M Earl et al. J Pediatr 2025; 285: 114673. Open Access! General and Autism-Related Neurodevelopmental Difficulties in Biliary Atresia
Methods: In this single-center, observational study, the authors deployed an anonymized survey of outcomes that was completed by 107 parents of children with BA who were younger than age 12 years. A detailed assessment of general neurodevelopment (Mullens Scale of Early Learning and Vineland Adaptive Behavior Scale) was carried out in 50 infants younger than 5 years old, and emerging autistic traits (Autism Diagnostic Observation Schedule) were assessed in those eligible. There were 93 matched controls.
Key findings:
- Neurodevelopmental concerns were raised by 37% of parents
- 47% of children required support from at least 1 service (such as speech and language therapy physiotherapy, play therapy, or seen a clinical psychologist), and a further 42% (n = 45) had used more than 1 service. The most accessed service was speech and language therapy (20%, n = 10)
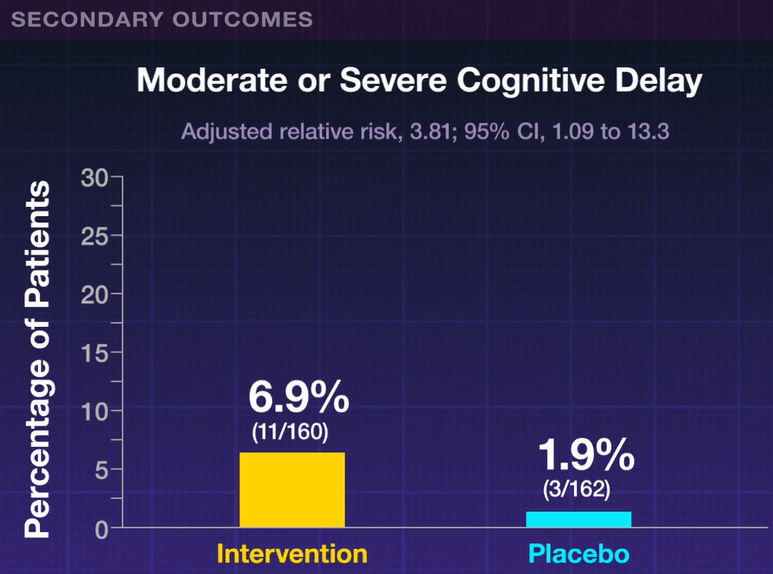
- A clinical or research diagnosis of autism was made in 30% of 35 children >2 years old
- Early surgical intervention and faster clearance of jaundice after surgery was associated with better general neurodevelopmental outcomes (F = 2.428, P = .042) but not with the presence of emerging autistic traits
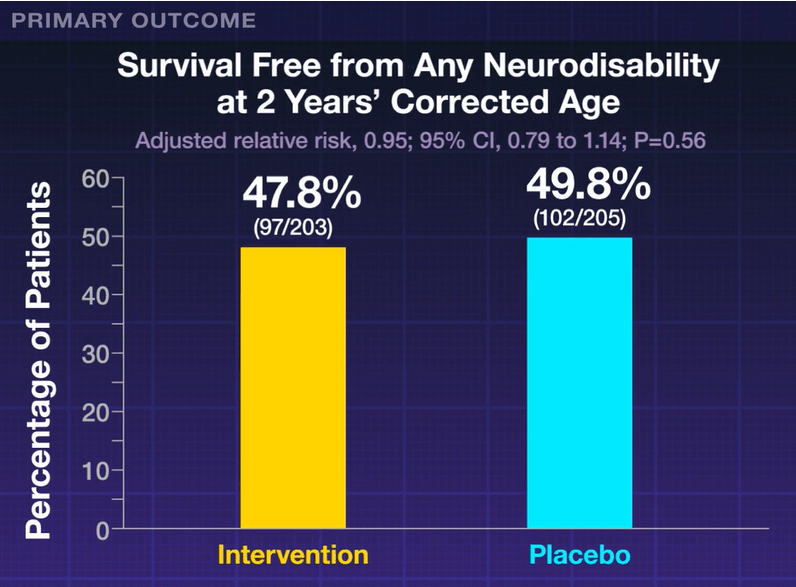
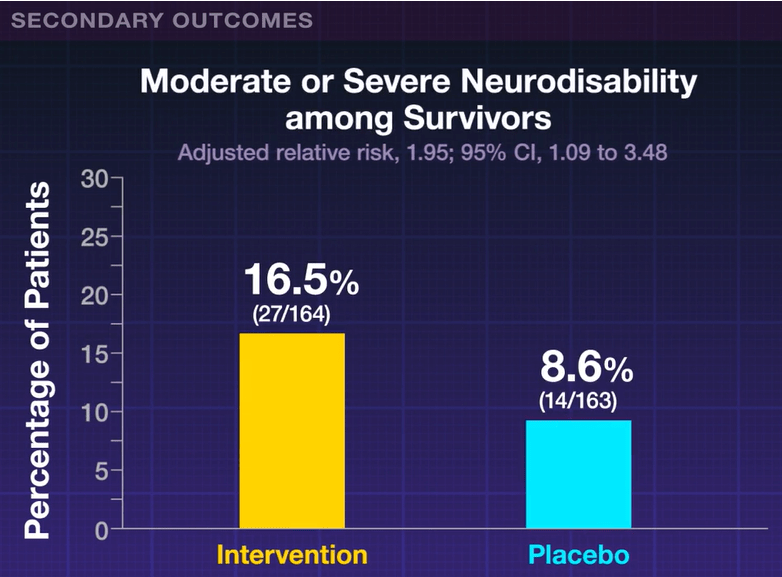
My take: High levels of neurodevelopmental difficulties occur in children with BA.

Related blog posts: