D Ahuja et al. Clin Gastroenterol Hepatol 2026. 24: 794-804. Comparative Safety of Janus Kinase Inhibitors vs Tumor Necrosis Factor Antagonists in Patients With Inflammatory Bowel Diseases
Methods: This retrospective cohort study used an administrative claims database and identified patients with IBD who were new users of either JAK inhibitors (n=856) or TNF antagonists (n=9422) between 2016 and 2023. Mean age was 45 years.
Key findings:
- There was no difference in the risk of VTE (1.3 vs 1.2; HR, 0.66; 95% CI, 0.28–1.57) and MACE (0.4 vs 0.7; HR, 0.50; 95% CI, 0.19–1.30)


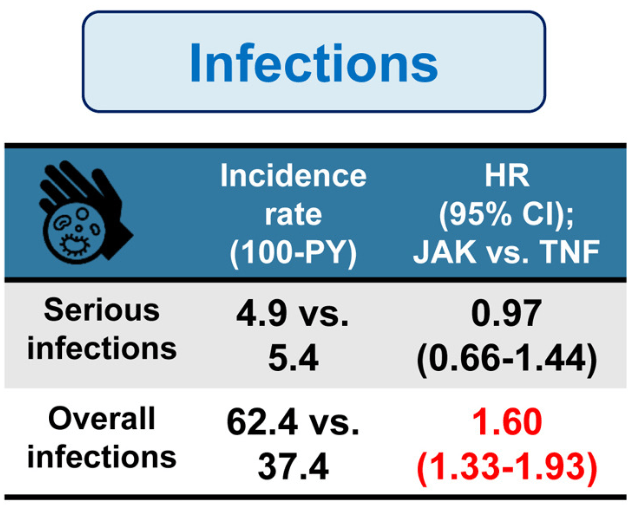
- There was no difference in the risk of serious infections (4.9 vs 5.4; HR, 0.97); however, JAK inhibitors were associated with an increase risk of overall infections (incidence rate, 62.4 per 100 person-years [PY] vs 37.4 per 100 PY; hazard ratio [HR], 1.60)
The authors note that their findings differ from the ORAL study (Ytterberg et al. NEJM 2022; 386: 316-326.) which showed higher risk of MACE in patients receiving tofacitinib. In the current study, even in patients deemed to be at higher risk for MACE (age >50 years with at least 1 cardiovascular risk factor), JAK inhibitors were associated with lower incidence and risk of MACE compared with TNF antagonists (IR per 1000 PY, 0.4 vs 2.1, HR 0.10).
My take (borrowed from the authors) “It is unlikely that JAK inhibitors are associated with higher risk of VTE and MACE compared with TNF antagonists in most patients with IBD.”
Related blog posts:
- Safety of JAK Inhibitors Compared to Anti-TNF Agents
- Prior Exposure to TNF Antagonists May Increase Response to JAK Inhibitors in Patients with Ulcerative Colitis
- FDA Slaps Restrictions on JAK Inhibitors Over Serious Safety Risks
- Dr. Joel Rosh: Positioning Therapies for Pediatric Ulcerative Colitis
- Lipid Changes with IBD Medications
