A Yerushalmy-Feler A et al. Clinical Gastroenterology and Hepatology, 2026 (In press); Upadacitinib Maintenance Therapy in Pediatric Ulcerative Colitis: 52-Week Multicenter Study From the Porto Group of the European Society for Paediatric Gastroenterology, Hepatology, and Nutrition
Background/Methods: There is limited data on the use of upadacitinib for pediatric inflammatory bowel disease. This retrospective data from 35 European centers analyzed its effectiveness in 105 children (95 with UC and 10 with IBD-U). Prior to upadacitinib, 103 of 105 children (98%) were treated with biologic therapies and 79 (75%) with ≥2 biologics. The induction dose was 45 mg in 86% of cohort; the maintenance dose was 30 mg in 87% (only 2 patients received 45 mg maintenance). Mean age at IBD diagnosis was 11.3 yrs and mean age at start of upadacitinib was 14.6 yrs. 65% of study participants had a pancolitis.
Key findings:
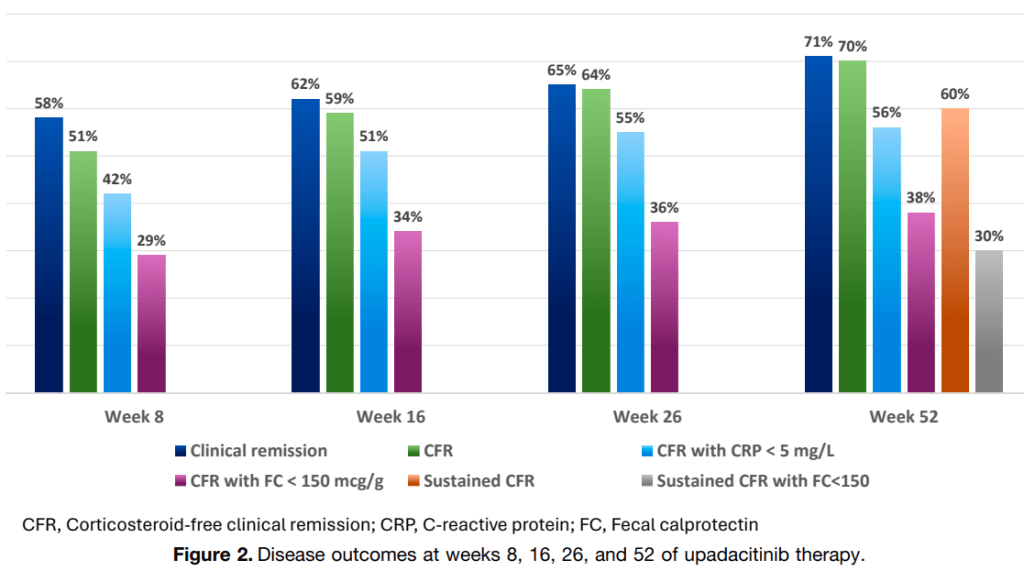
- Clinical remission and corticosteroid-free clinical remission (CFR) were observed after 8 weeks in 61 (58%) and 53 (51%) children, respectively
- By week 52, 75 children (71%) achieved clinical remission, 73 (70%) achieved CFR, and sustained CFR in 63 (60%); CFR with FC <150 mcg/g was reached 30 of 80 (38%) (29% of the ITT group)
- Adverse effects: There were two serious AEs: an appendiceal neuroendocrine tumor and cytomegalovirus colitis. The most frequent AEs were hyperlipidemia (n = 20), infections (n = 18), and acne (n = 14)
Predictors of response: “The baseline variables that were associated with achieving sustained CFR were prior failure of fewer biologic agents (≤2 vs >2), a lower PUCAI score, absence of corticosteroid therapy, and higher serum hemoglobin and albumin levels.”
Age: “Our findings suggest that upadacitinib provides comparable effectiveness in younger children weighing <40 kg, supporting its therapeutic potential across a broader pediatric age and weight range.”
My take: Upadacitinib is an important therapy for ulcerative colitis in the pediatric age range and in adults. It is effective in all age groups. Also, young children can now be prescribed a liquid version (Rinvoq LQ) which requires twice daily dosing (rather than once a day). Some patients who do not respond adequately or lose response may benefit from higher dosing.
Related blog posts:
- “Real-World” Experience: High Dose Upadacitinib Recaptures IBD Response
- Long-term Efficacy and Safety of Upadacitinib for Ulcerative Colitis
- Dr. Joel Rosh: Positioning Therapies for Pediatric Ulcerative Colitis
- More Data: Upadacitinib “is Effective and Safe” Plus 2 in Kids
- Pediatric Guidelines for Ulcerative Colitis (Part 1)
- Pediatric Guidelines for Ulcerative Colitis (Part 2: Acute Severe Colitis)
- Comparative Evidence and Positioning Advance Therapies for Inflammatory Bowel Disease)
- AGA Living Guideline for Moderate-to-Severe Ulcerative Colitis –The Good and The Bad
- Upadacitinib vs Risankizumab for Crohn’s Disease
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.

Would use of Rinvoq as the initial agent in treating IBD be more beneficial than using corticosteroids or biologics?
I don’t think there is enough data to know with certainty about whether it would be more beneficial than all of the biologics (particularly in children). In those with ulcerative colitis or Crohn’s disease (without small bowel disease), my expectation is that it would likely to comprare favorably with biologics in terms of treatment response. As for corticosteroids, these are not effective long-term therapy. An agent like Rinvoq as well as the anti-TNF therapies which also have rapid onset could help avoid steroids and their adverse effects.