D Turner et al. J Pediatr Gastroenterol Nutr. 2026;82:867–894. Reshaping study design for faster extrapolation‐baseddrug approval in pediatric inflammatory boweldiseases: An ESPGHAN–NASPGHAN position paper


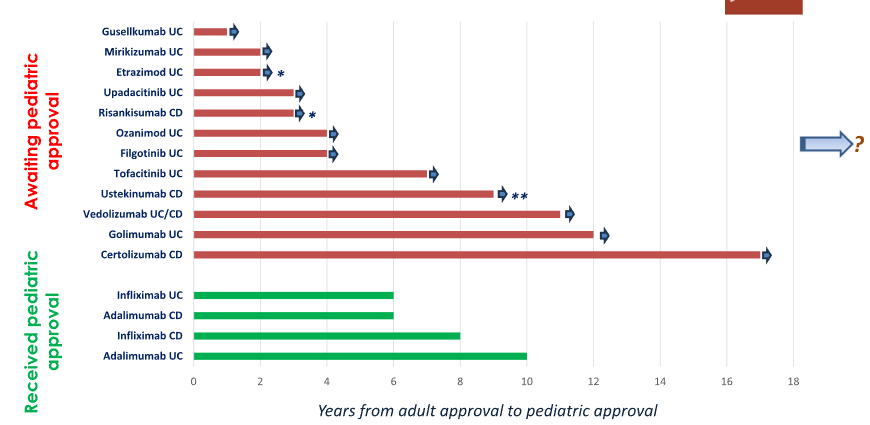
“It has been 11 years since the adult approval of vedolizumab and 9 years since the approval of ustekinumab, yet neither is currently approved for use in children (Table 1). Instead of taking the necessary steps to shorten this unacceptable delay, there are now extra hurdles for clinical trials such as three ileocolonoscopies over 1 year in both pediatric CD and UC, as suggested by the FDA…”
Addendum: See comment by Matthew Kowalik, MD who is the Director (Acting) of Division of Gastroenterology for FDA/CDER/OND/OII. There has been recent approval recent of Stelara (ustekinumab) for the treatment of pediatric patients 2 years and older with moderately to severely active Crohn’s disease. Here’s a link: Johnson & Johnson (JNJ) Gains FDA Approval for Pediatric Crohn’s Disease Treatment
“Pediatric extrapolation is based on assessing relevant similarities in disease characteristics, drug pharmacology, and treatment response between the target pediatric population and adults or other reference pediatric populations. While, historically, extrapolating safety data was considered unacceptable, the recent International Council for Harmonization (ICH) E11A Guideline on pediatric extrapolation that has been endorsed in 2024 by the EMA”[European Medicines Agency (EMA) ]

Selected Summary Statements (total of 24 are in the report):
- 1.There is no evidence that differences in pathogenesis and pathobiology between pediatric and adult patients with IBD are associated with differences in response to pharmaceutical therapies, except for monogenic disease (agreement 21/22).
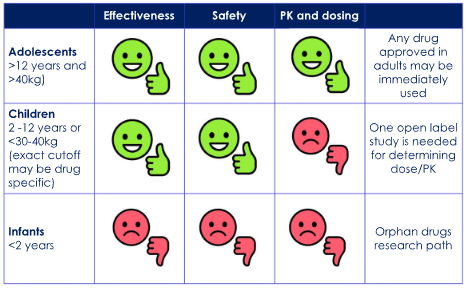
- 2.In general, efficacy and drug-related safety outcomes in children older than 2 years are comparable to adults (agreement 22/22).
- 9. Optimal dosing should be used in all study arms in pediatric trials. Placebo, sham, or doses demonstrated to be subtherapeutic in prior studies should not be permitted. They are unethical in children, reduce feasibility of enrollment, and are not expected to be informative given the underpowered sample size of pediatric studies (agreement 22/22).
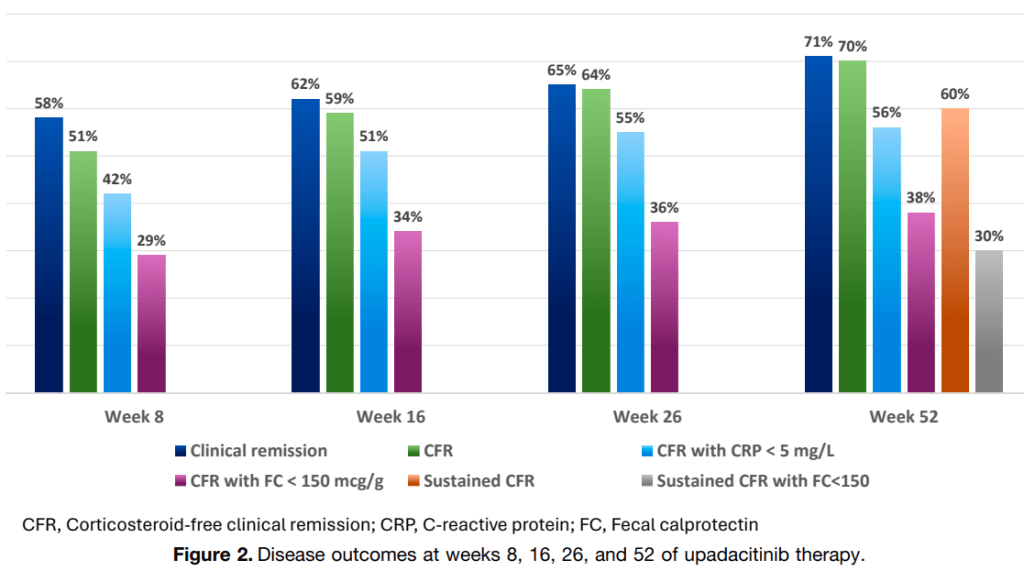
- 13.Ileocolonoscopic assessment is the gold standard for assessing mucosal healing (MH) and should be required at most twice in each study: that is, at baseline and study end (agreement 22/22).
- 14. Noninvasive objective measures, including serum and fecal biomarkers, magnetic resonance enterography (MRE), and/or intestinal ultrasound (IUS), should be used for assessing postinduction interim therapeutic response between the two ileocolonoscopies rather than requiring a third ileocolonoscopy (agreement 22/22).
My take (borrowed from the authors):
- “While it is paramount to achieve a precise and comprehensive approval process for new drugs in pediatric IBD, it is equally important to expedite the process, so children and adolescents are not denied effective treatment available for adults with IBD…”
- “Under these assumptions, future trial designs should be single-arm and open-label to focus on dosing and pharmacokinetics in children weighing <30–40 kg while mandating long-term safety registries, disease- and not necessarily drug-specific. Data should be supported by meticulously collected real-world evidence. Pediatric data must be collected as soon as a confident signal of efficacy and safety is achieved in adult studies…”
- “This will resolve the current paradox in which children have the most severe and extensive disease and the highest efficacy of drugs, yet very limited access to these drugs.”
Related blog posts: