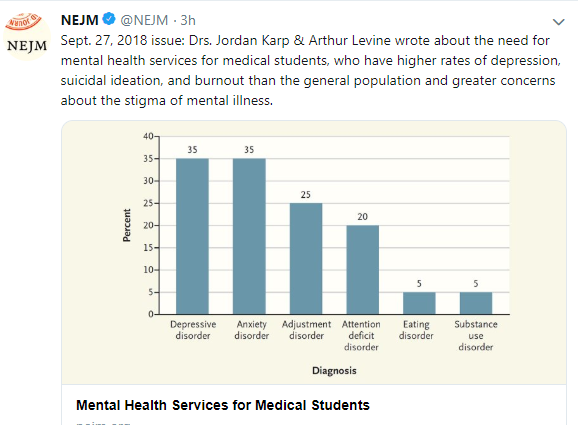
We had an brilliant lecture given to our group by Danielle Wendel who leads Seattle Children’s Intestinal Rehabilitation team. My notes below may contain errors in transcription and in omission. In addition, the information provided is based on what is done in Seattle. However, there is not a lot of evidence for much of what is done in intestinal rehabilitation. Thus, there is variation in practice at different centers and what works for one patient might not work for another. Following my notes, I have included many of her slides.
Key points:
- Enteral nutrition is key for adaptation. At Seattle, oral feeds rather than GT feeds are preferred.
- Time is the thing that helps the most. Unclear which additives help.
- Though prediction models often look at >50 cm, it is important to counsel families, even with more bowel length, that the care is going to be quite challenging.
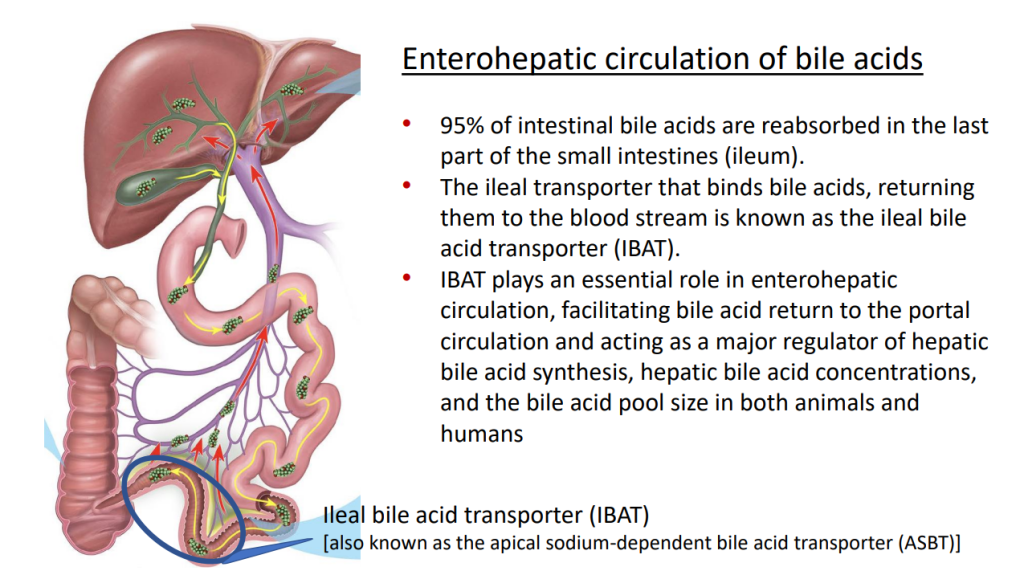
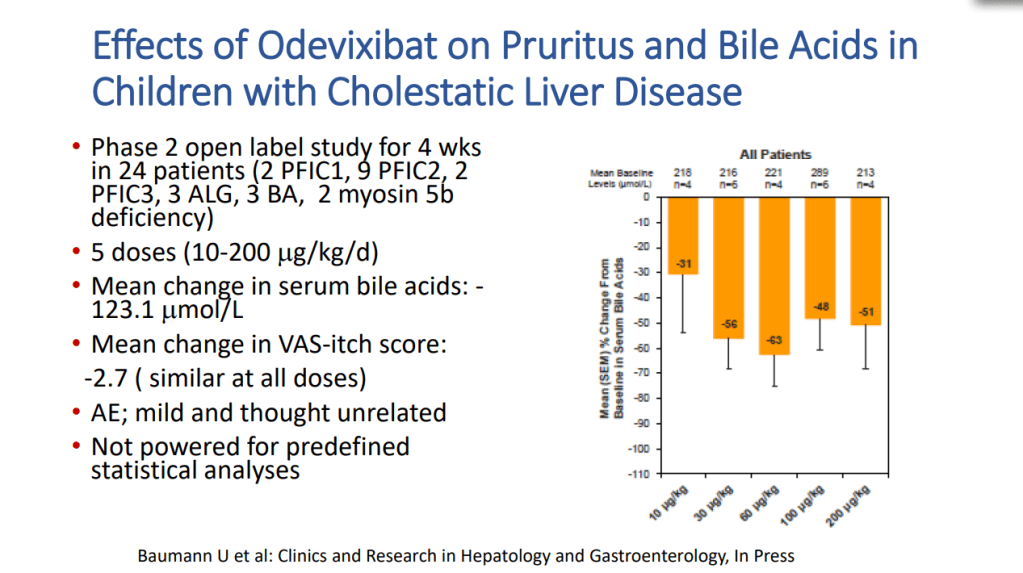
- Acid suppression can be helpful in the early phase after resection especially if problematic high stoma output.
- Iron: if given enterally, typically given everyday or every other day. Iron dextran can be given in TPN (cannot be given with lipids). Ferric carboxymaltose is a good choice for parenteral administration. Due to the need for few infusions (~1-2 per year), it is safer (less line entry) and cost-effective compared to iron sucrose.
- Seattle shares an “Emergency Care Letter” with their patients (template available in EPIC)
- Yearly doppler ultrasound is recommended to assess vascular access.
- If a patient has more than one thrombosis, the Seattle team recommends long term prophylaxis, though it might take treatment levels to prevent further thrombosis.
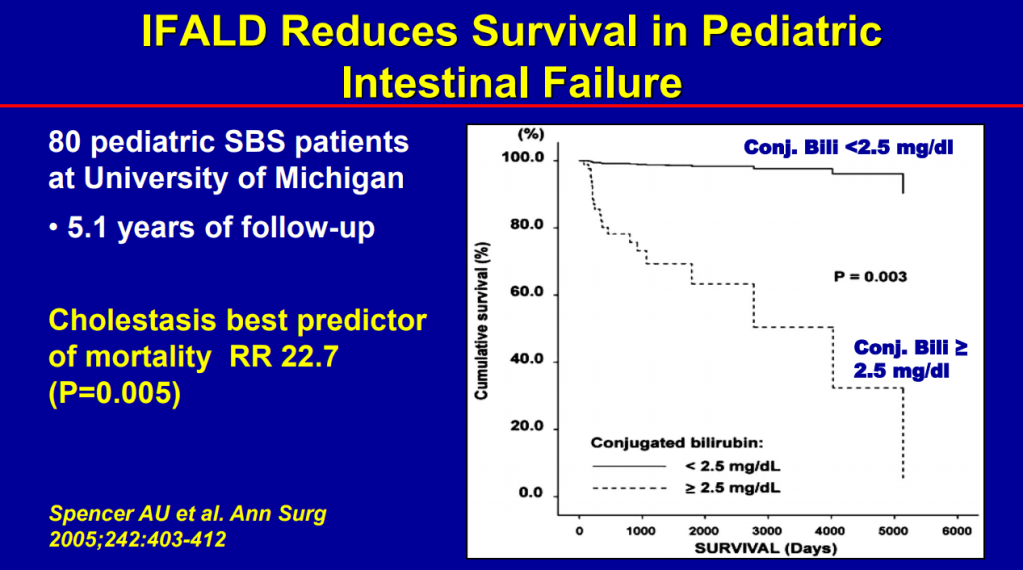
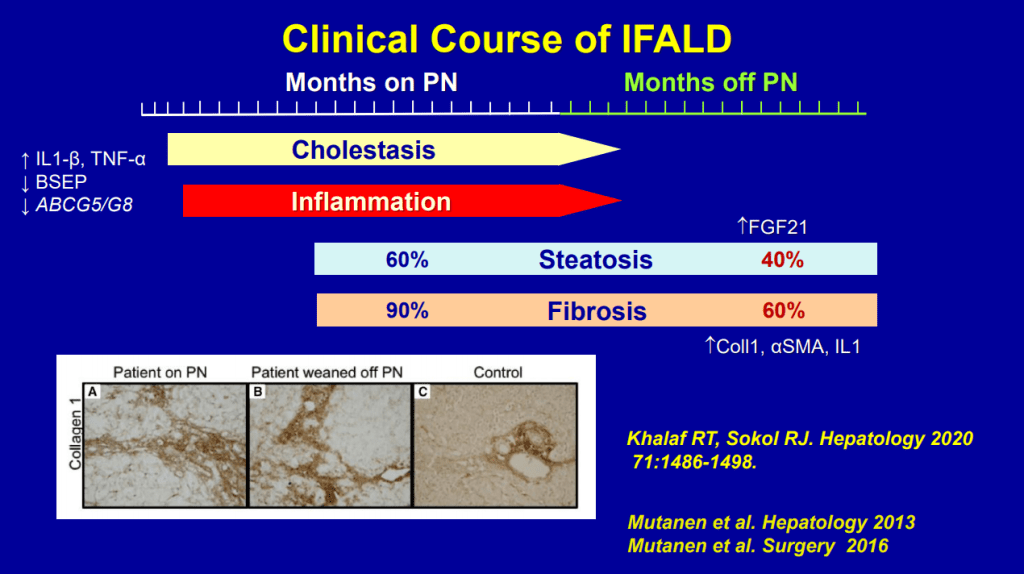
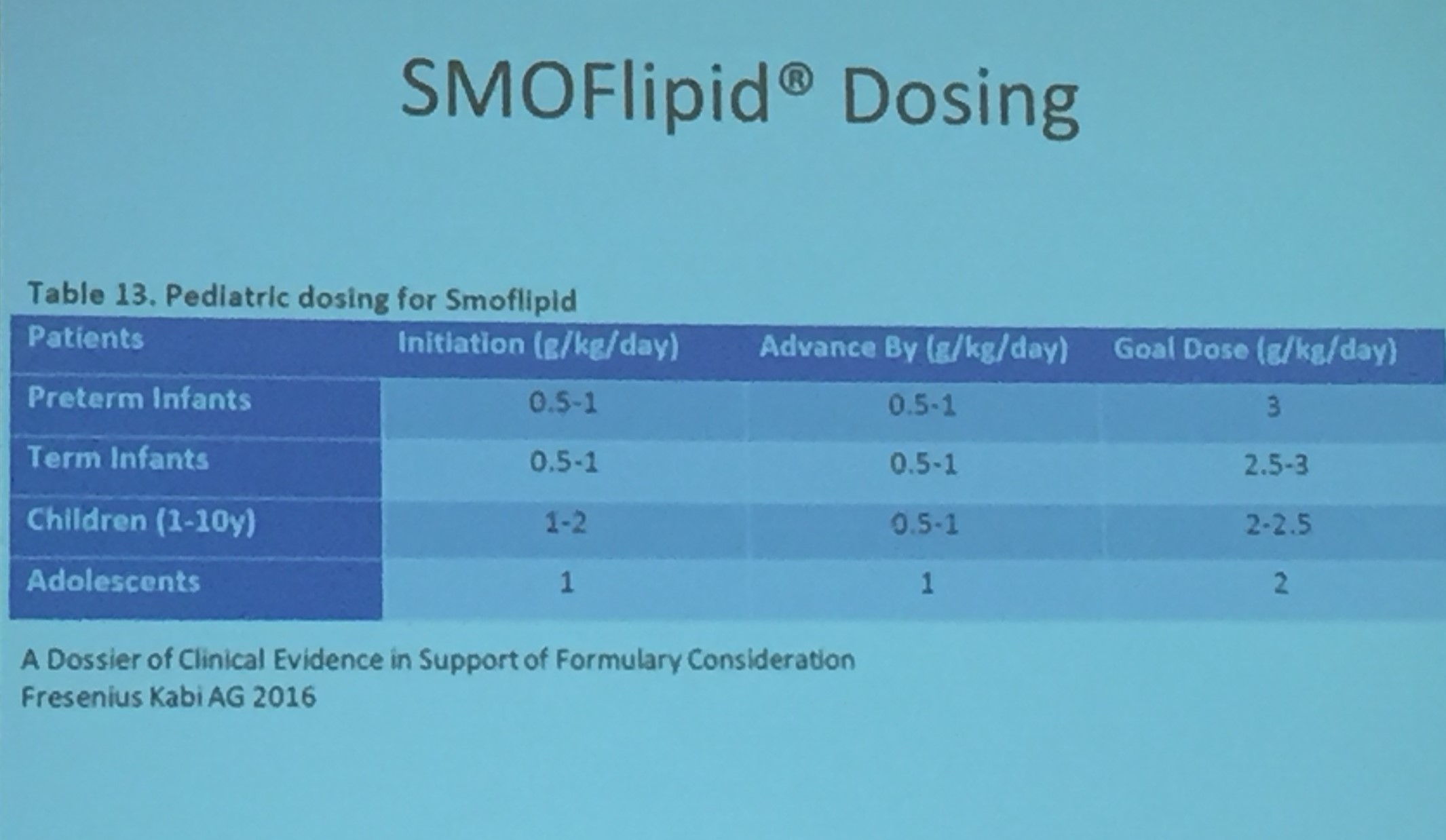
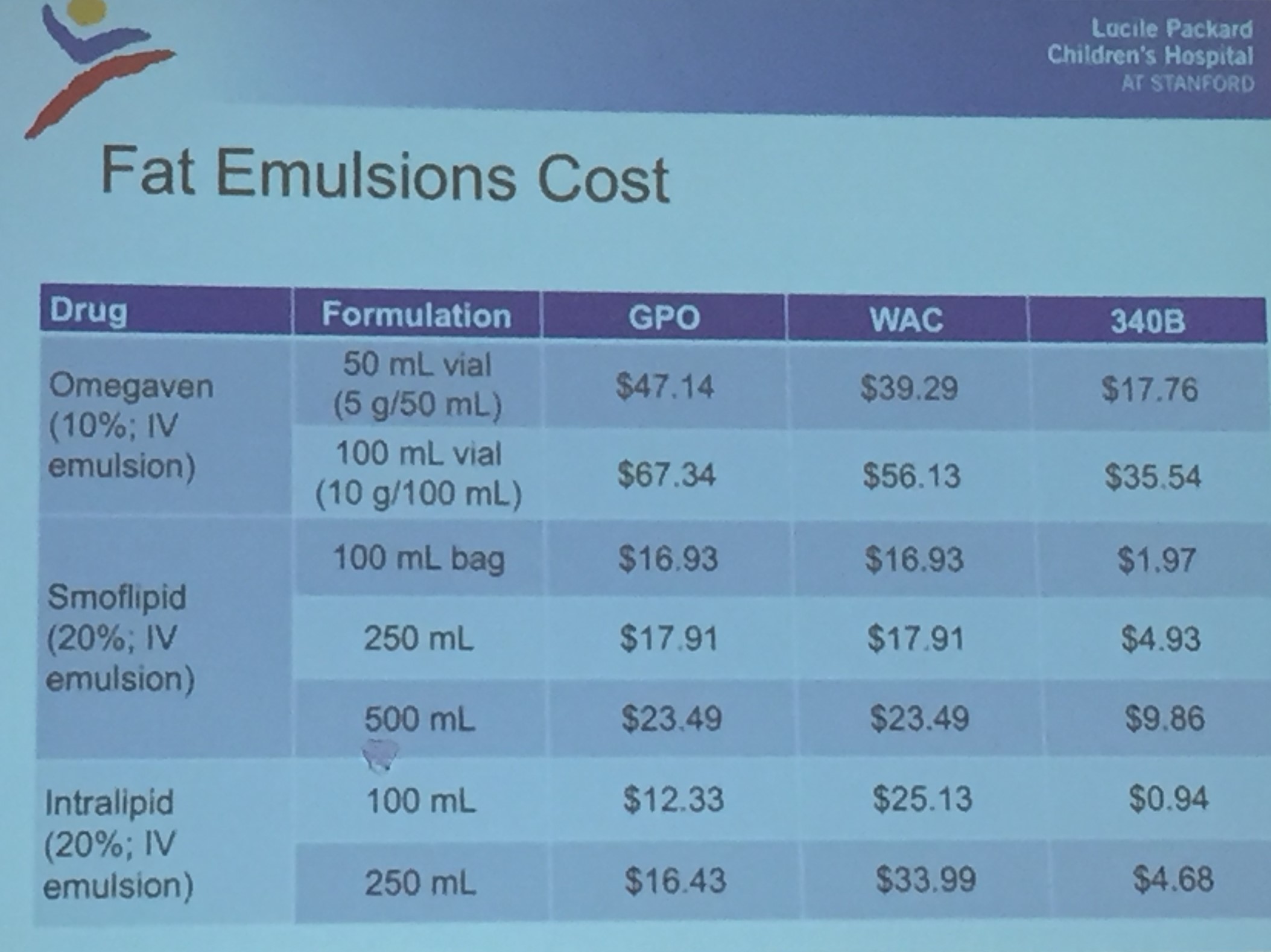
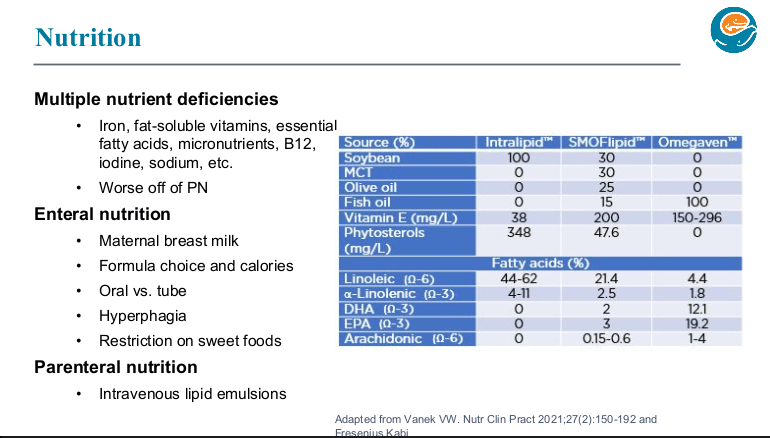
- SMOFlipid is the most frequently used lipid at Seattle. It can be given in higher doses than Omegaven which is important nutritionally; omegaven is generally given at only 1 to 1.5 mg/kg/day and used for treatment of IFALD.
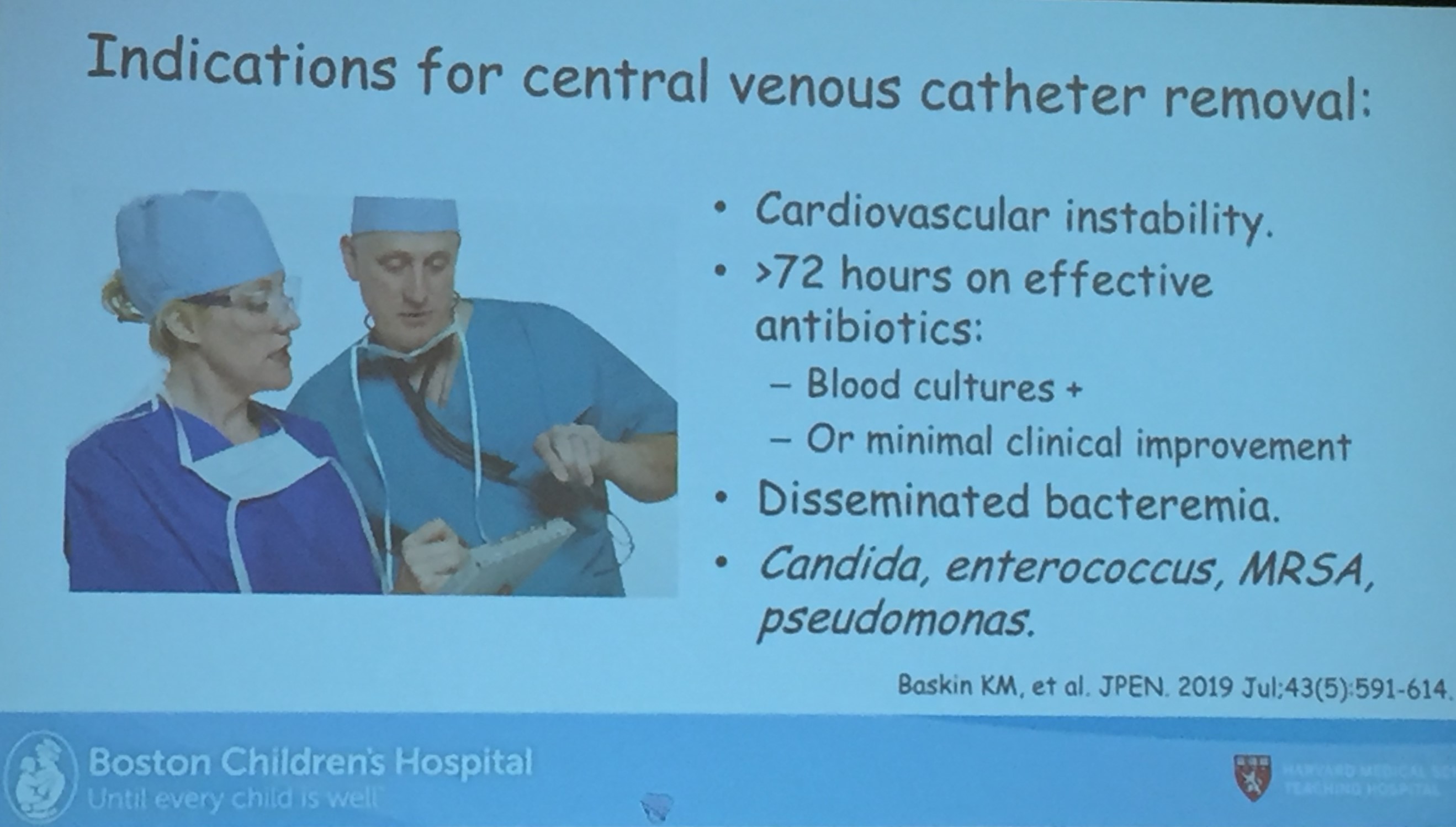
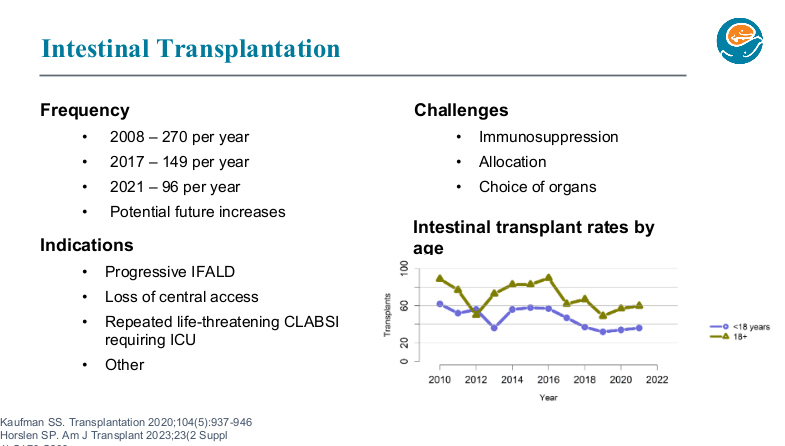
- Long term outcomes are just as good with chronic TPN as with intestinal transplantation. So, referral generally needed based on complications like losing central access (3 of 4 upper central sites) or progressive liver disease.
Diet Pointers:
- -In infancy, standard formula and breastmilk are preferred and thought to help with adaptation. Some infants need elemental diets but it is not routinely given across the board (some other institutions feel strongly about using elemental diets, but there is limited data)
- -Kids with short bowel syndrome may tolerate volume better than concentration
- -The Seattle program strictly restricts sweet tasting food/drink for first 3-4 years of life to help educate the child’s palate and recommends limiting these food/drink for IF patients in general.
- -Addition of solid foods usually helps with stoma output
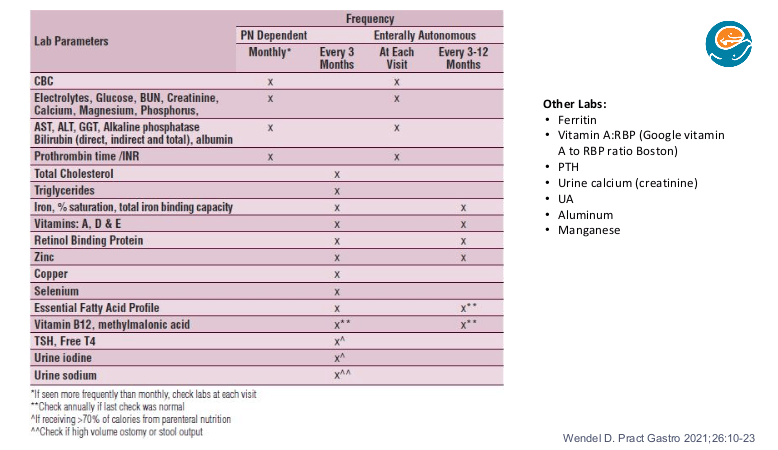
Nutrient Monitoring Pointers:
- Usually best to batch them all lab tests for micronutrients [many micronutrients are affected by inflammation and this may affect timing of lab testing]
- At Seattle, aluminum and manganese are not routinely checked as they are contaminants in PN that cannot be removed
- Serum thyroid testing is a marker for iodine deficiency in patients receiving most of their calories from PN (>70) which may be more frequent now that betadine is not used for dressing changes. Their goal for urine iodine is >100 (can be treated with ultra-dilute
dpotassium iodide which needs to be compounded by the pharmacy) - When testing for EFA (essential fatty acid) deficiency, lipids should be off for 4 hrs (or more). Urinalysis is checked to monitor for chronic kidney disease. Urine sodium goal is >30 and is checked quarterly in patients with high ostomy output or excessive rectal stool output with poor growth.
- Hypokalemia may be a sign of total body sodium depletion due to the kidneys dumping potassium to conserve sodium










Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.