BE Sands et al. Clin Gastroenterol Hepatol 2026; 23: 525-534. Open Access! A Phase 2 Study of MORF-057, an Oral α4β7 Integrin Inhibitor in Moderately to Severely Active Ulcerative Colitis
Background: MORF-057 is an orally administered small-molecule drug that inhibits α4β7 integrin-mediated recruitment of α4β7-expressing lymphocytes to the gut. It has a similar target as vedolizumab, a monoclonal anti-integrin antibody used to treat ulcerative colitis (approved in 2014), but it requires parenteral administration.
Methods: This open-label, phase 2a, single-arm, multicenter trial (EMERALD-1) comprised a 6-week screening period, a 52-week active treatment period (including a 12-week induction period and 40-week maintenance period), and a 4-week safety follow-up period. Of the 35 participants enrolled in the main cohort, 18 participants received 100 mg of oral MORF-057 twice daily for the entire treatment period.
The primary efficacy endpoint was a change in the Robarts Histopathology
Index (RHI) score from baseline to week 12. “RHI was chosen as the primary efficacy assessment because it is an objective measure that can be assessed in a blinded fashion, which was critical given the open label study design. RHI also allows for a deeper, more quantitative probe than endoscopy.”
Key findings:
- MORF-057 was well tolerated, and no treatment-emergent serious adverse events were
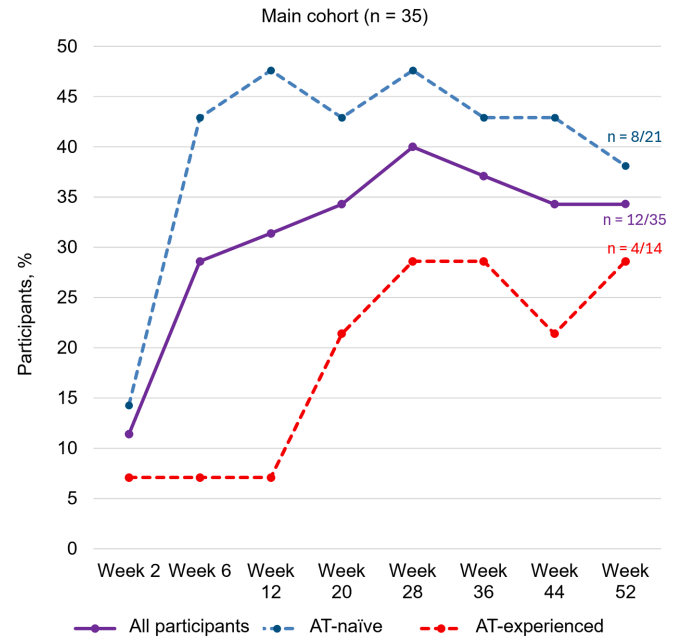
observed - At week 12, participants (n= 35) exhibited a mean change from baseline in RHI
score of ‒6.4 (standard deviation, 11.2). Additionally, 22.9% of participants (8/35) achieved
RHI remission (RHI score ≤3) - In participants with evaluable data (n=18), the effects of MORF-057 on pharmacokinetics, pharmacodynamics, and clinical efficacy were achieved at week 12 and remained consistent to week 52
My take: This small open label study shows that an oral medication targeting α4β7 integrin has potential as an effective therapy for ulcerative colitis. If effective, then it would be important to understand how it compares to vedolizumab.
Related blog post: In Trials: An Oral IL-23 Antagonist Peptide
