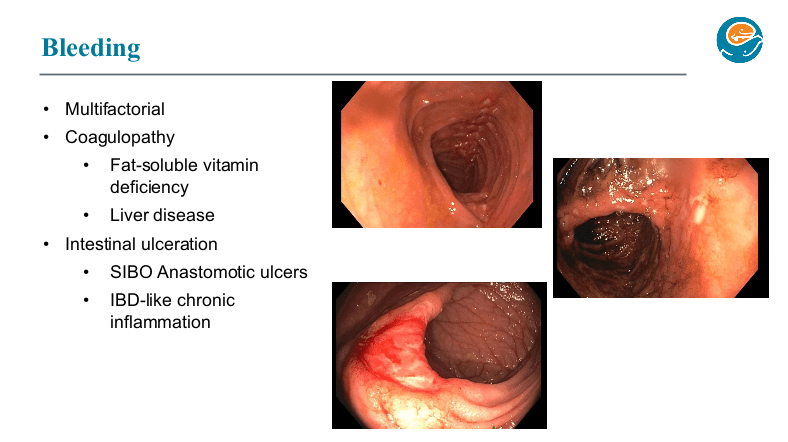
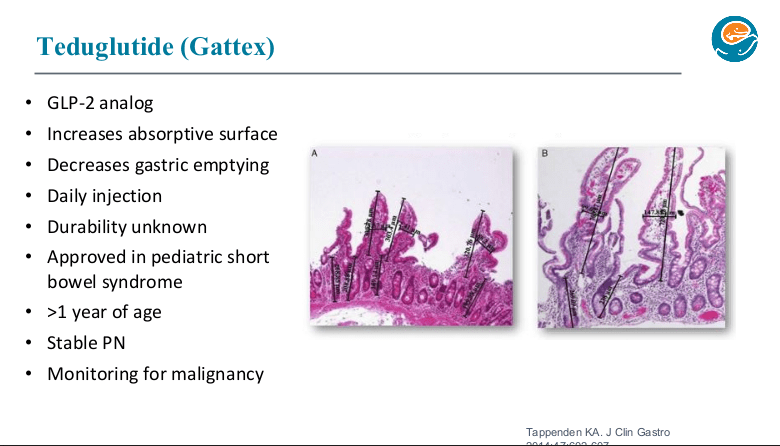
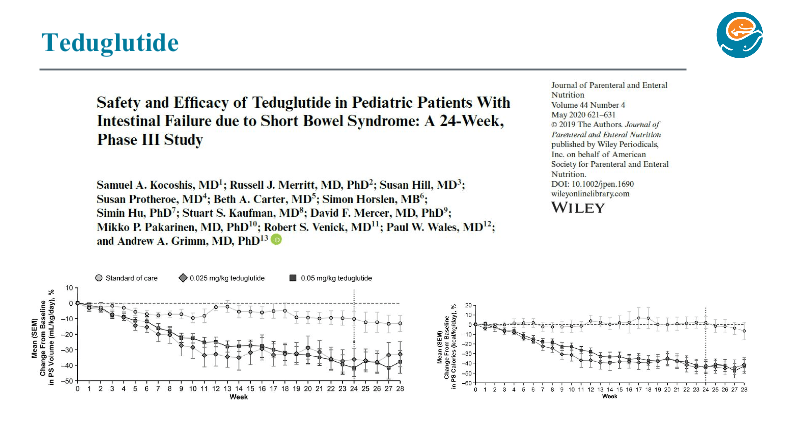
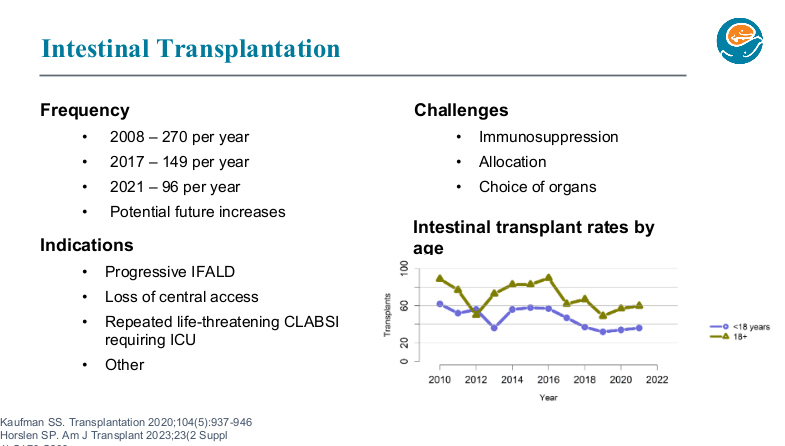
Two years ago, Dr. Wendel gave our group a great lecture on short bowel syndrome (SVS). One of the neonatologists in attendance invited her back to provide a state of the art update. While this 2026 lecture covered some of the same issues, there were important updates and insights.
My notes below may contain errors in transcription and in omission. In addition, the information provided is based on what is done in Seattle. However, there is not a lot of evidence for much of what is done in intestinal rehabilitation. Thus, there is variation in practice at different centers and what works for one patient might not work for another. Following my notes, I have included many of her slides.

Diet:
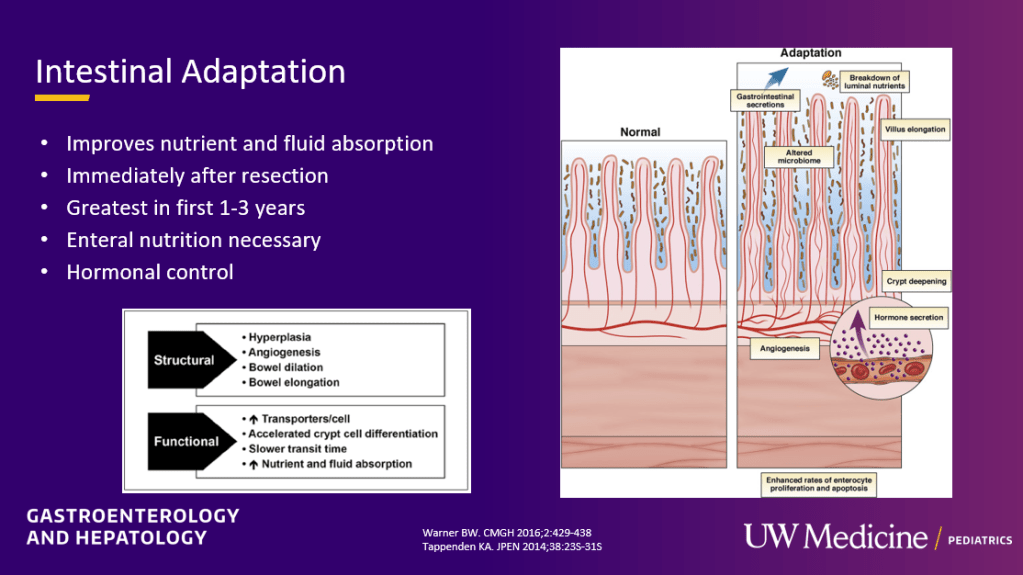
- Enteral feedings promote intestinal adaptation. Pediatric patients with SBS require much higher calories with enteral nutrition and may have hyperphagia as a compensatory mechanism
- Breastmilk and/or Standard formula likely help promote intestinal adaptation better than hydrolysates and elemental formulas. In addition, it may help reduce the development of food allergies which are increased in children with SBS
- Oral feedings have many advantages over NG or GT feedings when feasible. The ability to consume solid foods is quite helpful in reducing diarrhea. Also, encouraging oral feedings may help reduce feeding aversions. As such, GT placement is avoided if possible in Seattle
- Key diet advice: avoid sweet tasting food/drink, especially in the first few years of life while they are developing their palate/food preferences
- Feed osmolarity/caloric density: Most children with SBS tolerate lower caloric density (15-20 cal/oz) and more volume orally rather than higher caloric density/lower volume feeds

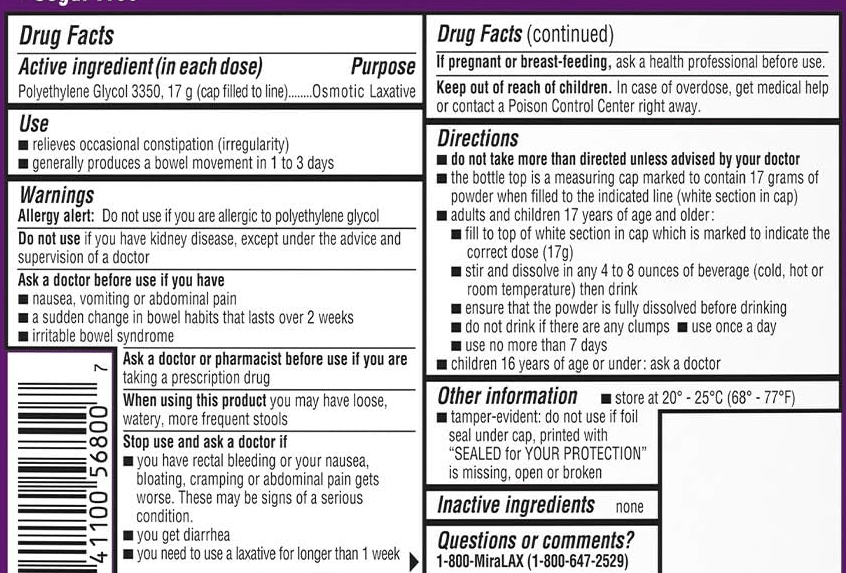
Parenteral Nutrition:
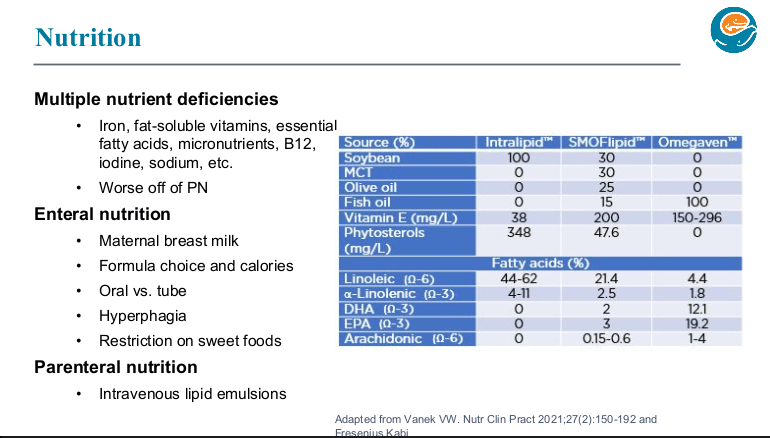
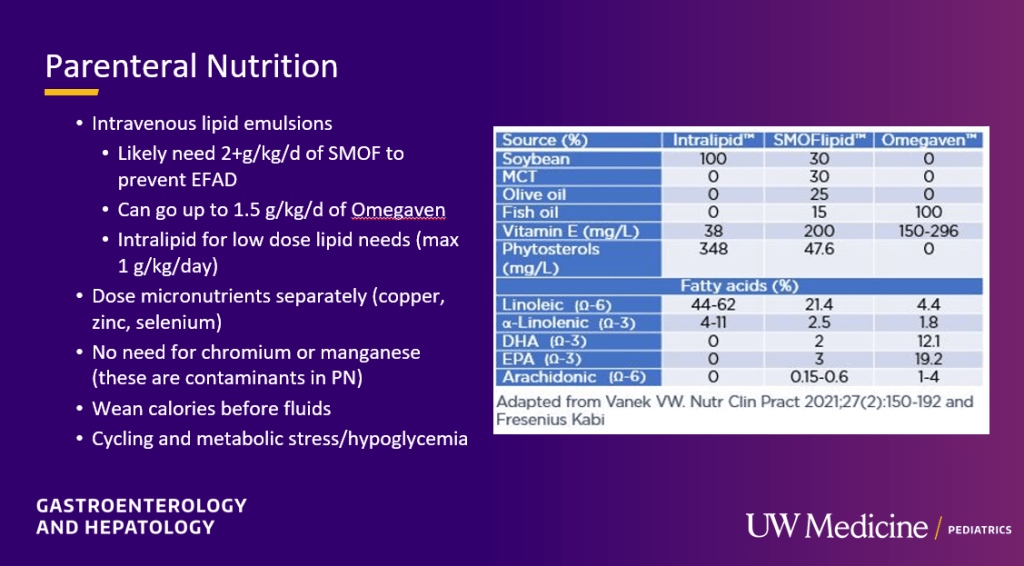
- Lipid emulsions: SMOFlipid at 2 gm/day can help prevent essential fatty acid deficiency (EFA). Omegaven may need to be dosed at 1.5 gm/day to prevent EFA. If used for short-term and low dose, standard intralipid can be useful
- HAL (aka TPN): Typically weaning calories is done before weaning volume. Cycling HAL (delivering over fewer hours) can be started prior to discharge. Watch for tolerance of the glucose infusion rate (JH: I prefer the terms HAL = hyperalimentation or PN=parenteral nutrition. TPN =total parenteral nutrition. Most patients are receiving parenteral nutrition but not total parenteral nutrition.)
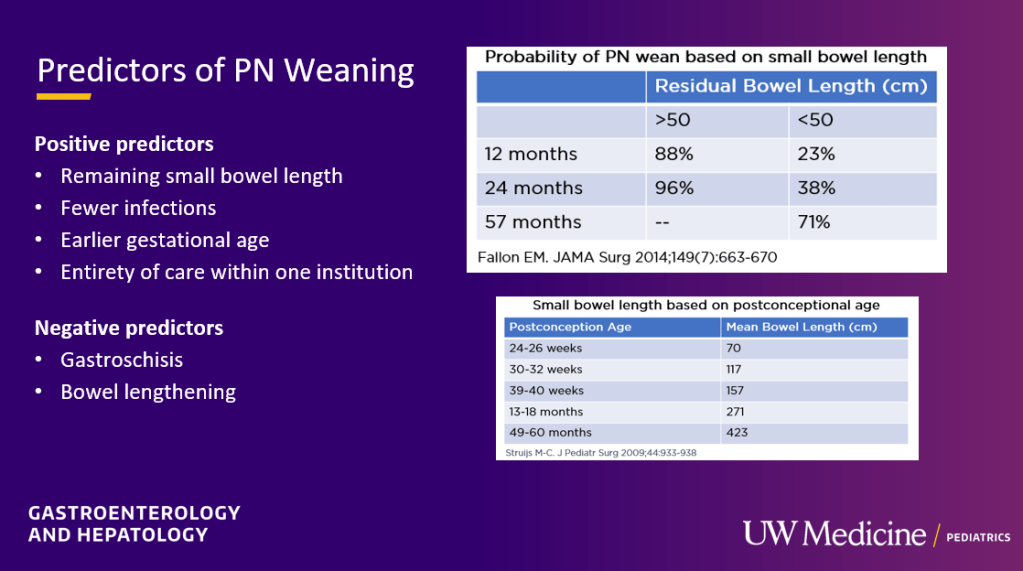
Ostomy/Stool Output:
- Output goals: Most pediatric patients can tolerate output of 50 mL/kg/day of ostomy output (if being supported by PN), though less than 30 mL/kg/day is more physiologic
- Iron: Parenteral iron is typically needed. Seattle team prefers ferric carboxymaltose as it may deliver enough iron for 6-12 months in one infusion
- Acid suppression: While acid suppression can sometimes be beneficial by lowering gastric output, if possible avoid long-term use as it may increase risk of bacterial overgrowth along with other infections
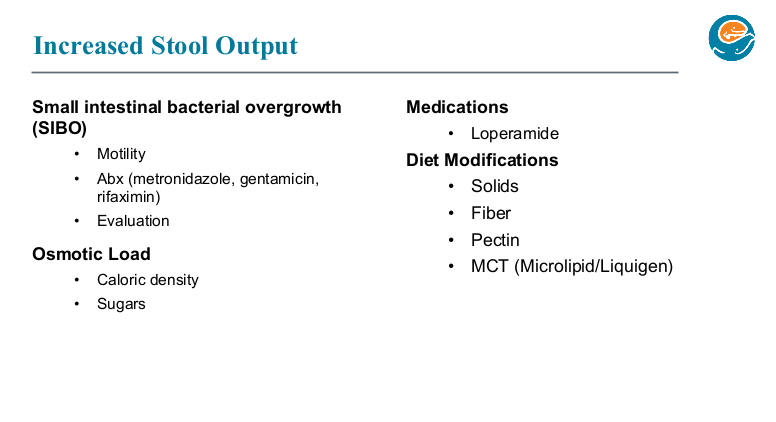
- Excessive stool output (via stoma or per rectum) is when it is more than the patient’s baseline. This should prompt investigation for potential causes including diet/osmotic agent, bacterial overgrowth and infections
- Pancreatic enzymes: It is unclear if pancreatic enzymes (PERT, Relizorb) will improve stool output due to lack of data
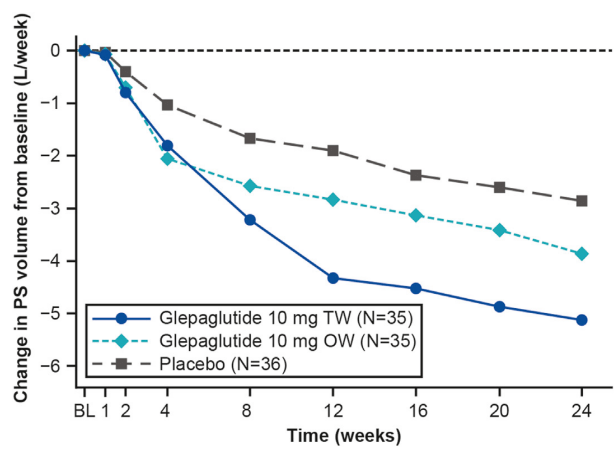
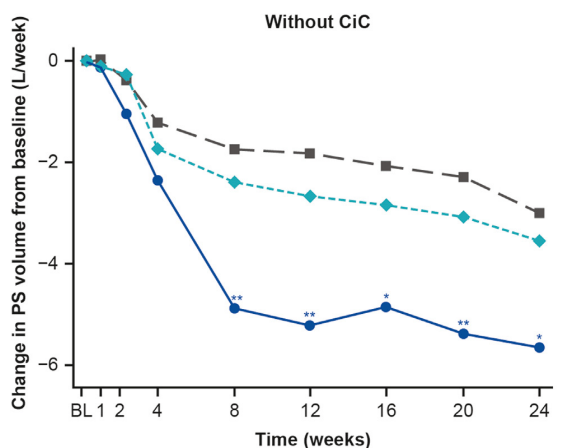
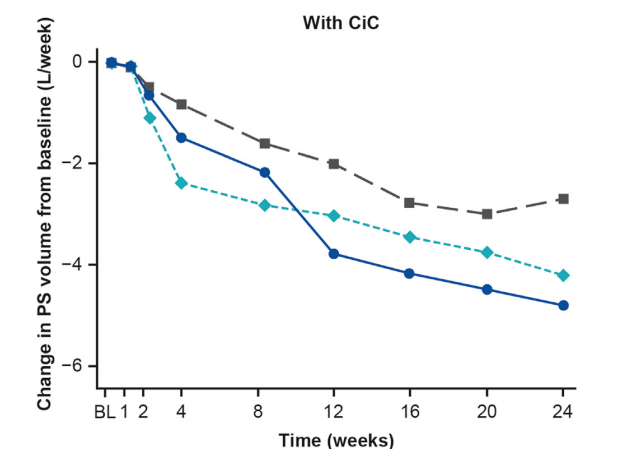
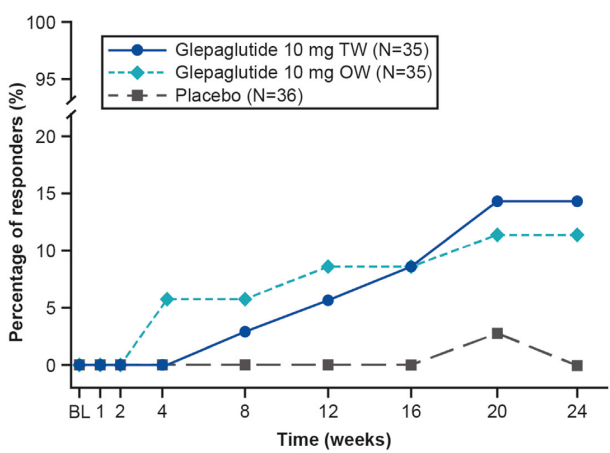
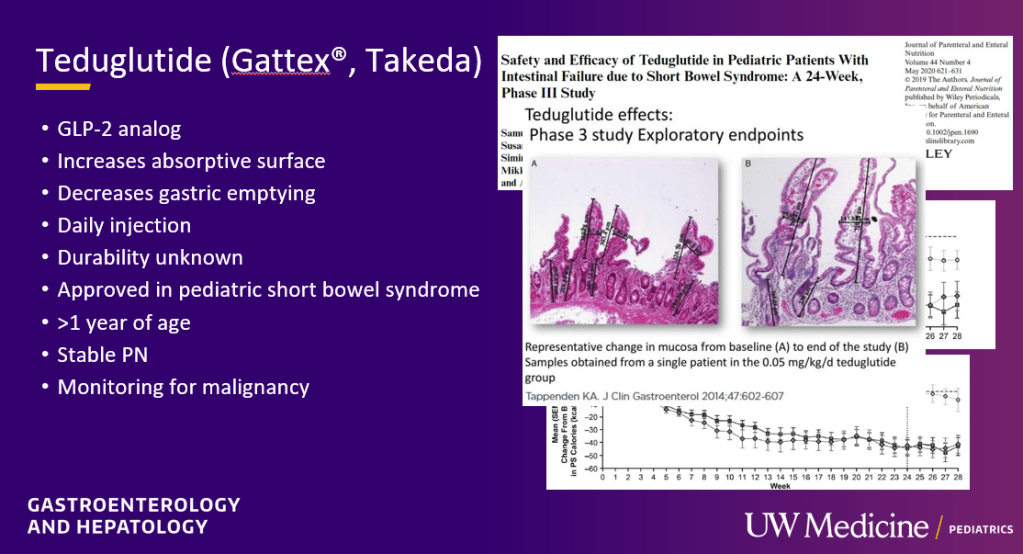
- Teduglutide can reduce the need for HAL. It is a hormone (like insulin) and sustained effects are generally not seen when it is stopped. However, especially in patients close to coming off HAL, it may be beneficial


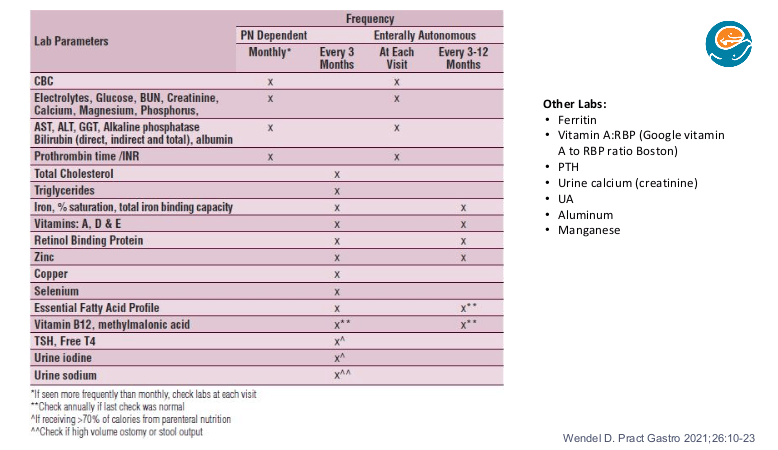
Monitoring:
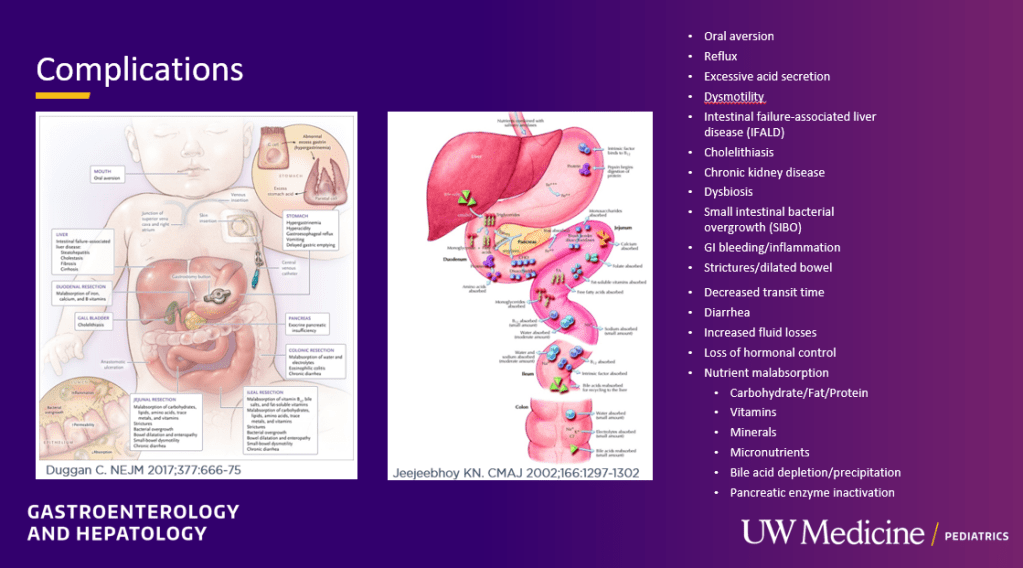
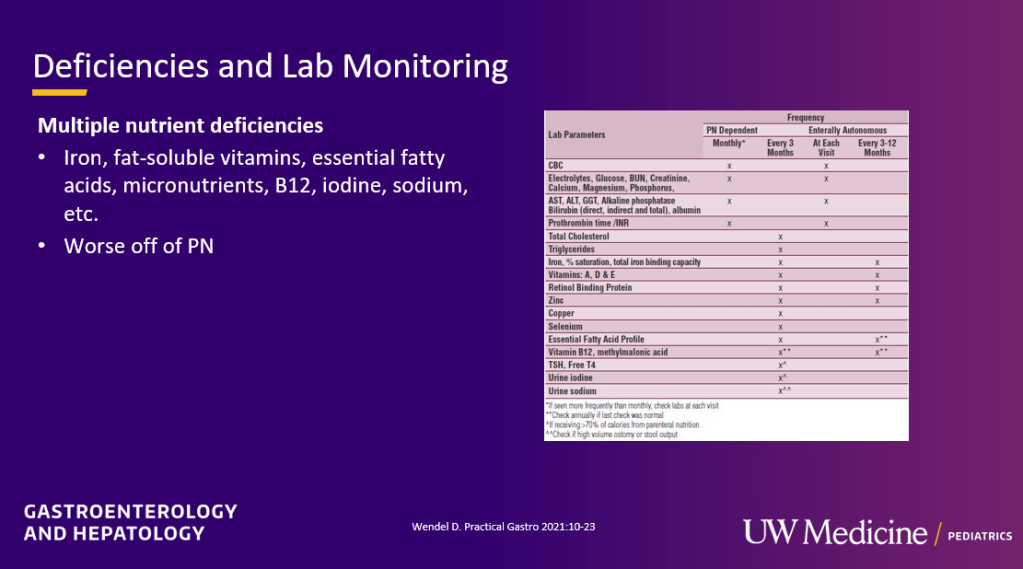
- Nutrient deficiencies: Close monitoring for nutrient deficiencies is needed and often even more important when no longer receiving HAL
- Urine sodium more than 30 is a goal. Sodium depletion interferes with growth and can contribute to other electrolye disturbances (eg. hypokalemia)
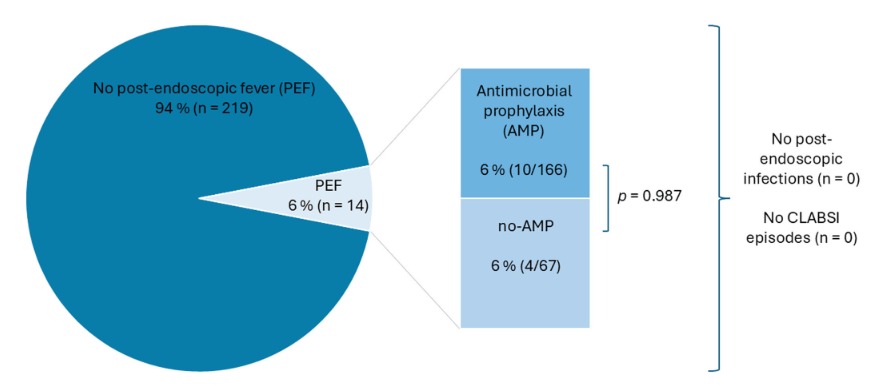
CLABSI:
- Antibiotics: Treatment starts with a broad-spectrum antibiotic and wait to add specific gram-positive coverage unless ill-appearing or gram-positive organism starts growing. Vancomycin is not used frequently in Seattle due to concerns of renal toxicity. In patients with gram-positive infection, linezolid is often used
- Minimum of 48 Hours For All Fevers: Everyone with SBS and with fever (greater than or equal to 100.4) stays for at least 48 hrs on broad spectrum IV antibiotics
- Locks: Sodium bicarb locks help prevent CLABSI and appear to have similar infection prevention as ethanol locks. Ethanol locks have been difficult to get coverage.
SIBO:
- Medications: Metronidazole is generally 1st line agent and gentamicin (IV formulation given enterally) is a 2nd line agent in Seattle. Rifaximin would be potentially their 1st line agent if it were easier to get covered
Related blog posts:
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.