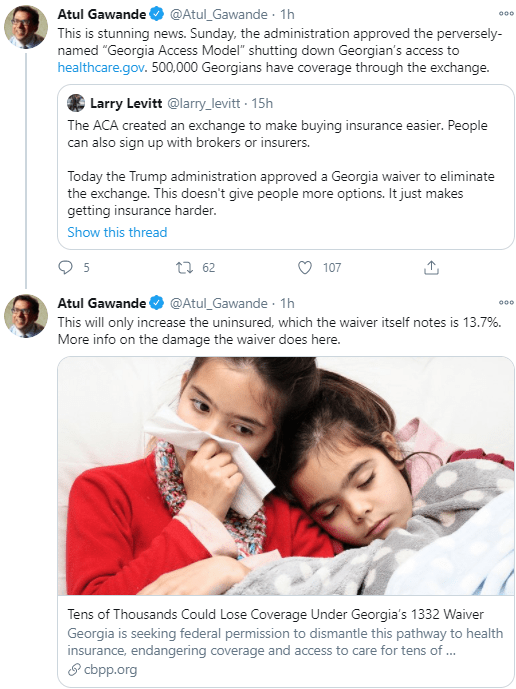
AAP News (Nov 2021): First oral pan-genotypic HCV drugs approved for children as young as 3 years
“The Food and Drug Administration (FDA) has approved Epclusa (sofosbuvir and velpatasvir) and Mavyret (glecaprevir and pibrentasvir) for treatment of chronic hepatitis C virus (HCV) infection in pediatric patients as young as 3 years. These products are the first all-oral, pan-genotypic (genotypes 1-6) HCV treatment regimens for pediatric patients 3 years and older…Both products are available as oral tablets and as newly approved oral pellets in dosing strengths suitable to accommodate the recommended weight-based dosing in pediatric patients.”
From HCVGuidelines.org (updated September 2021):

The following images are from Abbvie patient-provider handouts. I do not receive any funding from any pharmaceutical company but think these instructions are useful.



Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.